Retinal arterial macroaneurysms (RAMs) are acquired vascular dilations that usually occur in the first three bifurcations of retinal arteries. They typically present unilaterally in females over the age of 60 and are commonly associated with systemic hypertension. RAMs are generally asymptomatic; however, hemorrhaging may cause an acute loss of vision. Prolonged edema and hemorrhaging may lead to degeneration of the RPE and photoreceptors and thus, a permanent loss of vision within two weeks after the onset of the hemorrhaging.
In the following case, a presumed RAM resulted in this patient’s loss of central vision.
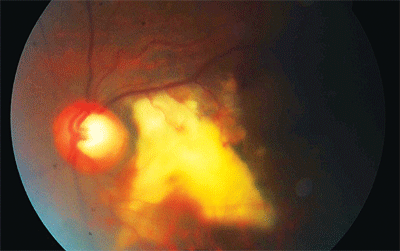 |
| This patient presented with signs in his left eye that seemed to point to a retinal arterial macroaneurysm. |
Patient History
A 73-year-old black male presented to the University Eye Instuitute with complaints of seeing a “curtain” or “veil” over his left eye. He reported that the vision in his left eye had progressively decreased at all distances over the past six years. The patient denied any headaches, flashes, floaters, ocular trauma or infections.
The patient had been diagnosed with primary open-angle glaucoma seven years prior. But, he self-discontinued the prescribed medication after just one year of use.
The patient’s last physical exam was about four months prior. He reported no chronic or infectious diseases. He denied diabetes, hypertension, cardiovascular disease, cancer, tuberculosis, shortness of breath and cough. He also denied any acquired or iatrogenic immune problem, hospitalizations or major injuries. He reported that he was not taking any medications and had no known drug allergies. He said he did not smoke, drink or use steroids. The patient did, however, report that in a past tour of military duty, he received multiple unknown vaccines and traveled extensively.
The patient’s family history was negative for diabetes, cardiovascular disease, cancer and glaucoma.
Diagnostic Data
The patient’s blood pressure measured 150/98mm Hg in the left arm while sitting. Entering visual acuity was 20/40- O.D. with pinhole and finger counting at one meter O.S. Spectacle correction in the right eye was +1.00D -1.00 X 095 with a +2.50D add, which resulted in 20/40+ vision. The left eye revealed no red reflex with retinoscopy secondary to small pupils and a posterior subcapsular cataract.
Pupils were equal in size, with a positive afferent pupillary defect in the left eye. Confrontation fields were full O.D. and restricted in the superonasal field O.S., and extraocular muscles were smooth and unrestricted O.U. Intraocular pressure measured 13mm Hg O.U., with pachymetry readings of 549µm O.U. Biomicroscopy revealed corneal guttae with edema, and anterior chambers were deep and quiet in both eyes. Grade 2+ cortical and nuclear cataracts were noted in both eyes, and a grade 2+ posterior subcapsular cataract was noted in the left eye.
The posterior segment of the left eye, when viewed by indirect ophthalmoscopy and biomicroscopy, revealed a white, feathery growth temporal to the optic disc that obscured the macula. Presumed feeder vessels were apparent, and a horseshoe-shaped subretinal and preretinal hemorrhage was located temporal to the lesion. No retinal breaks or detachments were noted.
Pertinent fundus findings in the right eye revealed a 2.2mm vertical diameter disc with a large cup-to-disc ratio of 0.85 x 0.85 and no inferior rim. The right eye had a faint foveal reflex and even pigmentation of the macula. The left disc was also 2.2mm in vertical diameter. The cup-to-disc ratio in the left eye was judged at 0.65 x 0.65 with difficulty because of the cataracts. The inferior rim also appeared to be absent in the left eye. Both discs had bayoneting and bean potting with good coloration of the remaining rim tissue.
