| |
Volume 11, Number 3 |
August
2015 |
|
|
Inside
This Issue |
|
|
|
|
|
This e-newsletter is provided free to doctors through industry support from |
 |
| |
FROM
THE DESK OF THE EDITOR
Recently, I was asked to serve as a Clinical Advisor to “ODs on Facebook” by Dr. Alan Glazer, the founder and administrator of the website. After several emails with Alan, I decided to do so, as I felt it was an interesting project. And it is. The amount of interesting cases, photos and questions posted on a daily basis on this website by its over 20,000 members (as of about two weeks ago) is astonishing! I often try to inject my opinion on some of the cases, but often find it is unnecessary, as many ODs have a great knowledge base to share with each other. Also, I have learned a lot myself, not only in the retina arena, but many other topics that I am less familiar with, such as practice management, coding, billing, etc., etc. If you are not a member of the site, I suggest you check it out. I am sure you will learn something, and hopefully be able to contribute as well.
LIVE
POLL
|
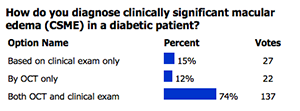
How much do you think a patient would be willing to pay for genetic testing to help assess their risk for advanced AMD?
|
|
On the topic of learning, please consider attending our ORS meeting December 4th and 5th in Anaheim, CA. Also, if you are attending the Academy in New Orleans this October, I will be moderating a session on Saturday for the Retina Special Interest Group (SIG), entitled “Diabetes: A Comprehensive Team Approach” from 10 am to noon. We will have an OD, retinal specialist, and endocrinologist share their thoughts on diabetes and how we can work to together to care for our patients with diabetes. Love to see you there!
Lastly, please look at the preliminary results of the VAST (Vitreoretinal Adhesion Prevalence Study) study by Julie Rodman. Julie, along with Diana Shechtman, are the primary investigators for this study evaluating the prevalence of VMA (vitreomacular adhesion) in patients over age 40. I am also involved in the study, as are many of my ORS colleagues, but really it is due to Julie and Diana’s hard work and dedication that this important study exists. The study will hopefully help elucidate the prevalence of VMA, as well as its clinical characteristics. This in turn should help us take better care of our patients with VMA, by having a better understanding of the condition. The final results are expected next summer.
Steven Ferrucci, O.D., F.A.A.O.
Editor in Chief
ORS/REVIEW OF OPTOMETRY MEETING
We are excited about bring the ORS/Review of Optometry Retina Update back to the west coast this year! A distinguished slate of speakers from both optometry and ophthalmology will present clinically relevant information on vitreoretinal disease prevention, diagnosis, technology, and treatment/management. Don't miss optometry's premier posterior segment educational event as we focus on providing education and insight into best retina practices in 2015.
Click here for more information and to register.
Joe Pizzimenti O.D., F.A.A.O.
Conference Co-chair
ORS Fellow
Past-President
|
PRESIDENT'S MESSAGE
Recently, there has been talk of a potential treatment for retinal and choroidal neovascularization that is delivered via topical eye drops. This has, of course, caught the attention of many in the optometric community. I have even been contacted by an editor of one of our most popular trade journals who asked me if I thought this might revolutionize how optometrists participate in the care of patients suffering from conditions such as wet AMD, diabetic retinopathy, and retinal vein occlusions. Well, before we get too excited, just what is this exciting eye drop and what do we know about it? Squalamine is a topical anti-VEGF agent that is derived from the internal organs, mostly the liver, of the dogfish shark. It inhibits VEGF, PDGF, and BFGF. Ohr pharmaceuticals has completed a phase II study, known as the IMPACT study, looking at the use of Squalamine in the management of wet AMD. Essentially, there were two treatment arms. One group received Squalamine eye drops BID in addition to monthly Lucentis injections, while the second group received placebo drops BID with monthly Lucentis injections. In the end, twice as many patients in the Squalamine group gained three or more lines of vision than in the placebo group. Recruiting is currently under way for a phase III follow up study using visual acuity improvement as an end point. Studies are also underway to evaluate the use of Squalamine in the treatment of central retinal vein occlusion.
MAY 2015
POLL RESULTS
 |
So what do the results that have been obtained thus far tell us? Well, it seems that wet AMD patients do better when Squalamine is added to their monthly Lucentis injection, so at the least, Squalamine appears to augment the effect of traditional anti-VEGF therapy. Might we be able to use topical Squalamine alone some day in the management of neovascularization? Might the concomitant use of Squalamine enable us to lengthen the time interval between anti-VEGF injections? If it makes it to the market, how expensive will it be? Clearly, we don’t yet have the answer to any of these questions now, but time will tell. Until then, dogfish sharks everywhere can rest easy for a while.
Brad Sutton, O.D., F.A.A.O.
