| |
Volume 11, Number 4 |
November
2015 |
|
|
Inside
This Issue |
|
|
|
|
|
This e-newsletter is provided free to doctors through industry support from |
 |
| |
FROM
THE DESK OF THE EDITOR
Recently, I did a presentation with a friend of mine, Paul Chous. Paul is an Optometrist in the Tacoma, Washington area who has a private practice that specializes in taking care of patients with diabetes. He arguably knows more about diabetes than any OD I know, and perhaps more than many MDs. He is very passionate about his causes, and is a very good lecturer, whom I recommend you listen to if you have not already. Anyway, he told me we should not refer to our patients as “diabetics” , but rather as “patients with diabetes”, as we should not define their whole being by their disease. I mean, would we refer to a patient with AMD as a “macular degenerate”! Anyway, it really got me thinking, and defining a patient by one single aspect of their health is probably inappropriate, or insensitive at the least. I have since tried to change my nomenclature, but it is certainly not that easy. So, thanks, Paul, for the education. I promise I will try!
LIVE
POLL
|
How do you utilize your OCT?
|
|
Please look at the example later in the newsletter of some research that one of our new ORS fellows, Ava Bittner, has done. It’s great to see an OD doing such important, high-level research! Also, she serves as a mentor to optometry students, and helps instill in them the importance of research, education and life-long learning. Kudos Dr. Bittner.
Lastly, it’s not too late to register for the ORS Meeting Dec 4-5 in Anaheim CA. Co-sponsored with Review of Optometry, this meeting will feature lectures from ORS fellows as well as a local retinal specialist in a wide array of topics, from AMD to genetics, retinal imaging and more. Love to see you there!
Steven Ferrucci, O.D., F.A.A.O.
Editor in Chief
ORS/REVIEW OF OPTOMETRY MEETING
We are excited about bring the ORS/Review of Optometry Retina Update back to the west coast this year!
A distinguished slate of speakers from both optometry and ophthalmology will present clinically relevant information on
vitreoretinal disease prevention, diagnosis, technology, and treatment/management. Don't miss optometry's premier posterior
segment educational event as we focus on providing education and insight into best retina practices in 2015.
Click here for more information and to register.
Joe Pizzimenti O.D., F.A.A.O.
Conference Co-chair
ORS Fellow
Past-President
|
PRESIDENT'S MESSAGE
As the Thanksgiving Holiday approaches, it is an excellent time to reflect upon all that we have to be thankful for. For me personally, I have more than my share of blessings to count. In my personal life, I do not even know where to begin. I have an outstanding wife who has been my better half for over 21 years. She also happens to be a terrific optometrist as well, and it is truly special to be able to “talk shop” with your soulmate and best friend. I have two wonderful teenage daughters who, in spite of an affinity for drama, make Julie and me very proud. I also have a terrific extended family, both my own, and the one in to which I married. Not everyone is so fortunate. Professionally, we are all blessed to have been called to a profession that makes such a difference in people lives, day in and day out. I am personally grateful to work for a great University, allowing me to not only provide for my family’s needs, but also allowing me to impact the lives of countless future doctors over the years. In a way, when those future doctors go on to graduate, they take a small piece of us with them. When they care for their patients, we care for their patients too. How cool is that? It is truly something to be thankful for indeed.
AUGUST 2015
POLL RESULTS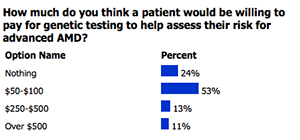
|
In Europe, OCT instruments are being utilized to perform a version of "dyeless angiography", and the
first commercial Swept Source OCT is being marketed. It is hard to conceive of providing high quality patient
care without the bells and whistles available to us today. But you know what else I am thankful for? I am
thankful that I began practicing BEFORE most of these instruments were available.
Brad Sutton, O.D., F.A.A.O.
ORS President

YOU
MAKE THE DIAGNOSIS
Answer appears later in newsletter.