Tentative Diagnosis
The assessment at this visit was an unknown retinal lesion. The patient was sent to a retinal specialist for a consult. We recommended that the patient report to his primary care physician for a physical and an evaluation for hypertension.
Follow-Up
At a one-week follow-up exam with a retinal specialist, the distance acuity of the right eye improved to 20/20- with correction. The left eye had a positive afferent pupillary defect and a vitreous hemorrhage. Fluorescein angiography in the left eye revealed sharp borders in the area of the subretinal lesion and no choroidal neovascular membrane.
Diagnosis
Gross observation and angiography findings led to a diagnosis of thrombosed retinal arterial macroaneurysm with subretinal and intraretinal hemorrhages. The patient was scheduled for follow up with the retinal clinic in two months.
Further Follow-Up and Treatment
The partner of the first retinal specialist evaluated the patient two months later. The specialist indicated that the diagnosis of a RAM was adequate, though uncertain because the focal aneurysm was not detectable. A photo review from six years earlier revealed an inferior nasal venous out-pouching close to the left optic nerve in the area of the current lesion. This led to speculation of the possibility of the lesion being a retinal venous macroaneurism.
A glaucoma evaluation was also initiated, and optic nerve assessment assisted with photos revealed a cup-to-disc ratio of 0.90 x 0.95 O.D. and 0.90 x 0.90 O.S. Thinning of the inferior rim in the right eye was noted, as was progressive cupping in both eyes. Visual fields revealed nasal defects approaching the paracentral field in the right eye and an overall depression that was densest superiorly in the left eye.
The patient was started on Xalatan (latanoprost, Pfizer) one drop before bedtime in each eye. Due to inadequacy of pressure lowering, a right eye trial of Timoptic XE 0.5% (timolol maleate, Merck) 1gtt in the morning was initiated in an attempt to lower the pressures more. This therapy was successful, and the patient was placed on Xalatan 1gtt O.U. before bedtime and Timoptic XE 0.5% 1gtt O.U. in the morning.
Since the patient will never regain vision in his left eye, he was educated about the importance of preserving the vision remaining in his right eye. We stressed the importance of continued compliance with glaucoma drops and follow-up exams. Also, we recommended polycarbonate protective eyewear for full-time wear.
In addition to treatment of the RAM and related ocular sequelae, it is vital to rule out and treat any causative systemic condition. A timely and proper referral for a pointed systemic work-up is essential for proper care, so the patient was sent for a full physical to rule out any possible associated systemic problems.
We sent a letter to his primary care physician to detail those systemic conditions to be ruled out. Because of the high association with hypertension, it is critical for the patient to be evaluated and treated for any possible hypertension. RAMs may also be associated with dyslipidemia, embolic and cardiovascular disease so the patient should be evaluated for these conditions also. Generally, this is all the medical testing required. In cases of an unsure diagnosis, diabetes should be ruled out.