ORS President

YOU
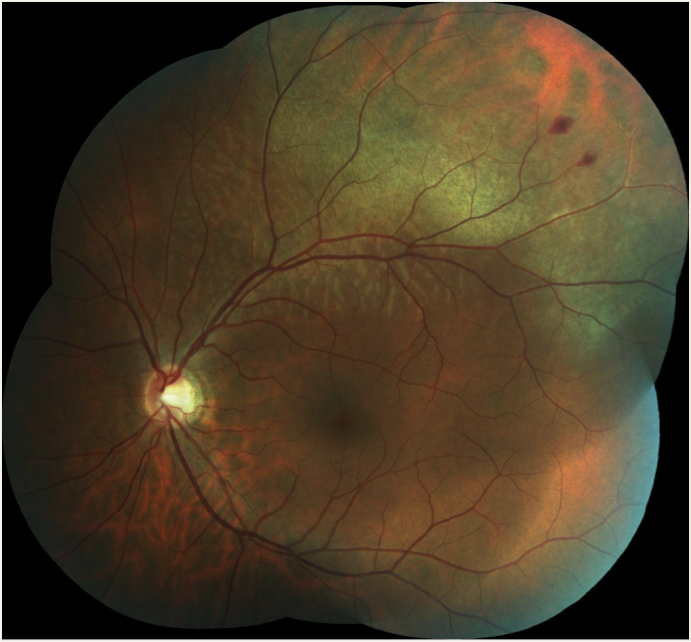
MAKE THE DIAGNOSIS
Answer appears later in newsletter.

CLINICAL
PEARLS
Selecting the correct patient for Jetrea
By Brad Sutton, OD, FAAO
ORS Fellow and President
Thus far, the adoption of Jetrea (Ocriplasmin) as a routine treatment for symptomatic vitreoretinal adhesion has been somewhat slow in the United States. This may be due, at least in part, to the fact that it was effective roughly one quarter of the time in the original clinical trials, each injection costs approximately $3000.00 to $4000.00 and is covered by some, but not all, insurances, and there have been reports of post injection visual disturbance and visual loss. Currently, post market approval effectivity is being further studied in the ORBIT study. In addition, looking at the results obtained in high volume Jetrea practices over time, certain factors have been identified that improve the success rate to approximately 50%. In other words, by choosing to use Jetrea in patients with certain characteristics, the chance of success is significantly enhanced. These characteristics include……..
1) VMA less than 1500 microns in diameter (focal adhesion rather than broad adhesion)
2) Age less than 65
3) The presence of a full thickness macular hole associated with VMA, if the hole is less than 250 microns in diameter
4) Phakic eyes
5) No ERM
VAST: The Vitreoretinal Adhesion (Prevalence) Studys
By Julie Rodman, O.D., F.A.A.O.
ORS Fellow
Recently, the preliminary results of the VAST study were presented at ARVO, The Association of Vision and Research in Ophthalmology.. This investigation analyzed the vitreomacular interface in patients 40 years of age and older with regard to vitreomacular adhesion through use of the SD-OCT. Due to the high association of maculopathy and visual disturbance as a consequence of vitreomacular adhesion, early detection of this condition could maximize the outcome and visual prognosis. Secondary evaluation included the correlation between VMA and gender, ethnicity, and refractive error.
Data was collected from 2179 eyes of 1120 subjects of which 43% were males. The mean age of the subjects was 59 years (range 40-90; SD 9.4). 8% of subjects were emmetropes, 35% were myopes, and 57% were hyperopes. 11% of eyes were pseudophakic; these were excluded from refractive error data. For subjects reporting ethnicity, 49% were Caucasian, 32% were Afro-American, 14% were Hispanic, 3% were Asian or Indian, and 2% were unknown or of mixed descent. Data were collected from a total of 9 centers in the US. A comprehensive eye examination, including medical history, amsler grid testing, dilated fundus examination, and SD-OCT was performed on each subject. The status of the posterior vitreoretinal interface was evaluated by a team of independent, masked readers, who determined the presence or absence of VMA from SD-OCT images. Classification was done according to the rubric established by "The International Vitreomacular Traction Study Group". Multiple logistic regression analyses with multilevel modeling to account for the correlation between eyes of a subject were used to explore the relationship between presence of VMA and other factors. Data was obtained over a one year period.
The presence of VMA was detected in 33.5% of eyes. VMA>1500 um was more prevalent (69%) than VMA<1500 um (31% of eyes with VMA). When comparing across 10-year age categories, VMA was most prevalent in 50-59 year olds (41.5%). With advancing age, the proportion of patients with VMA decreases (e.g., it is present in only 1.4% of patients 80-95 years of age). For every 10 year increase in age, there was a statistically significant reduced odds of having VMA by 55% (95% CI: 38-68%, p<0.001). In addition to age, ethnicity was a statistically significant factor in the prevalence of VMA. Afro-Americans were 74% less likely to have VMA than Caucasians (95% CI: 49-87%, p<0.001). Hispanics were 61% less likely to have VMA than Caucasians (95% CI: 6-84%, p=0.04). Gender (p=0.73) and refractive error (p=0.83 for myopia and p=0.92 for hyperopia) were not statistically significantly associated with VMA. VMA was present in approximately one -third of eyes representing a wide range of ages, demographics and refractive error. Patients who were 50-59 years of age or of Caucasian descent were more likely to have VMA. The anticipated completion date for the VAST study is summer 2016.
|

JOURNAL
ABSTRACTS
Increased Fundus Autofluorescence and Progression of Geographic Atrophy Secondary to Age-Related Macular Degeneration: The GAIN Study.
