and Susannah Marcus-Freeman, O.D.

Can you spot the difference between a benign tumor and a malignant one? Benign and malignant iris tumors may present similarly on routine clinical examination, but it is important to differentiate between the two to ensure proper management. The patients life may depend on it.
Until now, photodocumentation and serial observation for growth have been the gold standards for monitoring patients with suspicious lesions. But, recent advances in technology may be more helpful in differentiating iris tumors and other anterior segment lesions.
History
Walter, a 76-year-old white male, presented complaining of an inability to see small print and occasional blurred vision.
His ocular history was significant for moderate cataracts O.U., and he denied the use of any ophthalmic medications. His medical history was significant for tinnitus, hyperlipidemia, prostate cancer and arthritis. His prostate cancer was treated via radical prostatectomy with no history of recurrence. Medications included simvastatin 40mg q.d. and vardenafil hcl 20mg q.d. p.r.n.
Diagnostic Data
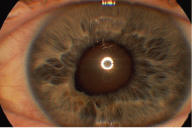
1. Elevated iris lesion infero-nasal to the left pupil with homogenous color and ectropion uvea.
Walters best-corrected visual acuity was 20/30+1 O.D. and 20/30 O.S. Biomicroscopy revealed normal corneas, normal anterior chambers, and +2 nuclear changes to the lens O.U. The iris of the right eye was flat and intact, but the left iris showed an ectropic/prolapsed frill adjacent to a solid-looking, grayish lesion inferonasal to the pupil (figure 1). The elevated area was homogenous in pigmentation with the rest of the visible iris. Pupils measured 4mm O.U. The right pupil was round, but the left pupil was slightly distorted inferonasally. Both pupils were reactive to light, with no afferent pupillary defect present in either eye.
Motilities and confrontations were normal O.U. Intraocular pressure (IOP) measured 14mm Hg O.U. Dilated funduscopic exam revealed bilateral posterior vitreous detachments and an epiretinal membrane (ERM) in the left eye. All other structures of the posterior segment were unremarkable. Anterior segment photos, optical coherence tomography (OCT), and B-scan were ordered. Gonioscopy was also scheduled.
OCT revealed an area of hyperreflective ectropion uvea with posterior signal blockage (figures 2 and 3). A slight thickening of the stroma in the area of the lesion was also evident. The lesion could not be detected with B-scan due to its small size. Ultrasound biomicroscopy (UBM) revealed a 0.7mm high, slightly thickened lesion of the anterior iris border with no apparent angle involvement (figure 4).
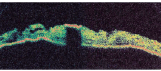
2. OCT showing a caliper thickness of 631m.
Diagnosis
We initially diagnosed Walter with a benign iris lesion, but were unsure due to the patients history of cancer. We wanted to rule out malignancy or metastasis, so we scheduled him for follow up in one week for photodocumentation.
Treatment and Follow-Up
At follow-up, we saw no changes in his iris appearance. Best-corrected visual acuity remained nearly 20/30 in each eye. Walters wife accompanied him to this visit and reported that her husband had been a professional boxer many years ago.
We referred him to a general ophthalmology clinic, where he was tentatively diagnosed with an iris cyst and scheduled for ultrasound biomicroscopy. Walter was scheduled for a follow-up visit in four months.
3. OCT also revealed hyperreflective iris pigment epithelium.
At four months, his best-corrected visual acuity measured 20/40 O.U., and there were no changes to the iris lesion. The UBM revealed a 0.7mm high, slightly thickened lesion of the anterior iris border with no apparent angle involvement. Gonioscopy of the left eye was performed at this visit and revealed open angles with trace pigment and no tumor involvement.
Although the lesion appeared stable, melanoma had not yet been definitively ruled out. A referral to a retinal specialist was made, and his opinion was that the lesion seemed nonmalignant based on clinical appearance; periodic evaluation on a six month interval was recommended to monitor for possible changes. Since the lesion appeared unchanged at the most recent visit, Walter was released for annual examination thereafter.
Discussion

4. UBM shows a small, solid, elevated mass at the anterior iris border.
Presumably benign lesions (e.g., nevi) are commonly encountered in patient care. Nevertheless, iris lesions can be hard to differentiate purely by clinical observation because they can display similar growth and behavior patterns.
