| |
Volume 14, Number 2 |
June 2018 |
|
|
Inside
This Issue |
|
|
|
|
|
This e-newsletter is provided free to doctors through industry support from |
 |
| |
FROM
THE DESK OF THE EDITOR
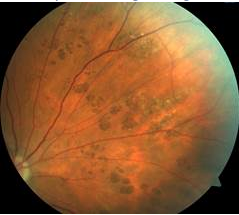
In the last newsletter, we featured a poll asking about the association between bear tracks and colon polyps and/or cancer. The subject came up in clinic between myself and a few other faculty optometrists just a few months prior. What type of presentation of CHRPE (congenital hypertrophy of the retinal pigment epithelium) necessitates a colonoscopy workup? A quick survey of a few colleagues revealed slightly different responses. A literature search was also unclear. Historically, the thought was that multiple CHRPEs or bear tracks necessitate a workup to rule out familial adenomatous polyposis (FAP). Others believe that the type of retinal lesions associated with FAP have a much different appearance than CHRPE. The majority of those surveyed in our last newsletter poll felt that there was an association between bear tracks and colon polyps/cancer. The patient featured in this picture is middle-aged and had a normal colonoscopy in the past.
But…she has a strong family history of colon cancer (three family members). Coincidence? Unfortunately, I don’t have an answer as to which patients need to be worked up. But I appreciate all your input as I continue to scratch my head in contemplation.
Have a happy summer! It is already miserably hot here in Indiana!
Anna Bedwell, OD, FAAO
Editor-in-Chief
POLL RESULTS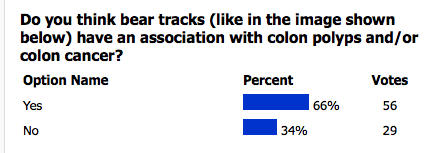
|
PRESIDENT'S MESSAGE
Summer is upon us. For me in California, that means warm weather, vacations to exotic locations, the beach, blockbuster movies and kids home from school driving us crazy!! But I still wouldn’t trade it for the world. It also means not as many conferences, and fewer work responsibilities, so hopefully a little extra time to relax as well as catch up on things. One thing I am particularly interested in learning more about are some of the new widefield imaging systems as well as some of the new analytics recently released for OCT angiography. Talk about fun summer reading!
After the summer, it’s time to start thinking about getting those needed hours to renew your license and perhaps travelling to a meeting or two. We are currently putting the finishing touches on our annual ORS meeting, RETINA UPDATE 2018, held November 30 to December 1 at the Fairmont Scottsdale in lovely Scottsdale, Arizona. The conference will include 11 hours of CE, including a two-hour hands on technology workshop, and will have wonderful speakers such as Jeff Gerson, Leo Semes, Mohammed Rafieetary, Brad Sutton and myself. Topics will include macular disease, diabetic eye care, and a lecture from a retinal specialist on therapies for genetic retinal diseases. So if you are interested in retina, need hours or just want to go to Scottsdale, please consider joining us. We would love to have you!
Sincerely,
Steven Ferrucci, OD, FAAO
ORS President

YOU
MAKE THE DIAGNOSIS
Answer appears later in newsletter.
Answer appears later in newsletter.

Image Gallery
Which of the following sets of images represents Leber’s Congenital Amaurosis?
