
Patients who present with dry eye, episcleritis and scleritis, or uveitis are often troubled by systemic immunologic disorders. For example, some 1% to 3% of the total American population suffers from rheumatoid arthritis, and about 0.5% has inflammatory bowel disease.1,2
We can successfully treat many of these secondary ocular disorders in our practice and offer information to other health-care providers to guide the management of systemic conditions. This article will discuss the basic pathophysiology of several common immunologic disorders, and will review both the diagnosis and treatment of their ocular sequelae.
Rheumatoid Arthritis
Rheumatoid arthritis (RA) is an autoimmune inflammation that results from the rheumatoid factor proteins attack on immunoglobulin G (IgG).3 Inflammatory cytokines, especially tumor necrosis factor (TNF)-alpha, are released from various white blood cells, leading to swelling and cell death. Joints and internal organs are particularly vulnerable to the damaging infiltration of inflammatory cells and pannus.3
Ocular involvement in rheumatoid arthritis results from cytokine-mediated inflammation. TNF may cause the release of metalloproteinases, which break down basement membranes and destroy ocular tissue.1 This process affects primary and accessory lacrimal glands; hence, dry eye is the most common manifestation of RA.1 Episcleritis is also seen commonly at the primary-care level. Scleritis is a more serious involvement (figure 1).
Also, rheumatoid arthritis patients are prone to non-granulomatous, bilateral uveitis, which is often chronic in nature. 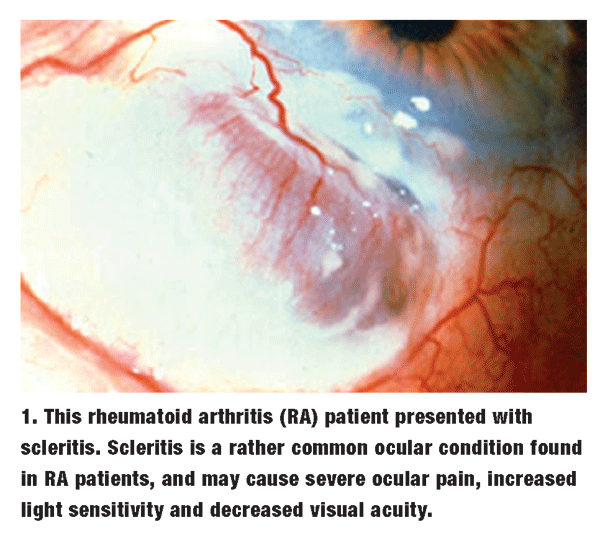 The severity of the disease dictates the appropriate level of systemic and/or ocular anti-inflammatory therapy. Many RA patients are treated with non-steroidal anti-inflammatory drugs (NSAIDs), while patients with more severe cases are medicated with disease-modifying anti-rheumatologic agents (DMARDS), such as Plaquenil (hydroxychloroquine, Sanofi-Aventis). Plaquenil initially was developed as an anti-malarial agent, and its effectiveness against rheumatoid arthritis and lupus is poorly understood.
The severity of the disease dictates the appropriate level of systemic and/or ocular anti-inflammatory therapy. Many RA patients are treated with non-steroidal anti-inflammatory drugs (NSAIDs), while patients with more severe cases are medicated with disease-modifying anti-rheumatologic agents (DMARDS), such as Plaquenil (hydroxychloroquine, Sanofi-Aventis). Plaquenil initially was developed as an anti-malarial agent, and its effectiveness against rheumatoid arthritis and lupus is poorly understood.
One word of caution: Toxicity resulting from macular and corneal deposition of Plaquenil can be problematic, although this does not often occur with the standard 200mg to 400mg daily dosage.4 Patients who take Plaquenil should have a dilated examination, Isihara color plates (performed monocularly), and central visual field testing annually.
Amsler grid is an effective chairside test for central visual fields. The Humphrey macular program is an excellent automated alternative. Suppression of macular sensitivity shown on these tests, as well as macular pigment changes on funduscopy, are reasons to alert the rheumatologist and attain retinal consultation.