The most common anterior segment neoplasms can be divided into four main categoriescysts, nevi, melanomas and metastases:1
Iris cysts, which can be further divided into two categories: primary, which have no recognizable etiology, and secondary, which have a recognizable cause.2 Primary iris cysts involve the posterior pigment epithelium of the iris and ciliary body.3,4
Iridociliary cysts (located at the iridociliary junction) are the most common type of primary cyst and account for nearly three quarters of all cysts diagnosed.2,3 Solitary iris or ciliary body cysts are less common.5
Primary cysts are usually round, thin-walled and filled with sonolucent material, which appears clear via ultrasonography.2 Most remain stable and require no surgical treatment, but larger primary cysts may cause angle closure.6-9
Secondary iris cysts, which are usually larger and surrounded by thicker walls, are the result of penetrating trauma, surgery, implantation, metastasis or chronic use of miotics.3,10 Three types of secondary implantation cysts have been noted: pearl cysts, serous cysts and epithelial downgrowth. The rare pearl cyst is a stromal, white, solid iris tumor consisting of three layers of differing reflectivity. The more common translucent serous cysts can cause iris atrophy via compression, erosion through the iris and invasion of the posterior chamber. Epithelial downgrowth of surface cells from the cornea, conjunctiva and skin occurs through a site of ocular penetration.6,10
Iris nevi, which are typically benign, avascular, pigmented lesions that measure less than 3mm wide and less than 1mm thick.11-12 They can occur in up to 50% of the population and can be differentiated from iris melanomas by size, thickness, absence of vascularity and growth.12 But, nevi may exhibit slow growth, cause ectropion uvea and involve the anterior chamber angle, which make them difficult to differentiate from melanomas.11 Nevi may also recur after excision.13
According to one study, most hypercellular, pigmented lesions of the iris stroma can be classified as nevi.13 In this study, researchers produced a classification of lesions into nine categories and concluded that most lesions were nevi, although they displayed some similar characteristics to melanomas, such as documented growth or presence of ectropion uveae. Reassignment to a benign category was based on cytology, lack of clinical sequelae and absence of mortality.13
Malignant melanoma, the most common primary iris malignancy, tends to occur more often in younger patients than choroidal melanoma.14,15 Malignant melanomas typically present as lesions with rapid growth and may be accompanied by increased IOP, an anomalous vascular pattern and satellite lesions.11,14 Iris melanomas begin as benign lesions.14 Melanoma often occurs in the inferior half of the iris, perhaps because the lower iris is more exposed to sunlight.1 One retrospective study of 1,043 patients found a 3% rate of metastasis, and there was a variable risk based on the histological subtype of the melanoma.14 Guidelines may aid in lesion differentiation based on the presentation and features of suspicious lesions (see Is it Benign or Malignant?, below).1
Metastatic tumors, which also form in the iris, most commonly migrate from the breast and lung in women, and the lung and gastrointestinal tract in men.16-18 The breast was found to be the most common primary site of iris metastasis in a case series of 40 patients.19,20 But, in a case series of 512 patients, lung cancer was found to be the most common primary metastatic cancer to the iris.20 This study also found that 7.8% of uveal metastasis involved the iris.20 Prostate carcinomas have been reported to rarely metastasize to the uvea with a shorter survival time compared with metastasis to other sites.19 Metastatic prostate carcinoma to the uvea presents similarly to other metastatic uveal cancers.16-18
Iris metastasis may vary in clinical presentation.20 Symptoms of iris metastasis include blurred vision, pain, redness, visible iris lesion and photophobia.20 It is typically found when the lesion is small and vision is still good.Sometimes presenting as an amelanotic lesion with moderately anomalous vasculature, iris metastasis may also involve the anterior chamber angle, and may be accompanied by glaucoma, iridocyclitis and hypopyon.18,20 Iris metastases can be very friable, resulting in the shedding of tumor cells and the formation of a pseudohypopyon, which leads to a misdiagnosis of intraocular inflammation.21
Differentiating benign and malignant lesions can be difficult, so histologic studies, such as fine-needle aspiration cytology, are warranted for highly suspicious lesions. Both fluorescein angiography and B-scan ultrasonography have been used in the evaluation of iris lesions. But, there is limited utility in examining smaller iris lesions with B-scan ultrasonography. The drawbacks of iris fluorescein angiography (IFA) include its invasiveness and its limitations in differentiating vascular patterns of both benign and malignant lesions. Small lesions should be monitored with controlled photography.22
OCT and UBM have achieved similar and perhaps superior utility; they provide non-invasive techniques for producing high-resolution photos that are most helpful with localizing and measuring tumors in very precise increments.3,23
Is It Benign or Malignant? These clinical characteristics differentiate benign from malignant anterior segment tumors.1 Features Associated with Lesions Elevation Number Yes
More Likely Benign
More Likely Malignant
Flat or slightly elevated
Nodular
More than one
Solitary
Laterality
Bilateral
Unilateral
Size
< 3mm
> 3mm
Growth
No
Yes
Vascularization
No
Yes
Ectropion uvea
No
Yes
Iris infiltration
No
Yes
Pupillary distortion
No
Yes
Cataract
No
Yes
Sentinel vessels
No
Yes
Glaucoma
No
Optical Coherence Tomography