The purpose of this prospective natural history cohort study is to define the role of increased fundus autofluorescence (FAF), a surrogate for lipofuscin content, as a risk factor for progression of geographic atrophy (GA).
The study contained 109 eyes of 82 patients with GA secondary to age-related macular degeneration to which a minimum follow-up of 6 months were included. Lipofuscin content was classified independently by 2 masked observers according to FAF patterns described previously. The main outcome was the progression of GA in mm2/year as measured with FAF.
Median follow-up was 18 months (range, 6-42). Median GA growth was 1.61 mm2/year. FAF, baseline area of atrophy, and time of follow-up were independently associated with GA progression (P < .004). FAF patterns and baseline area of atrophy were strongly associated (P < .0001), suggesting potential confounding. Mediation analysis suggested that most of the effect of FAF patterns on GA growth was actually caused by baseline area of atrophy.
The study concluded that FAF patterns, baseline area of atrophy, and time of follow-up were associated with GA progression. However, FAF patterns seem to be a consequence (not a cause) of enlarging atrophy and their effect on GA progression seems mostly driven by baseline area of atrophy.
Biarnés M, Arias L, Alonso J, et al. J. Am J Ophthalmol. 2015 Aug;160(2):345-353.
Progressing geographic atrophy: choroidal thickness and retinal sensitivity identify two clinical phenotypes
The purpose was to analyze changes in choroidal thickness and retinal sensitivity in patients with GA with or without choroidal neovascularization (CNV) in the fellow eye.
Subjects were placed in two groups: those with bilateral GA or those with unilateral GA with CNV in the fellow eye. Enhanced depth imaging optical coherence tomography (EDI-OCT), blue and near infrared-wavelength fundus autofluorescence, and microperimetry were evaluated every six months.
GA area, choroidal thickness, and retinal sensitivity were measured in the eyes with GA at baseline and every 6 months up to the last follow-up visit. 16 eyes in the bilateral GA and 11 eyes in the unilateral GA were studied. Mean GA area was not significantly different between groups at baseline or at follow-up.
Choroidal thickness was significantly greater in the bilateral GA group compared with the unilateral GA group both at baseline and at last follow-up. During follow-up, mean choroidal thickness significantly decreased only in the unilateral GA group. Conversely, mean retinal sensitivity significantly decreased only in bilateral GA group.
Changes in retinal sensitivity and choroidal thickness differed in patients with GA with or without CNV in the fellow eye. These results identify at least two GA phenotypes, in which the development and progression of GA may be primarily due to different pathophysiologic mechanisms.
Pilotto, E., Guidolin, F., Convento, E., et al. British Journal of Ophthalmology. August 2015. 99: 1082-1086.
Association of Nonarteritic Ischemic Optic Neuropathy With Obstructive Sleep Apnea Syndrome: Consequences for Obstructive Sleep Apnea Screening and Treatment
The purpose of this study was to determine the prevalence of obstructive sleep apnea syndrome in patients who have had nonarteritic anterior ischemic optic neuropathy in one eye as well as to determine the risk of the fellow eye becoming affected.ARMD.
89 of 118 patients with a history of unilateral NAION, which was determined on sudden onset of painless loss of vision and/or part of the visual field, clinical exam findings and laboratory results, were examined from 2003 to 2012 with polysomnography to determine the presence of OSAS. Follow up was done 1 month, 3 months, 6 months, and 12 months after initial onset of symptoms and annually thereafter to determine fellow optic nerve involvement.
75% (67) of the 89 patients who underwent polysomnography tested to have OSAS. Of the 73 patients that were followed for a total of 3 years, 13.7% (10) were shown to have fellow eye involvement, 8 with OSAS and 2 without OSAS. The risk for fellow eye involvement in patients who tested for severe OSAS, which is classified by greater than 30 events per hour, that were non-compliant with ventilation treatment with positive airway pressure was shown to increase with a 95% confidence interval.
This investigation showed that OSAS is common in patients with NAION and that polysomnography should be done in these patients. Additionally, patients with severe OSAS who are non-compliant with treatment are at an increased risk for fellow optic nerve involvement.