CLINICAL
PEARLS
Look for underlying systemic clues during your DFE
By Diana Shechtman, OD, FAAO
ORS Fellow
The importance of discovering underlying systemic disease through a dilated retail exam should not be minimized. While we are all familiar with looking for things such as diabetic and hypertensive retinopathy, a myriad of other retinal findings may also clue the clinician into underlying systemic diseases. As an example, ROTH spots, classically associated with bacterial endocarditis and pernicious anemia, can also be associated with many other disease processes, such as leukemia, as an example. Further, a comprehensive retinal evaluation often does not require additional equipment such as an OCT, but can be done with equipment already available, most notable condensing lenses, an indirect ophthalmoscope and slit lamp. Lastly, co-managing these patients with primary care providers can be rewarding, and demonstrate your ability to play a part in your patients’ overall health.
Which agent is best for DME? A look at DRCR.net Protocol T
By Steven Ferrucci, OD, FAAO
ORS Fellow
Recently , the results of a study by the Diabetic Retinopathy Clinical Research Network evaluating the role of anti-vascular endothelial growth factors drugs for diabetic macula edema (DME) was published. This study, called Protocol T, was designed , to compare ranibizumab ( Lucentis), bevacizumab (Avastin) and aflibercept (Eylea )head to head in the treatment of DME . The study was conducted at 89 sites, with 660 patients receiving either 2 mg of Eylea 1.25 mg, Avastin, and 0.3mg Lucentis as often as every 4 weeks.
Looking at the study group overall, it appeared the Eylea gave slightly better visual acuity result overall , with the mean visual acuity score improving by 13.3 letters, vs. 9.7 with Avastin and 11.2 with Lucentis; however, the results were not statistically significant. However, when the researchers looked at patients with worse visual acuity, the differences emerged. In patients with acuity worse than 20/50, the mean improvement with Eylea was 18.9 vs. 11.8 with Avastin and 14.2 with Lucentis. While the overall difference does not seems that great at first glance, the authors went on to explain that in eyes with worse vision at baseline, if you were treated with Eylea, you have a 63% better chance of gaining 3 lines of acuity than if treated with Avastin, and a 34% better chance than with Lucentis.
Based on the results of this study, many retinal specialist are therefore using Eylea as their drug of choice in patients with DME whose vision is worse than 20/50, despite the increased cost over Avastin. Year 2 results will be out next year, and may shed even more light on the best agent to use in our patients with DME.
Peripheral Retina Examination
Joseph Pizzimenti, OD, FAAO
ORS Fellow
This morning, I conducted a lab for optometry residents to hone their peripheral retina examination skills. The first step is to assemble the proper tools of the trade: dilating drops, slit lamp, contact and non-contact fundus lenses, BIO, and scleral depressor. The 60 D, 78 D, 90 D and super-field lenses offer good stereopsis and an inverted, reversed image of the fundus. The 90-diopter lens has the lowest magnification and the widest field of view. For BIO, 25 and 28 D lenses offer a wide field of view. You may also want to try out some of the newer generation slit lamp lenses such as Volk's Super VitreoFundus®. The 3-mirror contact fundus lens is a longtime favorite of mine for viewing all the way out to the pars plana.
For both slit lamp funduscopy and BIO, the patient should be positioned such that they are comfortable. Encourage your patients to use the handles on the slit lamp to ensure that their forehead and chin are correctly positioned. The intensity of the light source should not be too bright as to cause marked photophobia.
Scleral depression allows the clinician to view the peripheral fundus in profile. I find it helpful to explain to the patient beforehand that the technique will be a bit uncomfortable, as they will no doubt feel pressure on the globe. I also tell them that there will be periodic breaks during the procedure. I have the patient first look in the direction opposite where I indent, then ask them patient to look "into the pressure."
For more detailed "back to the basics" information on these techniques, check out the Lost Arts of Optometry series, beginning here: http://www.reviewofoptometry.com/issue_toc/i/2520/.
|

JOURNAL
ABSTRACTS
Image Artifacts In Optical Coherence Tomography Angiography.
The purpose of this article was to describe image artifacts of optical coherence tomography (OCT) angiography and their underlying causative mechanisms.
Artifacts are very common and can arise from the OCT image acquisition, intrinsic characteristics of the eye, eye motion, image processing, and display strategies. OCT image acquisition for angiography takes more time than simple structural scans and necessitates trade-offs in flow resolution, scan quality, and speed. An important set of artifacts are projection artifacts in which images of blood vessels seem at erroneous locations. Image processing used for OCT angiography can alter vascular appearance through segmentation defects, and because of image display strategies can give false impressions of the density and location of vessels. Eye motion leads to discontinuities in displayed data. OCT angiography artifacts can be detected by interactive evaluation of the images.