The principle of OCT is very similar to that of B-scan ultrasonography, with the exception that reflected light waves, rather than ultrasound waves, are utilized.23 This imparts some advantages over its predecessornamely, OCT provides significantly better resolution (light travels more quickly than sound), and light easily penetrates the ocular media, eliminating the need for fluid immersion or probe contact.24,25
OCT compares a partially coherent reference beam of 843nm to one reflected from human tissue; the two beams are then combined to produce an interference.26 Time of flight information is generated, which, in turn, provides spatial information about tissue microstructure.23 A tomographic image is produced by concurrently exhibiting up to 100 adjacent scans.26
OCT was first used to image the retina, and commercially available scanners, such as the Stratus (Carl Zeiss Meditec), utilize a 0.8m wavelenth.25 But, insufficient penetration and speed limit OCT scanners for anterior segment imaging applications.24,25 Increased scatter, both at the limbus and sclera, limits its ability to image the angle structuresthe shorter wavelength does not reliably penetrate all iris lesions.
The newer prototype cornea and anterior segment (CAS) OCT utilizes a longer, 1.3m wavelength that allows for full thickness penetration of a variety of iris lesions as well as angle structure visualizationa useful feature for determining whether an iris lesion has infiltrated the angle structures.25
The OCT signal produced by a particular tissue layer is a combination of its reflective, absorptive and scattering properties.25
One study described the OCT appearance of several different types of iris lesions.25 An iris cyst demonstrated low central reflectivity consistent with a fluid space. Iris nevi were visualized as areas of high anterior reflectivity with shadowing of the iris pigment epithelium. Hyperreflectivity at an area of ectropion uvea was observed. An iris melanoma appeared as a lesion with a highly corrugated surface and variable internal reflectivity; a thicker melanoma demonstrated shadowing of the otherwise highly reflective pigment epithelium.25
Standard criterion for use of the OCT in imaging the iris does not yet exist due to the newness of this technology and the paucity of examples found in the literature.
For our patient, Walter, a 0.8m wavelength OCT was used to image the iris lesion. Despite the known limitations, the increased thickness of the iris lesion was clearly demonstrable; furthermore, the ectropion uvea was clearly visualized. His OCT findings showed a caliper thickness of 631m and an area of high reflectivity at the anterior surface of the iris pigment epithelium.
B-Scan Ultrasonography
In years past, a B-scan was a conventional method of imaging the anterior segment. Most B-scans employ a 10MHz transducer that produces a resolution of 300m to 400m.27,28 Such resolution might permit imaging of larger iris and ciliary body lesions, but little useful information is gained in examining smaller lesions.28 And, ultrasonography may play little or no role in detecting iris metastasis, due partly to the size and location of the lesions.28
Likewise, a B-scan may not be capable of imaging iris cysts because of their small size. In one small study, eight iris cysts were imaged using both B-scan and UBM. Only two lesions were detected by conventional B-scan, but all eight lesions were detected by UBM.28
But, B-scan is still the method of choice for detecting posterior extension of larger lesions.28,29 In fact, for large lesions, B-scan can illustrate internal characteristics, such as acoustics or spontaneous pulsations, which is not possible with UBM.29
Ultrasound Biomicroscopy
UBM translates high-frequency sound waves into high-resolution images. Low-frequency systems produce better depth and penetration, while high frequency systems produce better resolutionUBM is only able to penetrate to about 4mm.30 Typically, a 50MHz to100MHz transducer is used to produce a resolution of up to 20m to 60m, 10 times higher than that of a conventional B-scan.28,30 With UBM, it is possible to accurately measure depth, tumor thickness and local invasion of lesions better than conventional ultrasound.28
UBM can provide very detailed imagery and localization of iris lesions, including excellent imagery of the anterior chamber angle, as with OCT. This is an excellent tool for accurately measuring lesions and monitoring for growth. There is good correlation in shape and location of lesions as compared with histopathological findings.28
Studies utilizing UBM to evaluate anterior segment tumors have found clinical features that might aid in characterization of lesions, including surface characteristics, internal reflectivity, anterior bowing of the iris plane and extension or involvement of the angle.3 It has also been useful in identifying posterior chamber and ciliary body lesions, such as melanomas and iridociliary cysts.28
In a study of 130 patients with anterior segment lesions, 70% were found to demonstrate neuroepithelial cysts.29 Such cysts were found to coexist with both nevi and ciliary body melanoma. And, the same study found that UBM was superior at identifying ciliary body and peripheral iris involvement of melanomas vs. B-scan.29
Visualization of lesions by UBM depends on lesion morphology. Typically, cysts are characterized by lack of internal reflectivity, or echolucency.27 Iris nevi may appear as lowly reflective surface plaques, or with fusiform thickening of more central lesions, or as anterior bowing of the iris root when located peripherally.28 Malignant melanomas may be seen as solid, irregularly shaped lesions with variable reflectivity within the iris stroma.27
UBM of Walter illustrated a small, focal, solid elevated mass at the anterior iris border. Malignant melanoma could not be specified using the UBM, but it could not be ruled out. Lesion size, lack of echolucency and lack of involvement of the anterior chamber were elicited; however, monitoring is still warranted.