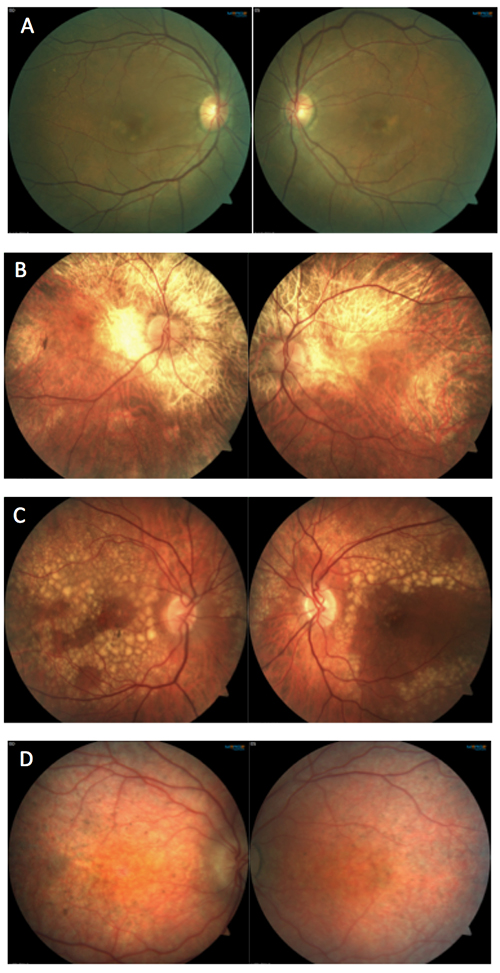
Answer appears later in the newsletter.
|

JOURNAL
ABSTRACTS
Posterior Vitreous Detachment as Observed by Wide-Angle OCT Imaging
Posterior vitreous detachment (PVD) occurs with increased gel liquefaction and declining vitreoretinal adhesion. Most OCT imaging is restricted to viewing the macula and optic nerve, leaving the peripheral retinal unexamined. The current study employed wide-angle, montaged OCT images to study the vitreoretinal interface and the process of PVD in its entirety.
The study enrolled 144 healthy eyes of 98 normal subjects to undergo wide-angle montage OCT images of the complete, obtainable vitreoretinal interface. The subjects ranged in age from 21 to 95 years (mean 51.4±22 years) and had refractive error between +3D and -5D. The group classified the eyes into 5 stages: stage 0, no PVD; stage 1, peripheral PVD (paramacular, peripheral); stage 2, perifoveal PVD extending into periphery; stage 3, peripapillary PVD still attached at optic nerve head; stage 4, complete PVD.
Only two eyes were classified as stage 0 (both were age 21). There were 88 eyes that fell into stage 1 (mean age 38±16.2 years). In stage 2, there were 12 eyes (mean age 67.9±8.4 years). This decreased in stage 3 to 7 eyes (mean age 70.9±11.9 years). Lastly, 35 eyes were classified as stage 4 (mean age 75.1±10.1 years). The authors found that PVD started in the paramacular-peripheral region rather than the traditionally thought perifoveal location. Furthermore, the first signs of PVD occurred in a younger population, starting in the third decade of life. The authors also found vitreoschisis (splitting of the posterior vitreous cortex) as a sign of initial PVD development in 40% of the studied normal eyes. Though this study was limited by the number of eyes, it provided excellent insight into the vitreoretinal interface, particularly in the paramacular to peripheral location. Further study with wide-angle OCT will be essential to a better understanding of PVD.
Tsukahara M, Mori K, Gehlbach PL, et al. Posterior vitreous detachment as observed by wide-angle OCT imaging. Ophthalmology. 2018; Apr 6. doi: 10.1016/j.ophtha.2018.02.039. [Epub ahead of print].
Prevalence of Reticular Pseudodrusen in an Elderly UK Caucasian Population—The Bridlington Eye Assessment Project (BEAP): a cross-sectional study (2002–2006)
The Bridlington Eye Assessment Project (BEAP) studied reticular pseudodrusen (RPD) in a UK population, and analyzed the prevalence, associations and risk factors. Participants were all age 65 or older. Color fundus photographs were taken of each eye and adjusted to the red-free and blue channels. All images were analyzed by reviewers for characteristics of RPD. To be considered RPD, there had to be round/oval lesions (non-confluent) with uniform size and color that were more appreciable on photo enhancement of the red-free channel. Distribution of RPD was recorded using a superimposed ETDRS grid centered at the fovea.