The conclusion of this study was that image artifacts are common and can lead to incorrect interpretations of OCT angiography images. Because of the quantity of data available and the potential for artifacts, physician interaction in viewing the image data will be required, much like what happens in modern radiology practice.
Spaide RF, Fujimoto JG, Waheed NK. Retina. 2015 Nov;35(11):2163-80.
Optical Coherence Tomography Angiography Features Of Diabetic Retinopathy.
The purpose of this study was to describe the OCT angiography features of diabetic retinopathy. The OCT angiography of the macula of 4 patients with diabetic retinopathy were obtained and compared with fluorescein angiography for features cataloged by the Early Treatment of Diabetic Retinopathy Study.
OCT angiography detected enlargement and distortion of the foveal avascular zone, retinal capillary dropout, and pruning of arteriolar branches. Areas of capillary loss obscured by fluorescein leakage on fluorescein angiography were more clearly defined on OCT angiography. Some areas of focal leakage on fluorescein angiography that were thought to be microaneurysms were found to be small tufts of neovascularization that extended above the inner limiting membrane.
OCT angiography does not show leakage but can better delineate areas of capillary dropout and detect early retinal neovascularization. This new noninvasive angiography technology may be useful for routine surveillance of proliferative and ischemic changes in diabetic retinopathy.
Hwang TS, Jia Y, Gao SS, et al. Retina. 2015 Nov;35(11):2371-6.
Retinal and Choroidal Vasculature in Birdshot Chorioretinopathy Analyzed Using Spectral Domain Optical Coherence Tomography Angiography.
The purpose of this study was to describe retinal and choroidal vascular changes in eyes with birdshot chorioretinopathy using OCT angiography.
Four of eight eyes (50%) had birdshot lesions in the posterior pole as demonstrated on fundus photography. All of these eyes demonstrated the areas of decreased choroidal blood flow below the disrupted RPE. Larger choroidal vessels bordered the birdshot lesions. All eyes analyzed showed retinal thinning, telangiectatic vessels, and an increased intercapillary space. Capillary dilatations and loops were each seen in 7 of 8 eyes (88%).
OCT angiography provides precise microvascular detail of the retinal vasculature and choriocapillaris that allows for the noninvasive visualization of the birdshot lesions and changes in the inner retina. The OCT angiography images delineated widespread retinal vascular findings not previously described in the literature. In the future, OCT angiography could be a useful tool to evaluate the natural history of birdshot chorioretinopathy, its progression, and the effect of treatment in these patients.
de Carlo TE, Bonini Filho MA, et al. JS. Retina. 2015 Nov;35(11):2392-9
North Carolina Macular Dystrophy Is Caused by Dysregulation of the Retinal Transcription FactorPRDM13
The Purpose of this paper was to identify the specific mutations causing North Carolina macular dystrophy (NCMD) using whole genome sequencing coupled with reverse transcription polymerase chain reaction (RT-PCR) analysis of gene expression in human retinal cells.
The analysis included 141 members of 12 families with NCMD and 261 control individuals.
The genotyping supported the theory that NCMD is a developmental abnormality and found PRDM13 as the responsible gene. PRDM13 is a member of a large family of DNA-binding proteins that play key roles in controlling gene expression during development.
They were able to identify 5 rare mutations each capable of arresting human macular development. This has implications for better understanding of macular development and therefore potential reversal of or prevention of macular degenerations.
Small, KW, DeLuca, AP., Whitmore, SS, et al. Ophthalmology. Oct 2015
Intravitreal Aflibercept for Macular Edema Following Branch Retinal Vein Occlusion: 52-Week Results of the VIBRANT Study
The purpose of this study was to determine the week 52 efficacy and safety in eyes with macular edema after branch retinal vein occlusion (BRVO) treated with 2 mg intravitreal aflibercept injection (IAI) compared to grid laser.
The eyes in the IAI group received IAI every four weeks through week 24 and every 8 weeks through week 48 with rescue laser at week 36 if needed. The eyes in the grid laser group received grid laser at baseline and if rescue criterion were met, 1 additional laser from week 12 to 20 and IAI every 8 weeks after 3 monthly doses from week 24 onward (laser/ IAI group).