IFA is performed on atypical iris lesions, though its usefulness in differentiating lesions is debatable.31 The rate of adverse effects with IFA is less than 5%.32
To perform IFA, the pupil must not be dilated. Color photos or green monochromatic photos should be taken before the procedure in order to locate the abnormality. There is no fluorescein pattern standard for IFA of the iris, because all irides are different, so it is more complicated to diagnose iris abnormalities specifically by vascular pattern alone.33 The iris of a normal adult under 40 years of age should show no dye leakage.33 But, by the fifth decade, the iris may show some age-related leakage.33 Normal iris vasculature does not leak dye because the iris vessels are nonfenestrated and have tight junctions between the cell tissues.33
The appearance of iris cysts vary on IFA based on type and size; iris vasculature may look completely normal, or demonstrate more dramatic angiographic changes.33 Lesion pigmentation that masks underlying fluorescence is typically considered benign if no dye leakage or uptake is observed at any phase of the IFA. Or, if continuous diffuse leakage, an irregular disorganized vascular network with alteration of the normal radial iris pattern, or a ring of hyperfluorescence develops around the lesion during angiography, it is more likely that the lesion is malignant. But, this evidence is not conclusive.34,35 Melanomas may show disorganized vasculature with gross leaking, hyperfluorescence and late diffuse staining.1,31
Walter has not undergone IFA at this point, and instead he will be monitored with serial photography.
Iris lesions are often difficult to differentiate from one another, in part because benign and malignant lesions often share clinical characteristics, including growth and appearance. Careful monitoring, including serial photography, is still considered a gold standard for managing iris lesions when clear characterization is not possible using standard diagnostic measures. The use of UBM and CAS OCT will enhance the clinicians ability to provide prudent care, and safeguard the patients well-being and overall health.
Dr. Williams is currently in private practice in
1.Marigo FA, Finger PT. Anterior segment tumors: current concepts and innovations. Surv Ophthalmol 2003 Nov-Dec;48(6):569-93.
2. Shields JA. Primary cysts of the iris. Trans Am Ophthalmol Soc 1981;79:771-6.
3. Marigo FA, Esaki K, Finger PT, et al. Differential diagnosis of anterior segment cysts by ultrasound biomicroscopy. Ophthalmology 1999 Nov;106(11):2131-5.
4. Gogos K, Tyradellis C, Spaulding AG, Kranias G. Iris cyst simulating melanoma. J AAPOS 2004 Oct;8(5):502-3.
5. Augsburger JJ, Affel LL, Benarosh DA. Ultrasound biomicroscopy of cystic lesions of the iris and ciliary body. Trans Am Ophthalmol Soc 1996;94:259-71.
6. Shields JA, Kline MW, Augburger JJ. Primary iris cysts: a review of the literature and report of 62 cases. Br J Ophthalmol 1984;68(3):152-66.
7. Vela A, Rieser JC,
8. Tanihara H, Akita J, Honjo M, Honda Y. Angle closure caused by multiple, bilateral iridociliary cysts. Acta Ophthalmol Scand 1997 Apr;75(2):216-7.
9. Haller JA, Stark WJ, Azab A, et al. Surgical approaches to the management of epithelial cysts. Trans Am Ophthalmol Soc 2002;100:79-84.