Aptel F, Khayi H, Pépin JL, et al. JAMA Ophthalmol. July 2015; 133(7):797-804
Association Between Anatomical Resolution and Functional Outcomes in the Mivi-Trust Studies Using Ocriplasmin to Treat Symptomatic Vitreomacular Adhesion/Vitreomacular Traction, Including When Associated with Macular Hole
The purpose of this study was to evaluate visual function in patients with symptomatic vitreomacular adhesion (VMA)/vitreomacular traction including when associated with macular hole after ocriplasmin treatment, and the association between resolution of the underlying condition and improvement in visual function.
Six hundred and fifty-two patients from 2 Phase 3 trials received a single intravitreal injection of ocriplasmin 125 μg (n = 464) or placebo (n = 188).
Overall, 42% of patients who achieved VMA resolution at Day 28 had a ≥2-line improvement in best-corrected visual acuity at Month 6, and 20% had a ≥3-line improvement. Likewise, 69% of patients with nonsurgical full-thickness macular hole closure at Day 28 had a ≥2-line improvement at Month 6, and 48% had a ≥3-line best-corrected visual acuity improvement. Mean improvements in 25-item Visual Function Questionnaire scores were associated with achieving VMA resolution and nonsurgical full-thickness macular hole closure.
The authors concluded that in patients with symptomatic VMA/vitreomacular traction, VMA resolution and nonsurgical full-thickness macular hole closure were each associated with improvements in visual function. Resolving the underlying anatomical condition in symptomatic VMA/vitreomacular traction will increase the probability of achieving a clinically meaningful improvement in visual function.
Gandorfer A, Benz MS, Haller JA, et al. Retina. 35 (6): 1151–1157, June 2015.
Effects of posterior vitreous detachment on aqueous humor levels of VEGF and inflammatory cytokines
The purpose of this study was to investigate the association of PVD with aqueous levels of VEGF and other inflammatory cytokines. 98 and 80 eyes undergoing cataract extraction were sampled for aqueous humor concentrations of VEGF and other cytokines, respectively. Eyes were divided into two groups based on either presence or absence of PVD. The concentrations of VEGF cytokines were compared between the two groups.
Multiple regression analysis revealed that the aqueous concentrations of both VEGF and other inflammatory cytokines were significantly lower in the eyes with PVD than without; this finding was independent of age, sex and axial length.
Because aqueous humor levels of cytokines were previously reported to correlate with vitreous levels, it is rational to speculate the VEGF concentration in vitreous is higher in the eyes without PVD than in the eyes with PVD.
Although the pathogenic association of PVD and the lower concentration of these cytokines remain to be clarified, concentrations of VEGF and other inflammatory cytokines appear to be affected by the presence or absence of PVD. One may speculate that this relationship may have an effect on the treatment outcome of exudative AMD, and possibly macular edema due to branch retinal vein occlusion.
Takahashi, H., Nomura, Y., Tan, X. et al. British Journal of Ophthalmology. August 2015. 99: 1065-1069.
Diagnostic Sensitivity of Indocyanine Green Angiography for Birdshot Chorioretinopathy
The purpose of this article was to determine the sensitivity of indocyanine green angiography in patients with Birdshot Chorioretinopathy but who do not display any birdshot lesions.
3 patients with a history of BSCR that only displayed retinal vasculitis, low-grade to moderate vitritis, and hypocyanescent lesions on ICGA but not detectable on fluorescein angiography were examined from 2007 to 2014. All patients were positive for HLA-A29.
This retrospective study suggests that BSCR choroidal lesions may manifest on ICGA before choroiditis and retinitis is evident on clinical exam as well as characteristic macular edema. Therefore, it is beneficial that in patients with retinal vasculitis, optic disc edema, low-grade vitritis and other clinical findings suspicious of BSCR, an ICGA is performed to help diagnose and initiate treatment earlier on in the course of the disease.
Reddy AK, Gonzalez MA, Henry CR, et al. JAMA Ophthalmol. July 2015; 133(7):840-843.

|
ANSWER
TO "YOU MAKE THE DIAGNOSIS"
Derek MacDonald, OD, FAAO
ORS Fellow
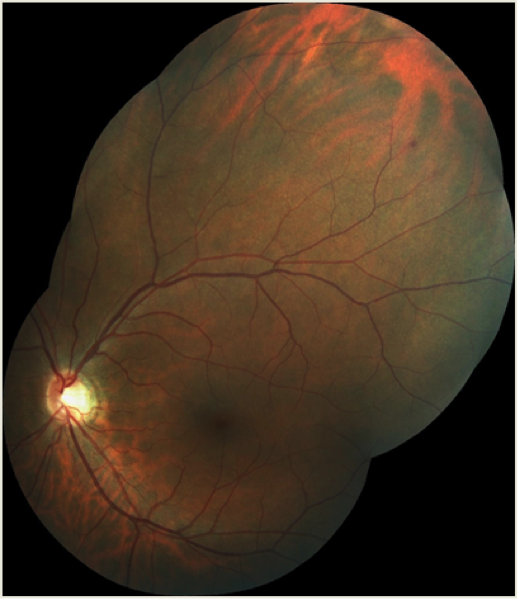
This photo represents commotio retina in a 39-year old Caucasian male with a previously uneventful ocular history approximately 18 hours after being hit on the left side of the face by a soccer ball kicked from close range. He reported an immediate and persistent disturbance in the inferior field of the left eye, but denied any central blurring, diplopia, pain, floaters, or photopsia. Entering corrected acuity was 6/6 in both eyes, albeit with an inferior-central ‘smudge’ in the left. Confrontation visual fields were full. Aside from the retinal appearance as noted, including two small peripheral retinal hemorrhages, the balance of the exam, including anterior segment, was normal. At scheduled follow-up in two weeks, the inferior field disturbance had completely resolved. His objective status remained stable with the exception of the expected near-complete resolution of the commotio retinae and associated retinal hemorrhaging. (see photo below).