A total of 3,476 participants had gradable photos in at least one eye. Overall, RPD was appreciable in 281 eyes (176 participants) equating to a prevalence of 5.06%. In 44/281 (15.7%) of eyes, RPD were only appreciable after digital photo enhancement to the red-free channel. Mean age was 81.1 years and prevalence increased with age group from 1.18% (65 to 69 years group) to 27.27% (≥90 years old group). RPD was subcategorized as dot type, discrete dots in reticular pattern, in 18.5% of participants; ribbon type, appeared as interlocking ribbons, in 36.7%; and the rest were labeled mixed dot/ribbon. Age, female gender and history of diabetes mellitus were associated with higher risk of RPD whereas a history of hypertension treatment had a reduced risk of RPD. The location of RPD was most often outside the ETDRS grid (88%) followed by the outer superior subfield. RPD were present in 25.9% of those studied with stage 4 AMD.
In conclusion, the authors found that RPD occurred more often than previously thought. The prevalence in this study was much higher than in the Beaver Dam Eye Study (0.7% in age 43 to 86 years old) but had similar results to the Rotterdam Study (4.9% ages 65 and older). This study provided further insight into the understanding of RPD.
Wilde C, Poostchi A, Mehta RL, et al. Prevalence of reticular pseudodrusen in an elderly UK Caucasian population—The Bridlington Eye Assessment Project (BEAP): a cross-sectional study (2002–2006). Eye (Lond). 2018; Mar 1. doi: 10.1038/s41433-018-0049-8. [Epub ahead of print].
A Study of the Natural History of Vitreomacular Traction Syndrome by OCT
Vitreomacular traction syndrome (VMTS) is a result of persistent vitreomacular adhesion caused by anomalous PVD. This retrospective cases series examined the natural history of VMTS over a follow up of 17.4±12.1 months. In total, 183 eyes of 159 patients fit the criteria for VMTS, as set by the International Vitreomacular Traction Study (IVMTS) Group, and were free of confounding retinal disease. Surgery (vitrectomy) was offered for development of macular hole or for symptoms of distortion and/or decline in vision. Otherwise, patients were followed (over a minimum six months) for spontaneous resolution. OCT scans were graded by a single reading center.
Of the 159 patients with VMTS, 70% were female and the mean age was 72±10 years. In 15% (24) of patients, both eyes had VMTS. Focal (<1,500 µm) VMTS was more common at 87% over broad VMTS (≥1,500 µm). Macular pucker was noted in 37 eyes (20%) and had a significant association with broad adhesion. The majority of cases (60%) did not resolve over follow up. Spontaneous resolution occurred in 20% (on average at 15 months) while progression to full thickness macular hole (FTMH) occurred in 12%. Surgery for worsening symptoms was elected in 8%. Overall, visual acuity remained relatively stable. Vision improved with spontaneous VMTS resolution and worsened in cases with either development of FTMH or worsening symptoms that necessitated surgery.
A better understanding of the natural history of VMTS is necessary to properly educate patients on the possibility of progression vs. spontaneous resolution. This would allow for patients to properly make an informed decision when offering the alternative treatments of surgery, ocriplasmin injection or gas injection.
Errera MH, Liyanage SE, Petrou P, et al. A study of the natural history of Vitreomacular Traction Syndrome by OCT. Ophthalmology. 2018; May;125(5):701-7.
Consistent Long-Term Therapy of Neovascular Age-Related Macular Degeneration Managed by 50 or More Anti-VEGF Injections Using a Treat-Extend-Stop Protocol
In order to maintain functional vision, some patients with neovascular macular degeneration (wet AMD) receive a tremendous number of anti-VEGF injections over time. This retrospective chart review looked at individuals who most certainly fit that criteria, as every eye included had received at least 50 shots.
This group included 71 eyes of 67 patients who were managed with a treat-extend-stop protocol. All eyes underwent three anti-VEGF injections, most primarily with bevacizumab (64.5%), at four to six-week intervals. Treatment windows were then extended on a strict protocol as the macula remained free of fluid, and repeat treatments were also performed as necessary when fluid returned. On average, it took 6.5 years to reach the 50th injection, with patients receiving around nine injections per year. The mean initial visual acuity in all eyes was 20/80, and the mean visual acuity at the final follow up visit was 20/50. At the time of the 50th injection, 42.9% of eyes had visual acuity of 20/40 or better, 42.3% of eyes had decreased vision compared to their baseline, and 8.5% of eyes had lost three or more lines of acuity. Of these eyes that lost three or more lines of acuity, more than half of them (four total) had developed central geographic atrophy.