10. Finger PT, McCormick SA, Lombardo J, et al. Epithelial inclusion cyst of the iris. Arch Ophthalmol 1995 Jun;113(6):777-80.
11. Van Klink F, De Keizer RJW, Jager MJ, Kakebeeke-Kemme HM. Iris nevi and melanomas: a clinical follow-up study. Doc Ophthalmol 1992;82(1-2):49-55.
12. Kaiser P, Friedman N, Pineda R. The
13. Jakobiec FA, Silbert G. Are most iris melanomas really nevi? A clinicopathologic study of 189 lesions. Arch Ophthalmol 1981 Dec;99(12):2117-32.
14. Geisse LJ, Robertson DM. Iris melanomas. Am J Ophthalmol 1985 Jun;99(6):638-48.
15. Katz NR, Finger PT, McCormick SA, et al. Ultrasound biomicroscopy in the management of malignant melanoma of the iris. Arch Ophthalmol 1995 Nov;113(11):1462-3.
16. Shields JA, Shields CL. Metastatic tumors to the intraocular structures. In: Shields JA, Shields CL, eds. Intraocular Tumors: A Text and Atlas.
17. Shields JA, Shakin EP, Shields CL. Metastatic malignant tumors. In: Gold DH, Weingeist TA, eds. The Eye in Systemic Diseases.
18. Font RL, Ferry AP. Carcinoma metastatic to the eye and orbit III. A clinicopathologic study of 28 cases metastatic to the orbit. Cancer 1976 Sep;38(3):1326-35.
19. De Potter P, Shields CL, Shields JA, Tardio DJ. Uveal metastasis from prostate carcinoma. Cancer 1993 May;71(9):2791-6.
20. Shields JA, Shields CL, Kiratli H, DePotter P. Metastatic tumors to the iris in 40 patients. Am J Ophthalmol 1995 Apr;119(4):422-30.
21. Shields JA. Metastatic tumors to the uvea. Int Ophthalmol Clin 1993 Summer;33(3):155-61.
22. Baldridge M. Malignant melanoma of the iris.
23. Huang D, Swanson EA, Lin CP, et al. Optical coherence tomography. Science 1991 Nov;254:1178-81.
24. Huang D, Li Y, Radhakrishnan S. Optical coherence tomography of the anterior segment of the eye. Ophthalmol Clin N Am 2004 Mar;17(1):1-6.
25. Bakri SJ, Singh AD, Lowder CY, et al. Imaging of iris lesions with high-speed optical coherence tomography. Ophthalmic Surg Lasers Imaging 2007 Jan-Feb;38(1):27-34.
26. Hrynchak P, Simpson T. Optical coherence tomography: an introduction to the technique and its use. Optom Vis Sci 2000 Jul;77(7):347-56.
27. Reminick LR, Finger PT, Ritch R, et al. Ultrasound biomicroscopy in the diagnosis and management of anterior segment tumors. J Am Optom Assoc 1998 Sep;69(9):575-82.
28. Pavlin CJ, McWhae JA, McGowan HD, Foster FS. Ultrasound biomicroscopy of anterior segment tumors. Ophthalmology 1992 Aug;99(8):1220-28.
29. Conway RM, Chew T, Golchet P, et al. Ultrasound biomicromiscopy: role in diagnosis and management in 130 consecutive patients evaluated for anterior segment tumors. Br J Ophthalmol 2005 Aug;89(8):950-5.
30. Pavlin CJ, Harasiewicz K, Eng P, et al. Clinical use of ultrasound biomicroscopy. Ophthalmology 1991 Mar;98(3):287-95.
31. Fries PD, Char DH. Fluorescein angiography in ciliary body melanomas. Ophthalmologica 1990;201(2):57-65.
32. Butner RW, McPherson AR. Adverse reactions in intravenous fluorescein angiography. Ann Ophthalmol 1983;15(11):1084-6.
33. Brancato R, Bandello F, Lattanzio R. Iris fluorescein angiography in clinical practice. Surv Ophthalmol 1997 Jul-Aug;42(1):41-70.
34. Dart JK, Marsh RJ, Garner A, Cooling J. Fluorescein angiography of anterior uveal melanocytic tumors. Br J Ophthalmol 1988 May;72(5):326-37.
35. Jakobiec FA, Depot MJ, Henkind P, Spencer WH: Fluorescein angiography pattern of iris melanocytic tumors. Arch Ophthalmol 1982 Aug;100(8):1288-99.