Physicians have also considered systemic steroid dosing a fundamental treatment method for RA patients; yet, monoclonal antibody drugs target cytokine activity and do not have the broader effects of steroids. Enbrel (etanercept, Amgen/Wyeth), Remicade (infliximab, Centocor) and Humira (adalimumab, Abbott) are commonly used anti-cytokine drugs.
The anti-cytokine drugs are produced from monoclonal antibody technology. The most severe of cases require immunosuppressive therapies, such as methotrexate, cyclophosphamide or cyclosporine.1
For the ocular sequelae, episcleritis is responsive to q.i.d. dosing of the soft steroids: Vexol (rimexolone, Alcon), Lotemax (loteprednol etabonate, Bausch and Lomb) or FML Forte (fluorometholone, Allergan).
Intraocular pressure effects are still possible with the soft steroids, so regular monitoring is essential. Scleritis generally requires systemic immunosuppression therapy.1
Immunosuppression therapy should be highly considered for any patient with granulomatous, bilateral uveitis.5
Acne Rosacea
Acne rosacea (AR) produces papules, pustules, and teleangectasias, especially on exposed skin. As many as 20% of rosacea patients may experience ocular involvement, including chronic blepharoconjunctivitis, meibomian gland dysfunction, and even corneal thinning and perforation.
The exact cause is unknown, but delayed hypersensitivity reaction to Staphylococcus and Streptococcus exotoxin is one proposed mechanism of AR; however, research is both inconclusive and ongoing.6 Rosacea patients have elevated levels of cytokine, lipase and metalloproteinase enzymes, resulting in tissue damage.7 The condition has been predominantly associated with patients of Northern European origin, but substantive research evidence of this relationship is lacking.
Artificial tears and warm compresses can be very effective at entry level. Acute flare-ups of ocular rosacea may require antibiotic drops and/or ointments.
Topical steroids should be avoided if possible, as rosaceas inherent chronicity may create dependency on these short-term drugs. In our practice, we have used topical Metrogel (metronidazole, Galderma) on the eyelids, which has yielded positive results.8
This is an offlabel usage, as no ophthalmic preparation is available. Therefore, the patient must carefully avoid introducing the gel into the eye itself.
 Doxycycline (50mg to 100mg q.d.) is a common oral treatment for AR. While doxycycline and other tetracycline analogues possess antibiotic effects, the main purpose of these drugs is to reduce inflammatory cytokines, metalloproteinases and lipases.9 These drugs may also help to curtail the viscosity of sebaceous secrtion.9,10,11 There may be additional anti-inflammatory effects associated with doxycycline that are poorly understood.
Doxycycline (50mg to 100mg q.d.) is a common oral treatment for AR. While doxycycline and other tetracycline analogues possess antibiotic effects, the main purpose of these drugs is to reduce inflammatory cytokines, metalloproteinases and lipases.9 These drugs may also help to curtail the viscosity of sebaceous secrtion.9,10,11 There may be additional anti-inflammatory effects associated with doxycycline that are poorly understood.