Vision is typically unaffected in eyes with optic disc melanocytomas; however, decreased visual acuity may occur in some rare cases. Causes for this may include
secondary optic neuropathy, optic disc edema, spontaneous tumor necrosis, juxtapapillary or foveal choroidal neovascularization, subretinal fluid from tumor
exudation, cystoid retinal edema, retinal traction, epiretinal membrane, central retinal vein occlusion, or, perhaps most concerning, malignant transformation
of the tumor.2,8,9
Commotio retinae is a transient and usually self-limiting whitening of the neuroretina following contrecoup concussive closed-globe injury. , , It was first (and incorrectly) described in the late 1800s as Berlin’s edema when involving the macula. Subsequent histological studies revealed the pathophysiology to involve disruption of the photoreceptor outer segments (OS) and retinal pigment epithelium (RPE), with little retinal edema. , The relatively weak inter-digitation of the photoreceptor OS and RPE seems particularly susceptible to concussive injury, with immediate OS disruption followed by remote RPE phagocytosis of OS material, and potential permanent RPE damage in cases of more severe trauma. Despite blunt trauma being a common cause of visual impairment, the incidence of commotio retinae is unclear, likely due to its typically self-limiting nature.
The advent of spectral domain OCT and adaptive optics has allowed in-vivo confirmation of previous histological observations, and detection of subclinical retinal disruption. , Particularly when the macula is involved, OCT has become an invaluable tool for delineating the extent of outer retinal damage, and in conjunction with presenting visual acuity, aids in predicting the extent of visual recovery.
OCT imaging of mild to moderate commotio retinae typically shows hyper-reflectivity and/or disruption of the photoreceptor inner/outer segment (IS/OS) junction that is often reversible. Defects involving inner retinal structures (including photoreceptor IS and/or external limiting membrane damage, and migration of disorganized RPE toward a degenerating outer nuclear layer) following more significant trauma tend to be permanent, and if central portend poor recovery of visual acuity.
In most cases, including this one, peripheral commotio retinae resolves without sequelae. Significant trauma, however, may cause permanent macular damage not currently amenable to any treatment in up to one-third of patients. Careful assessment of anterior segment, macular, and peripheral retinal integrity at presentation, and diligent lifelong monitoring for remote complications including but not limited to cataract and angle recession glaucoma, are critical components in the care of any patient presenting with a history of blunt ocular trauma.
References
Williams DF, et al. Posterior segment manifestations of ocular trauma. Retina 1990;10:S35-44.
Atmaca LS, et al. Changes in the fundus caused by blunt ocular trauma. Ann Ophthalmol 1993;25:447-52.
Youssri AI, et al. Closed-globe contusion injuries of the posterior segment. Int Ophthalmol Clin 2002;42:79-86.
Berlin R. Zur sogenannten commotio retinae [So-called commotio retinae]. Klin Monatsbl Augenheilkd 1873;1:42-78.
Sipperley JO, et al. Traumatic retinopathy in primates. The explanation of commotio retinae. Arch Ophthalmol 1978;96:2267-73.
Mansour AM, et al. Histopathology of commotio retinae. Retina 1992;12:24-8.
Blanch RJ, et al. Neuroretinal cell death in a murine model of closed globe injury: pathological and functional characterization. Invest Ophthalmol Vis Sci 2012;53:7220-6.
Misko M. Ocular contusion with microhyphema and commotio retinae. Optometry 2012;83:161-6.
Bradley JL, et al. Ultra-high-resolution optical coherence tomographic findings in commotio retinae. Arch Ophthalmol 2011;129:107-8.
Stepien KE, et al. Subclinical photoreceptor disruption in response to severe head trauma. Arch Ophthalmol 2012;130:400-2.
Blanch RJ, et al. Visual outcomes after blunt ocular trauma. Ophthalmology 2013;120:1588-91.
Woo SJ, et al. Optical coherence tomography morphologic grading of macular commotio retinae and its association with anatomic and visual outcomes. Am J Ophthalmol 2013;156:994-1001.