So what does all of this mean? Clearly, these eyes represented somewhat of a success story. It is almost hard to imagine that after they all received 50 or more injections, they had considerably better visual acuity on average than where they started. It is likely that by looking only at eyes that had received 50 or more shots, this became a somewhat self-selecting group for success. In eyes that do well with anti-VEGF therapy, it tends to be repeated when needed. In eyes that stop responding to therapy and get worse, therapy is eventually discontinued. In other words, these eyes received so many injections because they worked. That also likely explains the very small number of eyes that developed central geographic atrophy. If severe geographic atrophy forms and substantially decreases vision, then it is very likely that anti-VEGF therapy would be discontinued well before reaching 50 injections. At any rate, this chart review indicates that some eyes with neovascular AMD can exhibit substantial vision gains that are maintained after a tremendous number of injections.
Adrean SD, Chaili S, Ramkumar H, et al. Consistent long-term therapy of neovascular age-related macular degeneration managed by 50 or more anti-VEGF injection using a treat-extend-stop protocol. Ophthalmology. 2018; Feb 10. doi: 10.1016/j.ophtha.2018.01.012. [Epub ahead of print].
Retinal Microvasculature in Predicting Risk of Stroke Subtypes
The eye is the only organ in the body where vasculature can be directly visualized, thus allowing practitioners to appreciate the vascular sequelae of systemic disease. As such, researchers theorized that the status of retinal microvasculature could be used to assess stroke risk.
Lin, et al. performed a sub-analysis of data collected in the very large Atherosclerosis Risk in Communities (ARIC) study. That study included 10,468 people between the ages of 45 and 65, all of whom underwent retinal photography. In that complete cohort, 673 strokes were reported, of which 578 were ischemic and 95 were hemorrhagic. Individuals with abnormal retinal microvasculature were more than twice as likely to have suffered a stroke when compared with those with normal retinal vasculature. Four percent of those with abnormal retinas reported having had a stroke, while only 1.9% of those with normal retinas did so. Abnormal retinal microvasculature included arteriovenous nicking, arteriolar narrowing, retinal hemorrhages and micro-aneurysms. The association was most pronounced with lacunar ischemic strokes, and increasing amounts of retinal vasculopathy were related to higher stroke risk.
So what can be gleaned from this information? It is rather intuitive that abnormal retinal vasculature could be associated with a higher risk of stroke, as all vasculature in the body would presumably be experiencing the same damage that can be directly visualized in the retina. Having seen this presumption borne out in such a large patient cohort, eye care providers could feel confident in counseling patients and physicians regarding stroke risk as it pertains to retinal microvascular findings, which may in turn lead to improved systemic outcomes.
Lin MP, et al. Presented at the 2018 American Academy of Neurology meeting.
Optical Coherence Tomography Angiography in Pre-Clinical Alzheimer’s Disease
Optical coherence tomography (OCT) and optical coherence tomography angiography (OCTA) are utilized in the management of a very large number of ocular conditions. In addition, the technology is playing an ever increasing role in the detection and monitoring of systemic neurological disease as well. Conditions such as multiple sclerosis, Alzheimer’s disease, Parkinson’s disease and stroke can all affect OCT measurements. But can OCT and OCTA be utilized to detect preclinical disease?
Researchers at Washington University in St. Louis attempted to answer this question as it pertains to Alzheimer’s. They performed OCT and OCTA on 30 cognitively normal patients who had undergone biomarker screening for Alzheimer’s disease. The individuals involved had undergone either positron emission tomography (PET) scanning or cerebral spinal fluid (CSF) analysis within the last year. Patients with a history of trauma, ophthalmic disease, elevated IOP, diabetes, hypertension or high refractive error were excluded. Of the 30 individuals included, 16 were Alzheimer’s biomarker negative and 14 were biomarker positive. All but one of the 30 patients were Caucasian. The researchers found that the biomarker positive group had thinner inner fovea’s (66µm vs. 75.4µm) and an enlarged foveal avascular zone (.40mm2 vs. .30mm2) compared with the biomarker negative group.