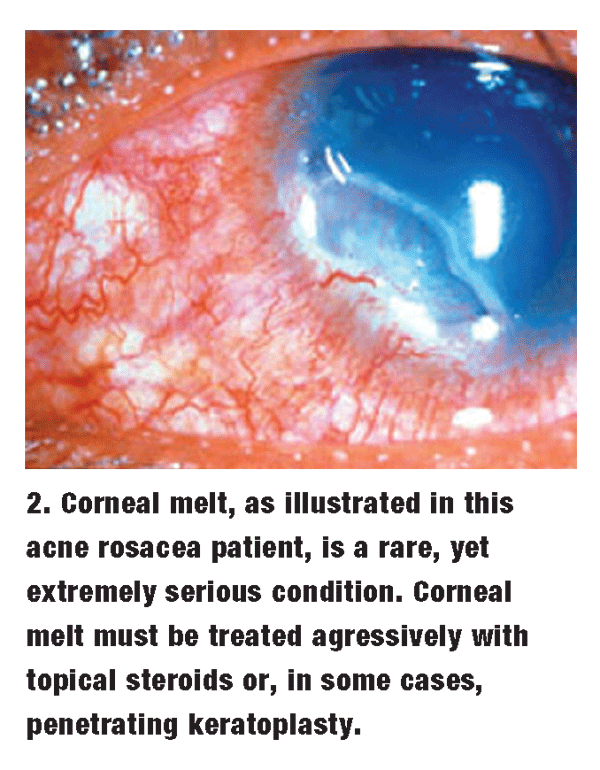
Rarely, AR patients have such severe corneal problems that penetrating keratoplasty (PK) is necessary (figure 2). If corneal epithelial cells adhere poorly, recurrent erosion, corneal thinning and scarring can result. If significant corneal thinning occurs, wide-diameter PK may be required.
Giant Cell Arteritis
Giant cell arteritis (GCA), while not commonly seen in a primary-care optometric practice, is a true ocular emergency when it does present. GCA results in the rapid development of severe vascular obstruction, which places the patients life at stake.
Giant cell arteritis causes an infiltration of inflammatory cells into the walls of the cranial arteries. This causes thinning of the lumen, resulting in an ischemic pathology. The eye is particularly vulnerable, presumably due to the narrow caliber of the ophthalmic artery.
Optometrists may be most familiar with GCAs effects on the optic nerve and its catastrophic effects on vision. If cranial nerves three, four, or six are involved in the ischemic process, diplopia, mydriasis or ptosis may result.12
GCA patients typically present with a dramatic reduction in unilateral visual acuity. They tend to suffer from severe headache and scalp pain along the course of the temporal artery. Some patients may even refrain from touching or brushing their hair. Chewing can also be affected, as the jaw muscles may suffer claudication, or cramping, from ischemia. Polymyalgia rheumatica and other systemic inflammatory diseases, such as polyarteritis nodosa, usually accompany GCA.3
The role of the primary-care optometrist is critical when managing a patient with GCA. When a patient presents with the telltale signs and symptoms, it is urgent to ensure that the vasculitic disease does not catalyze a stroke, which could result in death.
One study showed that 3% to 4% of patients with GCA have cerebral ischemic events.13 While the affected eye may have a poor visual prognosis, prompt action may protect the vision in the other eye or even save the patients life.
Upon suspicion of GCA, you should notify the patients primary-care provider or an emergency room physician. If indications are strong, systemic steroids should be started, even before attaining laboratory results. One treatment algorithm employs 12 doses of intravenous methylprednisone, given in 250mg doses every six hours, followed by 80mg to 100mg prednisone q.d. p.o.12
Temporal artery biopsy is a key tool in diagnosing a GCA patient, although it may need to be repeated if results are negative despite strong clinical evidence.12 Laboratory testing should include complete blood count (CBC), erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). The following algorithm, devised by the neuro-ophthalmologist Sohan Singh Hayreh, can help identify GCA cases: Patients with an ESR greater than 47mm/hr and a CRP greater than 2.45mg/dl are 97% specific for GCA.14
HLA-B27 Disorders
The HLA-B27 disorders are a group of inflammatory conditions that commonly exhibit the presence of a specific surface antigen on human leukocytes. The HLA-B27 disorders include ankylosing spondylitis, psoriatic arthritis, reactive arthritis and Behets disease.1
Patients with these disorders often present with a variety of systemic inflammatory ailments. For example, ankylosing spondylitis produces spinal arthritis with ligament calcification that is often most painful upon awakening, but is ameliorated with exercise. Psoriatic arthritis is a rheumatoid arthritis that is accompanied by psoriatic skin rashes.