Discussion
As previously mentioned, a retinal arterial macroaneurysm is an acquired vascular dilation that usually occurs in the first three bifurcations of retinal arteries or at arteriovenous crossings.1-4 The aneurysm weakens the vessel wall, which causes local damage. A hemorrhage follows in 50% of RAM occurrences.5 RAMs are generally asymptomatic; however, the most common presenting visual symptom experienced with a RAM is acute loss of vision secondary to hemorrhage.5
Typically, a RAM presents unilaterally (in more than 90% of cases), and the occurrence shows a marked predilection for females over the age of 60.5-6 RAMs are associated with systemic hypertension in more than 75% of patients.5,7,8 Other systemic implications include arteriosclerosis, cardiovascular disease, dyslipidemia and embolic disease.4,5,7,8,9
The histopathogenesis of a RAM can be directly correlated to physical changes associated with the systemic conditions described above. Damage to the arterial wall due to a loss of muscle tissue and replacement of fibrotic tissue decreases the elasticity in the artery. This damage, combined with increased intraluminal pressure, can cause arterial focal out-pouching. Other factors contributing to a RAM include retinal emboli, as well as retinal arteriolar bifurcations and venous crossings, which cause blood turbulence.5,9,10 Microaneurysms found in diabetic retinopathy differ from macroaneurysms; the capillaries and small venuoles are affected, rather than the arteries.8 Retinal venous macroaneurysms are reported, although they are significantly more rare than RAMs.11
RAMs are several times the size of the original artery width. The average diameter of a RAM is 280mm, but rare reports indicate they can reach 3,000mm.9,12 Generally, however, RAMs do not grow to such large sizes before rupturing.9,12 Involution of a RAM occurs after thrombosis and fibrosis. Occasionally, a Z-shaped kink in the artery remains after involution, leading the observer to the original site of the RAM.9
Other common fundus observations and sequelae associated with RAMs include hemorrhaging, exudates, edema and neurosensory retinal detachments.5,9 Hemorrhaging secondary to a RAM may occur subretinally, intraretinally, preretinally or intravitreally. Blood and exudates may move anteriorly or posteriorly through the retinal layers.5,9 Intraretinal yellow or white exudative lipid deposits are usually found encircling the RAM. Exudates also tend to collect in the macular layer, no matter the location of the RAM. Retinal edema can occur secondary to leakage from the RAM or from small damaged vessels surrounding the RAM.5,9 Prolonged edema and hemorrhaging can cause degeneration of the RPE and photoreceptors, as well as permanent loss of vision within two weeks of onset.1,8
Visual prognosis with symptomatic RAMs depends on the amount of macular damage. A submacular hemorrhage causes the largest visual deficit.2 Sub-RPE hemorrhages near or at the macula lead to the worst visual outcome, due to degeneration of the RPE and photoreceptors, as well as subsequent scarring. In short, the less time the edema, blood and exudates reside in the macula, the less the permanent visual deficit.9 Additional sight-threatening sequelae associated with RAMs, although rare, include macular hole, epiretinal membrane, neurosensory retinal detachment and angle-closure glaucoma.3,13
Ancillary tests aid in the diagnosis of a RAM. With intravenous fluorescein angiography, early filling of the aneurysm is observed, as well as late leakage and pooling of the lesion. If hemorrhaging is present, indocyanine green angiography allows for better observation of the aneurysm and lesion. Thus, IVFA and ICG help to differentiate between other clinical diagnoses, such as AMD or malignant melanoma.12 Angiography is paramount in the diagnosis of a RAM.
Differential diagnoses for RAM include Coats’ disease, Leber’s miliary aneurysms, angiomatosis retinae, retinal telengiectasia, diabetic retinopathy, retinal vein occlusion, ocular granuloma, retinal vein occlusion (including venous macroaneurysms) and age-related macular degeneration, as well as tumors, such as retinal capillary hemangioma, retinal cavernous hemangioma or malignant melanoma.2,12
Characteristic ophthalmoscopic and angiographic findings typically distinguish RAM from these other lesions, but the presence of extensive hemorrhage can make the diagnosis more challenging. The natural progression of most macroaneurysms includes spontaneous resolution without visual loss. Visual loss may occur secondary to related macular edema, exudate, hemorrhage and neurosensory retinal detachment, however.
Current treatment options of RAM include laser photocoagulation, laser photodisruption, vitrectomy and intravitreal tissue plasminogen activator (t-PA). Photocoagulation with an argon laser seals off the RAM or causes involution. The laser treatment can be directly aimed at the RAM, or indirect laser treatment can be used—but, indirect treatment is likely to provoke a hemorrhage.1 Nd:YAG laser photodisruption allows for the release of trapped blood into the vitreous, preventing damage to the RPE and photoreceptors.2,14 Laser treatment may expedite visual recovery. A vitrectomy can remove preretinal collected blood, and, at the same time, release a subretinal hemorrhage.1 Intravitreal injection of t-PA can break down a submacular hemorrhage by lysing the fibrin that is clotting the blood and inflicting damage.4,15 After the subretinal clotted blood has dissolved, it can be aspirated, or an intraocular gas bubble can pneumatically displace the submacular blood.4,6,15,16
Unfortunately, the patient has been lost to follow-up again. After numerous phone calls and letters, he is scheduled to return to clinic as this case is being written. Hopefully, his compliance will improve.