Blanch, RJ (2014) Understanding and preventing visual loss in commotio retinae. Ph.D. thesis, University of Birmingham
|

IN THE
NEWS:
 |
Endophthalmitis rate low after anti-VEGF injections
The rate of endophthalmitis after intravitreal anti-VEGF injections is low, although the risk is not entirely eliminated, according to a study in Ophthalmic Surgery, Lasers and Imaging Retina.
The noncomparative consecutive case series assessed the incidence and outcomes of endophthalmitis after intravitreal anti-VEGF injections by reviewing patient records at Bascom Palmer Eye Institute from 2005 to 2014. MarketScan data were used to establish the population-based rate of endophthalmitis for 2011 to 2013.
Approximately one patient per 6,064 injections (0.016%) at Bascom Palmer and one patient per 1,895 injections (0.053%) from the MarketScan database were clinically diagnosed with endophthalmitis. Of the 20 eyes diagnosed at Bascom Palmer, eight occurred after Avastin (bevacizumab, Genentech) injections, six occurred after Lucentis (ranibizumab, Genentech) injections and six occurred after Eylea (aflibercept, Regeneron) injections.
Nine of the 20 cases were culture-positive — five Streptococcus species, three coagulase-negative Staphylococcus species and one non-anthracis Bacillus species — with final visual acuity ranging from 20/40 to no light perception. Final visual acuity for all 20 eyes ranged from 20/25 to no light perception.
|
 |
AKB-9778 with Lucentis improves central subfield thickness in DME
Patients with diabetic macular edema treated with a combination of AKB-9778 and Lucentis showed a significant reduction in central subfield thickness compared with patients treated with Lucentis alone, according to the Time-2 study results announced in a press release from Aerpio Therapeutics.
The three-armed phase 2a study included 144 patients with diabetic macular edema (DME) who were equally randomized to receive AKB-9778 (15 mg twice daily) as a monotherapy or in combination with Lucentis (ranibizumab, Genentech) therapy compared with Lucentis therapy alone. AKB-9778 is a human protein tyrosine phosphatase beta enzyme inhibitor. The treatment period was 3 months.
The study’s primary endpoint was mean change from baseline in central subfield thickness (CST), and secondary endpoints included visual acuity and safety outcomes.
Patients treated with the combination therapy had a “clinically significant benefit” in reduction of CST compared with patients treated with ranibizumab alone (P = .008), according to the release. As well, there was a positive trend in favor of the combination therapy vs. ranibizumab alone with regard to gaining three or more lines of visual acuity, Aerpio reported in the release.
“Most importantly, the combination approach has the potential to provide better outcomes for DME patients. We are actively planning the next stage of clinical development for this promising compound,” Joseph Gardner, CEO of Aerpio, said in the release.
|
 |
Quick exposure to hand-held laser device may lead to full-thickness macular hole
Momentary exposure to high-powered hand-held blue-light laser devices was associated with retinal injuries such as full-thickness macular hole, according to a study in the American Journal Of Ophthalmology.
The retrospective case series included 17 eyes of 17 patients with full-thickness macular holes caused by exposure to high-powered hand-held blue-light laser devices. Mean patient age was 18 years (range: 11 years to 30 years).Data were culled from a larger comprehensive study of laser-induced ocular injuries.
Duration of exposure to laser light was less than 1 second. Mean approximate distance from the laser device to the injured eye was 0.95 m. The interval from time of injury to presentation in a hospital ranged from 2 days to 16 months.
The main outcome was macular hole closure; the secondary outcome was final visual acuity. Mean Snellen best corrected visual acuity at presentation was 20/210. Mean macular hole minimum diameter was 351 µm (range: 168 µm to 620 µm).Fourteen eyes underwent 23-gauge pars plana vitrectomy, internal limiting membrane peeling, and gas tamponade (12 eyes) or silicone oil tamponade (two eyes). Mean follow-up after surgical intervention was 7 months.
Eleven of the 14 operated eyes (78.6%) had complete closure of the macular hole. Of the three unoperated eyes, one eye with the smallest macular hole diameter closed spontaneously. Mean final BCVA was 20/62 (range: 20/20 to 4/200.)
|
 |
Incidence of late AMD quadruples per decade of age in white Americans
The incidence of age-related macular degeneration increased exponentially with age in white American subjects, and women had a higher annual incidence of late AMD compared with men, according to a study in American Journal Of Ophthalmology.
The authors conducted a systematic review and meta-analysis of 74 prospective cohort studies published in Medline, Embase and Web of Science on the incidence and prevalence of AMD in white populations with European ancestry.
Fourteen publications based on 10 population studies contained relevant incidence data and were included for analysis.
The authors culled data on age- and sex-specific incidence of late AMD, geographic atrophy and neovascular AMD, year of study recruitment, AMD grading method and continent.