They postulated that these findings might be the result of capillary deposition of amyloid and collagen, thus leading to cellular apoptosis and vascular dropout. This study certainly has some limitations, including a small sample size and lack of minority patients, so more research would need to be done to include a larger number of patients, more diverse patient populations and various sites. If the findings could be repeated under these circumstances, OCT and OCTA could become a valuable, noninvasive, inexpensive tool in the detection of preclinical Alzheimer’s disease.
Van Stavern G, et al. Presented at the 2018 American Academy of Neurology meeting.
Peripheral Changes Associated with Delayed Dark Adaptation in Age-related Macular Degeneration
Initial symptoms of age-related macular degeneration (AMD) include difficulty with night vision and moving from light to dark environments, even in the presence of normal visual acuity. Previous studies have suggested that impaired dark adaptation (DA) is strongly associated with AMD in the macula; however, AMD patients also develop peripheral changes, suggesting that AMD might not be limited to the macula. The goal of this study was to determine an association between peripheral changes in AMD and DA.
This prospective, cross-sectional study looked at 128 eyes of 72 patients; 32 eyes (25%) were controls, 19 eyes (14.8%) had early AMD, 64 eyes (50%) had intermediate AMD and 13 eyes (10.2%) had late AMD. Ultra-widefield (UWF) Optos pseudocolor photography and fundus autofluorescence (FAF) were performed on all eyes. FAF image patterns were categorized as follows: granular increased (small, discrete areas of bright hyperfluorescence), nummular decreased (small to medium areas of discrete, uniform hypofluorescence) and mottled decreased (areas of general hypofluorescence in an uneven/irregular pattern). DA was also performed over a 20-minute period on all patients and assessed with an AdaptDx dark adaptometer. The AdaptDx machine estimated the amount of time it took for visual sensitivity to become completely rod-mediated, termed rod-intercept time (RIT). The median time to dark-adapt was 4.3 minutes for control eyes, 6.4 for early AMD, 17.07 for intermediate AMD and 20 minutes for late AMD. Presence of reticular pigmentary changes in the midperiphery and far periphery were significantly associated with a more prolonged RIT. Presence of a mottled decreased FAF pattern in the midperipheral zone was also associated with impaired DA. No association was found between presence and number of peripheral drusen and RIT.
Dark adaptation is a promising functional outcome measure in AMD. This study appeared to be the first to assess and provide new insights into impairments in dark adaptation and peripheral changes in AMD. Further studies would be warranted to validate these findings.
Laíns I, Park DH, Mukai R, et al. Peripheral changes associated with delayed dark adaptation in age-related macular degeneration. Am J Ophthalmol. 2018; Jun;190:113-24.
Reperfusion of Occluded Branch Retinal Arteries by Transluminal Nd:YAG Laser Embolysis Combined with Intravenous Thrombolysis of Urokinase
Branch retinal artery occlusions (BRAO) and hemiretinal arterial occlusions (HRAO) result in retinal ischemia and sudden vision loss due to fibrin plaques, cholesterol plaques or calcified emboli becoming lodged in a retinal arteriole. Many treatment modalities have been attempted without success, though a few recent studies have shown successful restoration of retinal blood flow with Nd:YAG laser embolysis (TYE). The embolus is disintegrated by the laser; however, embolic debris still remains in the vasculature and may require further therapy. The goal of this study was to achieve reperfusion of the retina and improve therapeutic effect by administering fibrinolysis therapy via intravenous urokinase to further degrade a clot in addition to disrupting emboli with TYE.