At week 24, 52.7% of the IAI group improved by ≥15 letters from baseline visual acuity versus the 26.7% of the IAI/laser group. At week 52, however, it was 57.1% versus 41.1%. The mean improvement of visual acuity was 17 letters in the IAI group versus s 6.9 in the IAI/laser group at week 24 and 17.1 versus 12.2 letters at week 52.
After 6 monthly IAI injections, injections every 8 weeks maintained control of macular edema and showed visual benefit through week 52. In the laser group, recue lAI given from week 24 onward resulted in substantial visual improvements by week 52.
Clark, WC, Boyer, DS, Heier, JS, et al. Ophthalmology. Oct 2015
Joint Associations of Diet, Lifestyle, and Genes with Age-Related Macular Degeneration
The purpose of this study was to determine the association of AMD with risk genotypes and with lifestyle health.
The study included women from age 50 to 79. A healthy lifestyle score, which included diet and exercise, was assigned to each and genetic risk was based on Y402H in complement factor H (CFH) and A69S in age-related maculopathy susceptibility locus 2 (ARMS2).
The study found that women with low healthy lifestyle scores and high-risk CFH genotype were 3.3 times more likely to get AMD than women with a high healthy life style score and no high-risk CFH genotype. It also concluded that an unhealthy lifestyle increases AMD risk independent of high-risk genotypes.
Meyers, KJ, Liu Z, Millen, AE et al. Ophthalmology. Nov 2015
Correlation of choroidal thickness and body mass index
The purpose of this study was to investigate the relationship between choroidal thickness (CT) and body mass index (BMI) in healthy individuals.
160 eyes of 160 healthy individuals were enrolled. The participants were divided into four groups according to their BMIs. The groups were as follows: BMIs < 18.50 formed Group I, 18.50 - 24.99 formed Group 2, 25 - 29.99 formed Group 3, and 30 - 34.99 formed Group 4. No individuals falling under the World Health Organization International Classification of morbidly obese (BMI 35+) were included in this study.
The CT of subfoveal and pre-determined parafoveal points was measured using enhanced-depth imaging OCT (EDI-OCT). Comparisons were made between the four groups’ findings. There was found to be a statistically significant difference in regard to CT in all locations with those in Groups 3 and 4 having statistically significant decreased CT measurements.
Previous studies show that higher BMIs have association with narrowed retinal artery and wider retinal vein calibers. Obesity can play a role in microcirculation based on altered levels of nitric oxide (a vasodilator), endothelin, and angiotensin-II in the body. It is thought that this may also be the mechanism behind differences in CT in healthy individuals of varying weights.
Yilmaz, I. Ozkaya A, Kocamaz M et al. Retina. October 2015. 35:2085–2090.
Inner nuclear layer thickness as a prognostic factor for metamorphopsia after epiretinal membrane surgery
The purpose of this study was to use SDOCT in evaluating prognostic factors for metamorphopsia in individuals undergoing vitrectomy for idiopathic epiretinal membrane.
53 eyes of 53 patients undergoing vitrectomy were enrolled in the prospective study. Prior to surgery, examinations including visual acuity, severity of metamorphopsia (by M-CHARTS), and OCT macula were completed. OCT was done before surgery and 3 months and 6 months after the procedure.
The OCT studies analyzed foveal and parafoveal retinal thickness, macular volume, and the thicknesses of the ganglion cell layer, inner nuclear layer, and outer retinal layer (outer nuclear layer + outer plexiform layer).
The preoperative metamorphopsia score was found to correlate significantly with central foveal thickness, inner nuclear layer thickness, and outer nuclear layer thickness. The preoperative inner nuclear layer thickness yielded the highest regression coefficient with postoperative M-score. The postoperative M-score significantly correlated with postoperative central foveal thickness and inner nuclear layer thickness.
Consequently, inner nuclear layer and central foveal thickness values are likely to be good indicators of metamorphopsia both before and after surgery and a predictor of postoperative metamorphopsia in patients with idiopathic epiretinal membranes.
Okamoto, F., Sugiura, Y., Okamoto, Y., et al. Retina. October 2015. 35:2107–2114.