You can serve as a liaison to the patients general practitioner by providing a thorough history, treating the accompanying ocular pathology and securing a timely referral to a rheumatologist. The HLA-B27 disorders can be debilitating without systemic immunosuppressive drugs and/or physical therapy.
The primary ocular involvement associated with the HLA-B27 disorders is an acute, non-granulomatous anterior uveitis that is typically unilateral. HLA-B27 disorders account for approximately 18% to 32% of uveitis seen in western countries.15 Fortunately, most cases respond well to topical therapy, namely hourly applications of Pred Forte (prednisolone acetate, Allergan) and cycloplegic drops. An initial loading dose of Pred Forte every 15 minutes for the first two hours can help promote rapid symptomatic relief.
When clinical findings, specifically cells and flare, have improved, it is critical to taper the steroid drops in a deliberate fashion. In our practice, we typically continue the application of drops q.h. for the first week, then q2h for the second week, q4h for the third week and b.i.d. for the fourth week. Although this large cumulative dosage of prednisone increases the risk for cataracts and glaucoma, improper tapering may present an even greater risk.
A patient who has a recurrence of inflammation while tapering the dosage must restart the entire process, typically resulting in an even higher total amount of steroid being used.
Cycloplegia is also critical to successful treatment. Because dosing is recurrent, it is important to select a parasympatholytic agent that has minimal adverse effects, such as homatropine or scopolamine. We typically direct the patient use one of these agents t.i.d. until signs of inflammation are resolved. In this case, the dosing can be stopped without tapering.
Inflammatory Bowel Disorders
Inflammatory bowel disorders (IBDs), including Crohns disease and ulcerative colitis, result in recurrent bouts of constipation, diarrhea and indigestion due to inflammation of the intestinal wall. Many have an accompanying RA, as both conditions cause excessive release of TNF.1
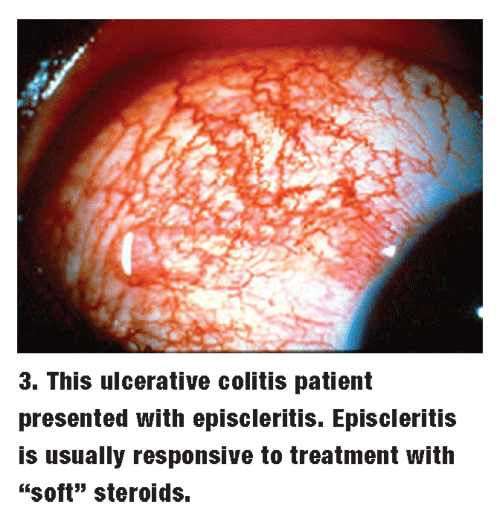 About 5% of patients with IBD develop ocular disease.5 Episcleritis and uveitis are the most common ocular pathologies in IBD patients. As with rheumatoid arthritis, episcleritis is usually responsive to q.i.d. dosing of the soft steroids, such as Vexol, FML Forte or Lotemax (figure 3). Extended dosage tapering is generally not necessary.
About 5% of patients with IBD develop ocular disease.5 Episcleritis and uveitis are the most common ocular pathologies in IBD patients. As with rheumatoid arthritis, episcleritis is usually responsive to q.i.d. dosing of the soft steroids, such as Vexol, FML Forte or Lotemax (figure 3). Extended dosage tapering is generally not necessary.
Uveitis is a much greater concern for an IBD patient, as it can be bilateral with posterior involvement.5 While topical treatment is indicated, posterior uveitic involvement requires systemic immunosuppression, and possibly periocular steroid injection. Because of the severity of inflammation and the use of toxic therapeutic agents, IBD patients also run a greater risk for cataracts, glaucoma and cystoid macular edema.
Sarcoidosis
Sarcoidosis is a multi-organ inflammatory process of unknown etiology. Patients with sarcoidosis have a characteristic sarcophagus, or coffin-shaped, inflammatory cell infiltration in the affected organs. Skin lesions, lung pathology and ocular inflammation are often associated with this condition.