This retrospective study looked at 34 eyes from 34 patients over a minimum of six months with a clearly visible obstruction point in acute BRAO or HRAO. An Nd:YAG laser was used to partially or completely disrupt the visible arterial embolus in all patients. Immediate thickening of the artery diameter and an alight reddening in the retinal occlusion zone were seen after TYE. Fluorescein angiography (FA) was then performed and results were divided into groups. Complete fluorescein filling of the artery and its branch vessels was defined as complete blood flow restoration; 2/3 fluorescein filling of the branch vessels was defined as mostly restored; 1/3 fluorescein filling of the branch vessels was defined as partially restored; and absence of dye filling any branch vessels was defined as ineffective.
Retinal blood flow on FA after TYE was completely restored in 13 eyes (38.2%), mostly restored in 11 eyes (32.4%), partially restored in five eyes (14.7%) and not effective in five eyes (14.7%). The day after TYE, 10 million to 20 million units of urokinase in 250mL of 0.9% sodium chloride solution were administered intravenously for 30 to 40 minutes twice a day for five days. After combined urokinase infusion therapy, the retinal occlusion zone became rosy, and blood flow on FA was completely restored in 16 eyes (47.1%), mostly restored in 15 eyes (44.1%) and partially restored in three eyes (8.8%). Visual acuity improved by two or more lines in 19 eyes (56%), four or more lines in nine eyes (26%) and six or more lines in six eyes (18%). There was no correlation with visual outcome and age, gender or type of occlusion.
Nd:YAG laser treatment combined with thrombolytic therapy had an improved therapeutic effect with further dissolution of emboli, rapid reperfusion of the retina and visual acuity improvement. TYE combined with urokinase thrombolysis should be considered an effective treatment for patients with visible emboli in BRAO or HRAO. Future studies would be needed with more cases and presence of a control group.
Chai F, Du S, Zhao X, Wang R. Reperfusion of occluded branch retinal arteries by transluminal Nd:YAG laser embolysis combined with intravenous thrombolysis of urokinase. Biosci Rep. 2018; Feb;38(1).

|
ANSWER
TO "YOU MAKE THE DIAGNOSIS"
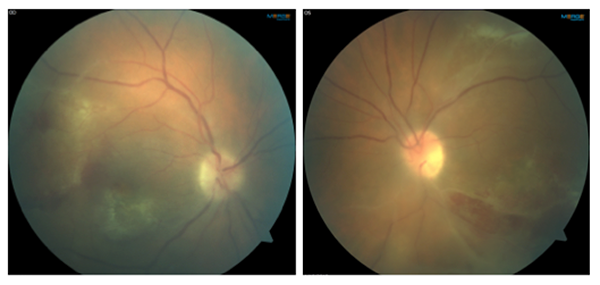
A 44-year-old Caucasian male presented to the clinic complaining of blurry vision OU and red eyes for the past month. He also noted photophobia and pain. The patient was HIV positive and had recently restarted HIV meds after not taking them for almost a year. He had a history of stroke four months prior. His last CD4 count was 20 to 50 (improved from zero), last viral load >100,000. 1
His visual acuities were 20/150 OD with pinhole to 20/80 and 20/400 OS with no improvement on pinhole. Pupils were round, equal and minimally reactive to light with a 3+ APD OS. Confrontation fields were full OD and restricted superior nasal OS. Goldmann IOP measured 19 mmHg OD and 22 mmHg OS. Slit lamp examination revealed diffuse conjunctival injection, multiple fine keratic precipitates on the endothelium, and 2+ cell and 1+ flare in the anterior chamber OU. Upon dilated fundus examination (photos above), both eyes had a dense vitritis, retinal hemorrhages, vascular sheathing and patches of retinal necrosis 360 degrees in the periphery and a few patches in the posterior pole.
Differential diagnoses included acute retinal necrosis (ARN), progressive outer retinal necrosis (PORN), CMV retinitis, toxoplasmosis and syphilis. Based on clinical picture, the patient was ultimately diagnosed with PORN and his anterior chamber tap was positive for varicella-zoster virus (VZV). He received multiple foscarnet and ganciclovir injections and the retinitis became quiescent. The visual outcome is unknown.