Comparison of intravitreal bevacizumab and dexamethasone implant for the treatment of macula oedema associated with branch retinal vein occlusion
The goal was to compare the functional and anatomical outcomes of intravitreal bevacizumab (IVB) with those of dexamethasone implant injection (IVD) for macular oedema associated with branch retinal vein occlusion (BRVO).
Seventy-two patients with center-involving macular oedema secondary to BRVO were retrospectively enrolled in the study; these patients were treated with either 1.25 mg IVB (mean injections: 2.92±1.38) pro re nata (PRN) by follow-up monthly or 700 µg IVD (mean injections: 1.71±0.47) given at 6-month intervals PRN and were followed for at least 12 months. Main outcome measures were changes in BCVA and central foveal thickness (CFT).
There was no statistically significant difference of mean change of the BCVA between IVB and IVD groups at monthly visits, up to 12 months (all p>0.05. Additionally, 52.6% in the IVB group and 50% in the IVD group gained two or more lines of Snellen VA at 12 months (p=0.85). The mean CFT decreased by 160 µm for the IVB group and by 140.7 µm for the IVD group at 12 months. Both the IVB group and the IVD group achieved statistically similar improvement of CFT at monthly visits, up to 5 months (all p>0.05); however, the CFT began to deteriorate after 5 months in the IVD group. After a second IVD injection at 6 months, the IVD group showed significant improvement of CFT, and there was no significant difference of CFT change between the IVB and IVD groups until 12 months.
For macular edema secondary to BRVO, IV bevacizumab administered PRN monthly and IV dexamethasone administered PRN at 6-month intervals, yielded functionally and anatomically comparable outcomes at 12 months.
Kim M, Lee DH, Byeon SH, et al. Br J Ophthalmol. September 2015;99:1271-1276.
Phase-Resolved Doppler Optical Coherence Tomographic Features in Retinal Angiomatous Proliferation
The purpose of this article was to study patients diagnosed with retinal angiomatous proliferation (RAP) based on conventional imaging techniques with phase-resolved Doppler OCT) to detect and localize blood blow in RAP lesions; and to compare these findings to conventional imaging, which are mostly invasive and give limited information concerning intra and transretinal blood flow.
12 treatment-naive patients diagnosed with RAP based on fundus examination, fluorescein angiography, and ICG were included. Patients were imaged with a 1040nm swept-source phase-resolved Doppler OCT instrument. Abnormal flow was defined as intraretinal neovascularization or retinal choroidal anastomosis.
In 11 patients adequate phase-resolved Doppler OCT images were obtained showing abnormal blood flow in the RAP lesion. In 4 patients a retinal angiomatosis proliferation was found, 3 patient showed intraretinal neovascularization connected with a pigment epithelial detachment, 2 patients showed only intraretinal neovascularization, and in 2 patients flow was limited to sub retinal or sub-retinal pigment epithelial space.
The study concluded that phase-resolved Doppler OCT is able to detect and localize abnormal blood flow within RAP lesions. Blood flow was mostly confined to the intraretinal structures with or without a connecting PED; 1/3 of patients was found to have retinal choroidal anastomosis. The potential of angiography with phase-resolved Doppler OCT to distinguish between normal and pathologic blood flow in addition to structural OCT data without invasive procedures will help to further elucidate both retinal and choroidal vascular pathologies like RAP.
Amarakoon S, de Jong JH, Braff B, et al. American Journal of Ophthalmology. November 2015. 160(5):1044–1054.

|
ANSWER
TO "YOU MAKE THE DIAGNOSIS"
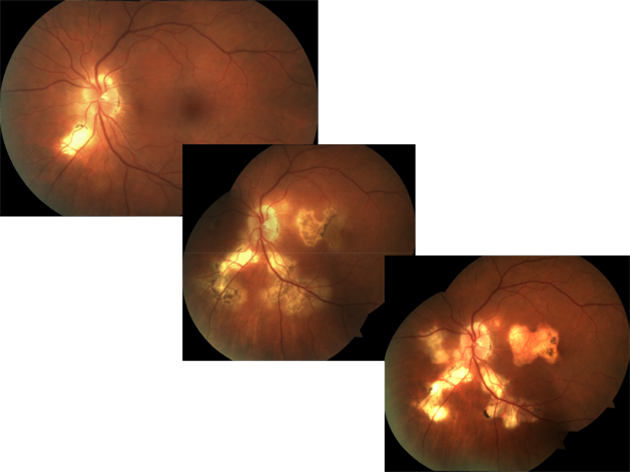
This photo represents a patient with progressiveSerpiginous choroiditis. Serpiginous choroiditis is a rare, typically bilateral, progressive, recurrent inflammation of the choroid, retinal pigment epithelium (RPE) and choriocapillaris. Its etiology is poorly understood.