Patients typically present with flu-like symptoms, including fever, malaise, respiratory problems and lymphadenopathy.3,16 They may also have sharply elevated blood pressure resulting from an excessive amount of angiotensin converting enzyme (ACE).
Systemic work-up includes gallium-enhanced chest X-ray, angiotensin converting enzyme level and serum calcium.3
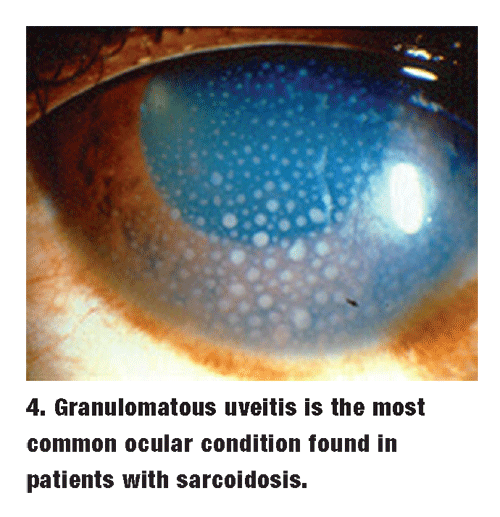 The classic ocular finding in patients with sarcoidosis is a granulomatous uveitis, but the lid, conjuntiva and lacrimal glands are also vulnerable to inflammation (figure 4). Lid granulomas, dry eye and conjunctivitis are possible results. This inflammation may be either anterior in nature or may additionally affect posterior structures. Aggressive topical treatment with Pred Forte q.h. may not be completely effective.
The classic ocular finding in patients with sarcoidosis is a granulomatous uveitis, but the lid, conjuntiva and lacrimal glands are also vulnerable to inflammation (figure 4). Lid granulomas, dry eye and conjunctivitis are possible results. This inflammation may be either anterior in nature or may additionally affect posterior structures. Aggressive topical treatment with Pred Forte q.h. may not be completely effective.
Close monitoring of these patients may suggest the necessity of adding periocular or systemic steroid therapy. These options should be considered if there is unresolved pain, cystoid macular edema, or unabated cells and flare. It is important to note that inflammation posterior to the iris is generally not treatable with topical agents alone.
Superior Limbic Keratoconjunctivitis
While not strictly considered a systemic disorder, superior limbic keratoconjunctivitis (SLK) has a correlation with hyperthyroidism that requires a physical examination and blood work. Superior limbic keratoconjunctivitis patients present with a history of stinging, burning and other ocular discomfort, although the eye remains relatively quiet.
Both mechanical friction by the upper lid and tear film deficiencies are the likely causes of SLK.17
Examination of the superior bulbar conjunctiva reveals injection and hypertrophy, with the appearance of superior corneal staining, filaments and haze. The superior tarsal conjunctiva may exhibit fine papillae and injection.
This chronic condition may require a multi-faceted therapeutic approach to keep the patient comfortable. Possible therapies include:
Topical steroids, which may be helpful for acute flare-ups; but, reliance on this modality has obvious pitfalls, including: steroid-induced glaucoma, cataract formation and potentiation of infection if steroids are used chronically for SLK.
Artificial tears.
Large-diameter bandage contact lenses, although our practice has not had great success with this modality.
Punctal plugging, which may provide better comfort if the ocular surface is kept moist.
Restasis (cyclosporine, Allergan) may increase tear production and have an anti-inflammatory effect, although long-term studies are lacking. Note that this is an off-label usage for Restasis that should be reviewed with the patient.
Beyond the primary-care level, silver nitrate and superior conjunctival resection may be necessary, although these options have produced varying results.12,18
Other Treatment Considerations
Dry eye syndrome is a common burden for rheumatologic patients. The primary and accessory lacrimal glands can be prime targets for systemic inflammation, resulting in poor tear volume and quality.If artificial tears prove inadequate, these patients may benefit from short-term therapy with soft steroids, such as Vexol.