Both ARN and PORN are types of necrotizing herpetic retinitis, though there are some key differences between the two. ARN is caused by latent VZV, HSV-1 or HSV-2 infection and more commonly develops unilaterally in otherwise healthy individuals.1 ARN typically presents as peripheral retinal necrosis associated with moderate to severe vitritis and retinal vasculitis.1 Comparatively, PORN is almost exclusively caused by VZV, a double-stranded DNA virus of the herpes group.2 The virus remains latent in the trigeminal and dorsal root ganglia, but can be reactivated during times of loss of T-cell regulatory control. PORN affects immunocompromised patients, specifically individuals with AIDS. Ocular manifestations of PORN include bilateral multifocal peripheral retinal necrosis. PORN initially may have minimal to no vitreous or anterior chamber inflammation and vasculitis, but it can quickly progress and involve the entire retina, ultimately leading to blindness.3 Secondary complications include rhegmatogenous retinal detachment secondary to atrophic thin retina with multiple holes, and optic nerve head involvement including edema, hyperemia and atrophy.3 Treatment of PORN includes various combinations of IV, oral and intravitreal antivirals (ganciclovir, acyclovir, foscarnet), HAART and barricade laser.2
Katelyn Lucas
Optometry Resident
Indianapolis Eye Care Center
1. Moorthy RS, Weinberg DV, Teich SA, et al. Management of varicella zoster virus retinitis in AIDS. British Journal of Ophthalmology. 1997;81:189-94.
2. Gore DM, Gore SK, Visser L. Progressive outer retinal necrosis: outcomes in the intravitreal era. Arch Ophthalmol. 2012; Jun;130(6):700-6.
3. Schachat AP, Murphy RP. (1994). Retina: Volume Two (2nd Ed.). St. Louis, MO: Mosby-Year Book, Inc.
|

IN THE
NEWS
 |
FDA Approves Genentech’s Lucentis Prefilled Syringe
Genentech announced that the FDA approved a Lucentis® (ranibizumab injection) 0.3 mg prefilled syringe as a means to administer the medicine in the treatment of diabetic retinopathy and diabetic macular edema. The Lucentis 0.3 mg prefilled syringe is packaged in a sterile, single-use tray and is made of borosilicate glass.
In October 2016, the FDA approved Lucentis 0.5 mg prefilled synringe to treat neovascular AMD, macular edema from retinal vein occlusion and myopic CNV. Genentech anticipates that the Lucentis 0.3 mg prefilled syringe will be available in the second quarter of this year.
|
 |
Bascom Palmer Receives $12 Million Gift to Create Research Center
Philanthropist Lois Pope is donating $12 million to Bascom Palmer Eye Institute in memory of her mother who went blind from macular degeneration. The gift will go towards establishing the Lois Pope Center for Retinal & Macular Degeneration Research on the Palm Beach Gardens campus. The gift marks the single largest donation to Bascom Palmer in its 56-year history.
|
 |
Arizona College of Optometry Starts Military Residency
Arizona College of Optometry (AZCOPT) has teamed up with the Department of Defense to create a new military optometric residency program. The one-year program, starting July 1, will be located at the Walter Reed National Military Medical Center. The program will enable educational training in ocular disease, vision rehabilitation and primary care for active duty optometrists from any service branch. Midwestern University and AZCOPT will oversee and help to obtain accreditation.
|
 |
GenSight Biologics Announces Phase III Clinical Trial Results for GS010 in LHON
The topline results from the Phase III REVERSE study were announced by GenSight Biologics. The trial assessed the safety and efficacy of GS010 by single intravitreal injection to treat Leber’s Hereditary Optic Neuropathy (LHON). Though the study failed to meet its primary endpoint, which was improvement in vision of gene therapy treated eyes over sham treated eyes, it still reported favorable results. Both the GS010 eyes and sham eyes improved +11 ETDRS letters over baseline at 48 weeks. Researchers were pleasantly surprised to see an improvement in the contralateral, untreated eye. In terms of safety, the most common adverse effects of GS010 were related to the injection itself. Additionally, there were some cases of intraocular inflammation, which were controlled without issue. There are two other ongoing phase III trials of GS010: RESCUE and REFLECT.