The choroidal lesions appear grayish-yellow and can be localized to levels of the inner choroid and overlying retinal pigment epithelium with a distinctive retinal distribution. The disease presents concentrated around the optic disc with peripapillary lesions and fingerlike projections extending outward. Once present, active lesions last from weeks to months often with a seemingly spontaneous resolution manifesting distinct areas of retinal pigment epithelial loss (atrophy) or clumping along with concomitant retinochoroidal atrophy. Patients frequently have recurrences at intervals varying from weeks to years. Each recurrence results in an extension further into the periphery. About one third of patients present with an inflammatory reaction in both the posterior and the anterior segment.
Regardless of the presenting form, the clinical course includes progressive choroidal inflammation with the potential for multiple recurrences and significant visual loss. The amount and quality of the visual disability corresponds to the extent of the involvement of the para-foveal and foveal regions. Here, tissue destruction may be the direct result of the inflammatory lesions or may come secondary to induced choroidal neovascularization (CNV). In one study CNV, occurred in approximately 50% of cases.
Other clinical entities which should be considered in the differential are categorized in the “white dot syndromes”. These include multifocal choroiditis, multiple evanescent white dot syndrome, birdshot chorioretinopathy, acute multifocal posterior placoid pigment epitheliopathy, punctuate inner choroidopathy and diffuse unilateral subacute neuroretinitis.
Due to disease’s insidious and progressive clinical course, reassessment is required over a lifetime . Generally, treatment is only initiated if the macula is affected, and sight is threatened. Long-term immunosuppressive treatment appears to prolong remission and preserve vision in patients with serpiginous choroiditis. Intravitreal steroid preparations such as triamcinolone acetonide and fluocinolone acetonide offer promise for local control of the disease without the projected systemic side effects. Recently, researchers have reported that anti-vascular endothelial growth factor (anti-VEGF) treatments with both CNV or macular edema in serpiginous choroiditis have demonstrated positive results.
Andrew Gurwood, OD, FAAO
ORS Founding Fellow |

IN THE
NEWS
 |
Applied Genetic Technologies files investigational new drug application for gene therapy product
Applied Genetic Technologies Corp. has filed an investigational new drug application with the FDA to conduct a phase 1/2 clinical trial of its gene therapy product candidate for treating achromatopsia, according to a company press release.
The adeno-associated virus-based gene therapy is under consideration as treatment for achromatopsia caused by mutations in the CNGB3 gene. A multicenter clinical study in the U.S. is planned to evaluate safety and efficacy of the gene therapy product, according to the press release
“In addition to finalizing our collaboration agreement with Biogen, initiating our phase 1/2 clinical trial for XLRS, and expanding our product pipeline, we remain on track to have a second product candidate in clinic shortly, pending the FDA’s acceptance of the IND application and approval from the sites’ institutional review boards,” AGTC president and CEO Sue Washer said in the release.
In July, Biogen and AGTC entered into a collaboration and licensing agreement to develop gene-based therapies for ophthalmic diseases.
|
 |
Ophthotech completes patient recruitment in phase 3 trial of Fovista in combination with Lucentis
Ophthotech has completed patient recruitment in the second phase 3 trial of Fovista in combination with Lucentis for the treatment of wet age-related macular degeneration, the company announced in a press release.
The two phase 3 clinical trials are investigating the superiority of Fovista (pegpleranib, Ophthotech) in combination with Lucentis (ranibizumab, Genentech) over Lucentis monotherapy. Data from the two clinical trials will be analyzed together “to optimize pooled analysis of certain relevant endpoints in accordance with the statistical analysis plan,” the release said.