Steroid response should be monitored, as even milder steroids can induce significant intraocular pressure increases in susceptible individuals. Over the long term, Restasis, which suppresses T cells, can benefit patients whose dry eye is inflammatory in origin.
Restasis provides an intermediate step prior to the instillation of punctal plugs. Although plugs can be an effective treatment for dry eye, their association with canalicular granulomas and tendency to dislodge makes them an imperfect option.
Also, punctal plugs may actually contribute to inflammation by limiting the exit of cytokines and other inflammatory mediators from the pre-corneal tear film.
Laboratory Testing
A good working relationship with the family practitioner, internist or rheumatologist that may be involved in the care of patients with autoimmune disorders is essential.
We play an important role in the diagnosis and treatment by informing the other doctor about the ocular findings, especially in cases of uveitis. For example, a uveitis that is identified as granulomatous raises greater suspicion of sarcoidosis, tuberculosis or syphilis, and will guide the systemic work-up accordingly. Attentive care to the rheumatologic patient can be quite rewarding for the primary-care optometrist. The modern practice can offer several diagnostic and treatment options that were unavailable during the last decade. Patient retention and professional satisfaction are the results of treating these challenging conditions.
Dr. Potter is chief of optometry and contact lenses with Millennium Eye Care, a multi-subspecialty optometry/ophthalmology practice in Freehold, N.J. He has a special interest in the ocular effects of systemic diseases.
1. Pleyer U, Foster CS. Uveitis and Immunological Disorders.
2. Loftus EV. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology 2004 May;126(6):1504-17.
3. Goljan E. Most Commons in Pathology and Laboratory Medicine.
4. Marmor MF, Carr RE, Easterbrook M, et al. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy. Ophthalmology 2002 Jul;109(7):1377-82.
5. Nussenblat R, Whitcup S,
6. Macsai M, Mannis M, Huntley A. Chapter 41: Acne Rosacea, Dermatology CD Library, Lippincott-Raven Publishers, 1996.
7. Baldwin HE. Systemic Therapy for Rosacea. Skin Therapy Lett 2007 Mar;12(2):1-5,9.
8. Bleicher PA, Charles JH, Sober AJ. Topical metronidazole therapy for rosacea. Arch Dermatol 1987 May;123(5):609-14.
9. Ta CN, Shine WE, McCulley JP, et al. Effects if minocycline on the ocular flora of patients with acne rosacea or seborrheic blepharitis. Cornea 2003 Aug;22(6):545-8.
10. Seedor JA, Perry HD, McNamara TF, et al. Systemic tetracycline treatment of alkali-induced corneal ulceration in rabbits. Arch Ophthalmol 1987 Feb;105(2):268-71.
11. Nicolaides N, Ruth EC. Unusual fatty acids in the lipids of stear and human meibomian gland excreta. Curr Eye Res 1982-1983;2(2):93-8.
12. Kunimoto D, Kanitkar K, and Makar M. The Wills Eye Manual.
13. Caselli RJ, Hunder GG, Whisnant JP. Neurologic disease in biopsy-proven giant cell (temporal) arteritis. Neurology 1988 Mar;38(3):352-9.
14. Hayreh SS, Podhajsky PA, Raman R, Zimmerman B. Giant cell arteritis: validity and reliability of various diagnostic criteria. Am J Ophthalmol 1997 Mar;123(3):285-96.
15. Chang J, McCluskey P, and
16. Sowka JW, Gurwood AS, Kabat AG. Handbook of Ocular Disease Management. Sarcoidosis Available at: www.revoptom.com/HANDBOOK/sect7d.htm (Accessed: August 30th 2007).
17. Sowka JW, Gurwood AS, Kabat AG. Handbook of Ocular Disease Management. Sarcoidosis Available at: www.revoptom.com/HANDBOOK/SECT12a.HTM (Accessed: August 30th 2007).
18. Papaliodis GN. Superior Limbal Keratoconjunctivitis: Case Reports.