|
 |
Optos Releases Monaco (Ultra-widefield Imaging Device) to US Market
Monaco, the first combination ultra-widefield imaging device and OCT, has been introduced into the U.S. market. Monaco, a product of Optos (Nikon Corp.), can produce a 200-degree optomap image in a half-second single capture. The compact, desktop device has the ultra-widefield capabilities of color imaging, 3-in-1 color depth imaging, autofluorescence in addition to central pole OCT.
|
 |
FDA Approves Fast Track Designation for MeiraGTx’s AAV-RPGR Treatment of X-linked RP
Gene therapy developer MeiraGTx was granted Fast Track designation for AAV-RPGR to treat X-linked retinitis pigmentosa (RP) by the FDA. AAV-RPGR is designed to target the RPGR gene, specifically RPGR-ORF 15. This defect is the most common cause of X-linked RP. Currently, AAV-RPGR is in Phase I/II clinical trials of adult and pediatric patients with the RPGR-ORF 15 mutation. Additionally, MeiraGTx is in enrollment for a natural history study of patients with X-linked RP.
|

|
IMAGE QUIZ ANSWER
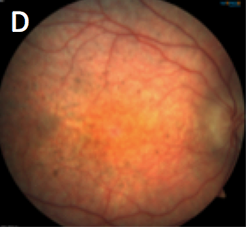
Image D in the gallery represents Leber’s Congenital Amaurosis (LCA), a congenital retinal dystrophy resulting in profound vision loss. LCA is inherited in an autosomal recessive pattern. There are several (at least 20) different gene mutations that can cause LCA. Children with LCA initially have a normal fundus appearance but eventually develop retinal changes such as: chorioretinal degeneration, bone spicules, subretinal flecks and/or optic disc pallor. Generally, the condition is diagnosed in infancy or early childhood based on signs of nystagmus, strabismus, sluggish pupil response, reduced visual acuity and the oculo-digital sign of Franceschetti. Scotopic and photopic electroretinogram is severely reduced or nonrecordable.
A: Butterfly pattern dystrophy B: Choroideremia
C: Doyne’s honeycomb dystrophy
|
WHY BECOME AN ORS FELLOW?
By Bill Denton, O.D., F.A.A.O.
Chair, Membership Committee
At some point in your career, you realize you just may be coasting. Your knowledge has been limited to the journals you receive and attempt to read, and the conferences that may not be as fulfilling as they once were. You simply need a challenge that will add an extra dimension to your professional learning.
Fellowship in the Optometric Retina Society (ORS) can provide several benefits in addition to the initial challenge of qualifying for this honor. Plenty of perks accompany your induction, but the coolest part is being associated with a body of knowledge and resources which can help you in many other ways. It is not uncommon to receive weekly thought-provoking emails about challenging cases and treatment dilemmas. Some fellows like to share their awesome cases they have diagnosed, while others post their cases with hopes that other Fellows will suggest an alternative differential diagnosis. At times it is like a round-table of brainstorming, but through the use of modern technology. Fellowship has little obligation with a huge opportunity for professional growth.
If you are up to the challenge of becoming a Fellow of the ORS, feel free to peruse the details and application at www.optometricretinasociety.org. Advice can be given to assist you in your quest. Feel free to contact us.
|
SPONSOR NEWS

Editor
in Chief
Anna K. Bedwell, OD, FAAO
Co-Editor
Brad Sutton, OD, FAAO |
Journal
Reviewers
Katelyn Lucas, OD
Senior Graphic Designer
Matt Egger
|
Review of Optometry® is published by the Review Group, a Division of Jobson Medical Information LLC (JMI), 11 Campus Boulevard, Newtown Square, PA 19073.
To subscribe to other JMI newsletters or to manage your subscription, click here.
To change your email address, reply to this email. Write "change of address" in the subject line. Make sure to provide us with your old and new address.
To ensure delivery, please be sure to add revoptom@lists.jobsonmail.com to your address book or safe senders list.
Click here if you do not want to receive future emails from Review of Optometry. |
|