The company expects to announce topline results from both phase 3 clinical trials in the fourth quarter of 2016.
|
 |
Compounded Avastin not linked to increased risk of endophthalmitis
Intravitreal injections of repackaged Avastin did not pose a greater risk of endophthalmitis than Lucentis, according to a study in JAMA Ophthalmology.
In response to reports of an endophthalmitis outbreak linked to repackaged bevacizumab in 2011, the FDA issued a preliminary draft guidance allowing the use of compounded bevacizumab with a beyond-use date that limits use to within 5 days of repackaging.
The authors retrospectively analyzed medical claims data on 383,810 intravitreal injections of Avastin (bevacizumab, Genentech) and Lucentis (ranibizumab, Genentech) given to 58,612 patients between 2005 and 2012.Specifically, 296,565 injections of bevacizumab were given to 51,116 patients and 87,245 injections of ranibizumab were given to 7,496 patients.Primary diagnoses were age-related macular degeneration, diabetes and retinal vein occlusion.Mean time to diagnosis of endophthalmitis was 4.75 days after injection.The authors wrote that, to the best of their knowledge, the study used the single largest data source to evaluate the risk of endophthalmitis after bevacizumab or ranibizumab injection.
There were 71 cases of endophthalmitis: 49 in the bevacizumab group and 22 in the ranibizumab group. The overall rate of endophthalmitis was 0.018%. Separately, the rates were 0.017% in the bevacizumab group and 0.025% in the ranibizumab group. The between-group difference was not statistically significant.
An analysis with a broader definition of endophthalmitis showed 97 cases of endophthalmitis: 69 in the bevacizumab group and 28 in the ranibizumab group. However, that analysis did not show a stronger correlation between bevacizumab and endophthalmitis, the authors wrote.
Overall, there was no correlation between intravitreal bevacizumab and the incidence of endophthalmitis, according to the study.
|
 |
Eyegate receives patent approvals for delivery system, pharmaceutical formulation
The U.S. Patent and Trademark Office issued a Notice of Allowance for a patent application related to Eyegate Pharmaceuticals’ Eyegate II Delivery System with pre-filled drug applicator, the company announced in a press release.
The patent is for a delivery device containing a hydrogel applicator used to transport therapeutic substance across and/or through the eye, the release said.
Additionally, a patent related to delivery of the company’s lead compound EGP-437 (dexamethasone phosphate) has been granted.
“The patent is not limited to any particular eye condition or to a particular iontophoretic device/method, but rather is directed at the delivery of this formulation through iontophoresis to the eye of patients in need of treatment,” the release said.
“The grant and issuance of these two patents represent important milestones in our effort to build a robust IP portfolio protecting our proprietary drug formulation and the revolutionary delivery system that accompanies it,” Stephen From, president and CEO of Eyegate, said in the release.
EGP-437 is being evaluated in clinical trials to treat non-infectious anterior uveitis and macular edema, according to the release.
|
 |
Spark’s SPK-RPE65 phase 3 trial show gains in functional vision, light sensitivity
SPK-RPE65 resulted in a sustained difference in functional vision and light sensitivity between study subjects and controls, according to additional data from a phase 3 pivotal trial, Spark Therapeutics announced in a press release.
SPK-RPE65 is the company’s lead product candidate for the treatment of RPE65-mediated inherited retinal dystrophies.
The additional data, presented at the Retina Society meeting in Paris, build on the top-line results of the phase 3 trial announced earlier this month. Among 20 intervention subjects, mean improvement in functional vision was 1.9 specified lux levels as measured by the change in bilateral mobility testing (MT) from baseline to 1 year in a modified intent-to-treat population.
Mean improvement among nine control subjects was 0.2 specified lux levels.
Thirteen subjects who received SPK-RPE65 were able to pass the MT at 1 lux at 1 year, the maximum improvement measurable on the MT score. None of the control subjects were able to pass MT at 1 lux at 1 year, the release said.
In addition, in full-field light sensitivity threshold testing, intervention subjects showed a statistically significant improvement of –2.06 log10 (candela second/m2). Control subjects showed a decline of 0.04 log10 (candela second/m2).
Spark Therapeutics also released durability data from a phase 1 study of SPK-RPE65. The data showed that after 3 years, all subjects who participated in the phase 1 open-label study continued to experience a sustained improvement in functional vision and light sensitivity. These subjects received the same dose and volume of SPK-RPE65 that was used in the phase 3 trial, the release said.
|
 |
Psoriasis may be highly associated with risk of developing retinal vein occlusion
Patients with psoriasis may have a higher risk of developing retinal vein occlusion, according to a study in RETINA. .