Dr. Pate is a clinical associate professor at the University of Houston College of Optometry. Dr. Cline is a graduate of the University of Houston College of Optometry. She practices in Pearland, Texas.
1. Zhao P, Hyashi H, Oshima K, et al. Vitrectomy for macular hemorrhage assocated with retinal arterial macroaneurysm. Ophthalmology. 2000 Mar;107(3):613-17.
2. Tonotsuka T, Imai M, Saito K, Iijima H. Jpn J Ophthalmol. 2003 Sep-Oct;47(5):498-502.
3. Arthur SN, Mason J, Roberts B, Girkin CA. Secondary acute angle-closure glaucoma associated with vitreous hemorrhage after ruptured retinal arterial macroaneurysm. Am J Ophthalmol. 2004 Oct;138(4):682-3.
4. Humayun M, Lewis H, Flynn HW Jr, et al. Management of submacular hemorrhage associated with retinal arterial macroaneurysms. Am J Opthalmol. 1998 Sep;126(3):358-61.
5. Rabb MF, Gagliano DA, Teske MP. Retinal arterial macroaneurysms. Surv Ophthalmol. 1998 Sep-Oct;33(2):73-96.
6. Wu TT, Sheu SJ. Intravitreal plasminogen activator and pneumatic displacement of submacular hemorrhage secondary to retinal artery macroaneurysm. J Ocul Pharmacol Ther. 2005 Feb;21(1):62-7.
7. Sekuri C, Kayikcioglu M, Kayikcioglu O. Retinal artery macroaneurysm as initial presentation of hypertension. Int J Cardiol. 2004 Jan;93(1):87-8.
8. Matthews D. Retinal macroaneurysms secondary to systemic hypertension causing retinal hemorrhage. J Am Optom Assoc. 1986 Feb;57(2): 26-9.
9. Adameczyk DT, Olivares GE, Petito GT. Retinal arterial macroaneurysm: a longitudinal case study. J Am Optom Assoc. 1989 Nov;60(11):840-5.
10. Cahill MT, Flitcroft DI, Acheson RW. Fundal findings preceeding retinal artery macroaneurysm. Eye. 1998;12(6):1023-4.
11. Khairallah M, Ladjimi A, Messaoud R, et al. Retinal venous macroaneurysm associated with premacular hemorrhage. Ophthalmic Surg Lasers. 1999 Mar;30(3):226-8.
12. Ohno-Matsui K, Hayano M, Futagami S, et al. Spontaneous involution of a large retinal arterial macroaneurysm. Acta Ophthalmol Scand. 2000 Feb;78(1):114-7.
13. Tashimo A, Mitamura Y, Ohtsuka K, et al. Macular hole formation following ruptured retinal arterial macroaneurysm. Am J Ophthalmol. 2003 Apr;135(4):487-9.
14. Iijima H, Satoh S, Tsukahara S. Nd:YAG laser photodisruption for preretinal hemorrhage due to retinal macroaneurysm. Retina. 1998;18(5):430-4.
15. Kokame G. Vitreous hemorrhage after intravitreal tissue plasminogen activator (t-PA) and pneumatic displacement of submacular hemorrhage. Am J Ophthalmol. 2000 Apr;129(4):546-7.
16. Hassan AS, Johnson MW, Schneiderman TE, et al. Management of submacular hemorrhage with intravitreous tissue plasminogen activator injection and pneumatic displacement. Ophthalmology. 1999 Oct;106(10):1900-7.