The annual incidence of late AMD in white Americans age 50 years and older, estimated from prevalence, was 3.5 per 1,000. The incidence was equivalent to 293,000 new cases among white Americans annually.
Annual incidence rates among people age 50 years and older were 1.9 per 1,000 for geographic atrophy and 1.8 per 1,000 for neovascular AMD.
Incidence rates roughly quadrupled per decade of age.The estimated total number of new cases rose rapidly from age 50 years to mid-80s and subsequently began to fall.
Late incidence of AMD was 38% greater among women than among men.
|
 |
Home monitoring program for high-risk AMD candidates may have public health impact
Broader implementation of a home monitoring program for detecting early AMD in patients at high risk for developing the disease may potentially impact public health in the U.S., according to an analysis presented at the European Society of Ophthalmology in Vienna. Carl D. Regillo, MD, discussed the potential public health impact of the Foresee Home AMD monitoring program, which uses preferential hyperacuity perimetry to help diagnose choroidal neovascularization (CNV) in its earliest stages.
Looking at the data from the AREDS2-HOME study, Regillo and colleagues estimated that, for the high-risk population using the Foresee program in addition to standard of care monitoring who develop CNV, 87% maintain a visual acuity of 20/40 or better, compared with 62% of patients who use only standard of care monitoring. This would result in approximately 101,000 more patients who would maintain vision.
Currently, it is estimated that only 36% of patients have 20/40 or better visual acuity following the initiation of treatment. Applying the results from the HOME study to the U.S. population at risk for developing late age-related macular degeneration (AMD), researchers estimate approximately 205,000 more people would maintain 20/40 or better vision.
These results are applied to estimates of the U.S. population at high risk of developing late AMD to estimate the potential public health effect of home monitoring with the device.
|
 |
Gevokizumab fails to meet primary endpoint in phase 3 trial for Behçet’s disease uveitis
Xoma today announced gevokizumab failed to meet its primary endpoints in a phase 3 trial in patients with uveitis caused by Behçet’s disease, according to a company press release.
The EYEGUARD-B study — a randomized, double-masked, placebo-controlled study of the efficacy of gevokizumab in the treatment of patients with Behçet’s disease uveitis — enrolled 83 patients with Behçet’s disease uveitis, with 40 patients randomly assigned to receive gevokizumab and 43 patients assigned to receive placebo. The primary endpoint was time to the first acute ocular exacerbation.
“Although the study did not achieve its main objective, we did see signals of drug activity such as preserved visual acuity, less severe ocular exacerbations and a reduced incidence of reported macular edema in patients treated with gevokizumab,” Paul Rubin, MD, senior vice president of research and development and chief medical officer for Xoma, said in the release. “We will continue to work closely with our partner, Servier, and uveitis experts to conduct a thorough analysis of the data to fully understand gevokizumab’s impact on several clinically relevant endpoints.”
|
 |
Topical squalamine shows encouraging results in phase 2 studies
Final results of the phase 2 IMPACT study of topical squalamine lactate in the treatment of neovascular AMD showed a trend toward increased efficacy in specific lesion types, according to a speaker at the American Society of Retina Specialists meeting .
Squalamine is a small molecule that counteracts multiple growth factors implicated in the angiogenic process, including VEGF and PDGF. The IMPACT study evaluated the effects of OHR-102 (0.2% squalamine lactate ophthalmic solution, Ohr Pharmaceutical) in combination with anti-VEGF in treatment-naïve patients with wet age-related macular degeneration (AMD) and vision between 20/40 and 20/320. Patients recruited across 23 U.S. sites received Lucentis (ranibizumab, Genentech) injection at day zero and then were randomized 1-to-1 to squalamine topical therapy or placebo eye drops administered twice daily. Patients were followed monthly and re-treated with ranibizumab as needed based on strict OCT-guided criteria. The number of re-treatments were similar between the two groups.
The topical therapy was generally well tolerated, it was reported. A phase 3 development program in a targeted population based on complete analysis of the IMPACT study results is under design.
|
 |
Ocata receives patents for retinal pigment epithelium transplant technology
Ocata Therapeutics has received three new U.S. patents that expand the scope of earlier patents for retinal pigment epithelium transplant technology, according to a company press release.
The expanded patent protection includes “all formats and formulations of RPE cells, including suspensions and sheets of cells, for use as therapeutic agents, and the use of these formulations for treating ophthalmic diseases such as dry age related macular degeneration and Stargardt’s macular degeneration,” according to the release.
The addition of these new patents brings the total number of Ocata’s patents to seven that “specifically protect retinal pigmented epithelial products derived from human embryonic stem cells as well as any other pluripotent stem cell source including induced pluripotent stem cell sources,” according to the release.
“For the first time a pluripotent stem cell derived therapy is about to enter a pivotal trial for Stargardt’s disease and phase 2 clinical trials for dry age-related macular degeneration, bringing the possibility of these long awaited therapies that much closer to reality,” Eddy Anglade, MD, chief medical officer of Ocata, said in the release
|
 |
Ophthotech reports net loss of $37.1 million in second quarter
Ophthotech Corporation announced a net loss of $37.1 million for the second quarter of 2015, according to a company press release
The loss, which translates to $1.08 per diluted share, compares with a net loss of $73 million, or $2.19 per diluted share, for the same period in 2014.
Collaboration revenue was reported at $1.6 million at the end of the quarter, compared with no collaboration revenue in the second quarter of 2014. Research and development expenses were reported at $32.1 million for the end of the second quarter, compared with $34.7 million for the second quarter of the previous year. The increase was primarily attributed to the company’s phase 3 clinical trial for Fovista, according to the release.
General and administrative expenses were $12 million at the end of the quarter, compared with $7.6 million for the second quarter last year. The increase correlates to an increase in costs to support Ophthotech’s expanded operations and public company infrastructure, the release said. |
| |
Regeneron reports $999 million in total revenue in second quarter
Regeneron reported $999 million in total revenue in the second quarter, a 50% increase compared with $666 million in the same quarter of 2014, according to a press release.
Net sales for Eylea (aflibercept) in the U.S. were reported at $655 million, a 58% increase compared with $415 million in the same quarter of 2014. Net sales for Eylea outside of the U.S. were reported at $338 million compared with $247 million in the same quarter of 2014.
Non-GAAP net income was reported at $328 million, or $3.29 per basic share and $2.89 per diluted share, in the second quarter compared with $289 million, or $2.88 per basic share and $2.47 per diluted share, in the same quarter of 2014.
GAAP net income was reported at $195 million, or $1.89 per basic share and $1.69 per diluted share, compared with $96 million, or $0.96 per basic share and $0.85 per diluted share, a year ago |

MEET
THE FELLOWS
In each issue, a Fellow of the Optometric Retinal Society is highlighted. In this issue, Dr. Derek MacDonald, a new ORS fellow, will be highlighted
Dr. Derek MacDonald graduated from the University of Waterloo School of Optometry in 1992, and began full-scope private practice in Waterloo, with special interest in ocular and systemic disease. He was elected to the Ontario Association of Optometrists Board of Directors in 1999, and served a two-year term as President that ended November 2007. He then assumed a staff position as a Policy Consultant, until being elected to the Council of the College of Optometrists of Ontario in 2010, and re-elected in 2012. Dr. MacDonald is a founding member of the Eye Health Council of Ontario, a Fellow of the American Academy of Optometry, and a member of the Optometric Glaucoma Society. When he’s not doing something optometry-related between October and April, he’s usually shoveling the snow from his driveway. Between April and October, he enjoys cycling through the Kitchener-Waterloo countryside in the early morning, and wondering why he bothers playing golf in the early afternoon.
|
WHY
BECOME AN ORS MEMBER?
By Bill Denton, O.D., F.A.A.O.
Chair, Membership Committee
At some point in your career, you realize you just may be coasting. Your knowledge has been limited to the journals you receive and attempt to read, and the conferences that may not be as fulfilling as they once were. You simply need a challenge that will add an extra dimension to your professional learning.
Fellowship in the Optometric Retina Society (ORS) can provide several benefits in addition to the initial challenge of qualifying for this honor. Plenty of perks accompany your induction, but the coolest part is being associated with a body of knowledge and resources which can help you in many other ways. It is not uncommon to receive weekly thought-provoking emails about challenging cases and treatment dilemmas. Some fellows like to share their awesome cases they have diagnosed, while others post their cases with hopes that other Fellows will suggest an alternative differential diagnosis. At times it is like a round-table of brainstorming, but through the use of modern technology. Fellowship has little obligation with a huge opportunity for professional growth.
If you are up to the challenge of becoming a Fellow of the ORS, feel free to peruse the details and application at www.optometricretinasociety.org. Advice can be given to assist you in your quest. Feel free to contact us. |

SPONSOR NEWS


Editor
in Chief
Steven Ferrucci, OD, FAAO
Co-Editor
Mark T. Dunbar, OD, FAAO |
Journal
Reviewers
Philip Kim, OD
Alison Bozung, OD
Leslie Small, OD
Paul Hammond, OD
Nabila Gomez, OD
Elliot Brafman, OD
Senior Graphic Designer
Matt Egger
|
Review of Optometry® is published by the Review Group, a Division of Jobson Medical Information LLC (JMI), 11 Campus Boulevard, Newtown Square, PA 19073.
To subscribe to other JMI newsletters or to manage your subscription, click here.
To change your email address, reply to this email. Write "change of address" in the subject line. Make sure to provide us with your old and new address.
To ensure delivery, please be sure to add revoptom@lists.jobsonmail.com to your address book or safe senders list.
Click here if you do not want to receive future emails from Review of Optometry. |
|