The retrospective population-based cohort study evaluated the risk and incidence of developing RVO in 30,198 psoriasis patients compared with 30,198 control subjects without psoriasis from the Taiwan National Health Insurance Research Database.
The RVO incidence was 1.46 times higher in psoriasis patients vs. non-psoriasis subjects, 1.97 times higher in psoriasis patients aged 65 years or older vs. subjects without psoriasis, and 1.82 times higher in women with psoriasis than without psoriasis.
In a subgroup analysis, the risk of developing RVO in psoriasis patients was 2.07 times higher in those with a hypertension comorbidity compared with those without psoriasis.
There was also a significantly higher risk of RVO in psoriasis patients who needed methotrexate as a second-line therapy.
|
 |
Study: Higher carotenoid intake reduced risk of advanced AMD over long term
An increased intake of lutein/zeaxanthin reduced the long-term risk of advanced age-related macular degeneration, according to a large study recently published in JAMA ophthalmology.
The prospective cohort study included 63,443 women and 38,603 men who participated in the Nurses’ Health Study and Health Professionals Follow-up Study between 1984 and 2010. All subjects were older than 50 years and were not diagnosed with AMD, diabetes, cardiovascular disease or cancer at baseline.
Food frequency questionnaires were used to calculate predicted plasma carotenoid scores. Regression models were used to account for bioavailability and reporting validity of different foods and to determine associations between predicted plasma carotenoid scores and AMD.
Over the course of follow-up of the studies, there were 1,361 cases of intermediate AMD and 1,118 cases of advanced AMD, primarily neovascular AMD, with visual acuity of 20/30 or worse.
Median age at onset was 73 years among women and 76 years among men.
A comparison of extreme quintiles of predicted plasma lutein/zeaxanthin scores showed a 40% reduction in the risk of advanced AMD in women and men (P < .001).
There was no association between predicted plasma scores or calculated intake of carotenoids and intermediate AMD.
|
 |
Only half of patients undergo appropriate screening for hydroxychloroquine toxicity
Slightly more than half of patients who presented for hydroxychloroquine toxicity screening received appropriate evaluation based on the 2011 revised guidelines, according to a study in the American Journal of Ophthalmology. Proper screening was defined as one subjective test, Humphrey visual field, and one objective test, spectral-domain OCT, fundus autofluorescence or multifocal electroretinography.
The study included 756 patients who took hydroxychloroquine from 2011 to 2014 and underwent 1,294 screening visits.
Initial screenings were performed on 21 patients at an outside institution. Of the 735 remaining patients, 560 (76.2%) presented solely for hydroxychloroquine screening and 175 (23.8%) presented with chief complaints not related to hydroxychloroquine.
Of the 560 patients who presented for screening only, 307 (54.8%) were appropriately screened, 144 (25.7%) were underscreened and 109 (19.5%) were inappropriately screened.
The most common initial screening tests were SD-OCT in 56.6% of patients, Humphrey visual field in 55% and Amsler grid testing in 40%.
The most common follow-up screening tests were SD-OCT in 63.5% of patients, Humphrey visual field in 42.4% and Amsler grid testing in 31.3%.
Fundus autofluorescence was performed in 97 patients (13.2%) and multifocal electroretinography was performed in three patients (0.4%) at initial screening.
Treating physicians found signs of toxicity in 12 patients (1.6%).
|
 |
RESPOND study: Iluvien effective at 6 months in DME patients
Iluvien was effective and well tolerated in reducing retinal thickness and improving visual acuity in patients with chronic diabetic macular edema, Alimera Sciences announced in a press release. Alimera Sciences Limited, the company’s European subsidiary, announced the interim 6-month results of the RESPOND study at the Euretina congress in Nice, France.
The nonrandomized, open-label, multicenter phase 4 pilot study is focusing on the efficacy and safety of Iluvien (fluocinolone acetonide intravitreal implant 0.19 mg) in patients with chronic DME that is insufficiently responsive to available therapies. The study is being conducted at four sites in Portugal.
Twelve patients received a single injection of Iluvien. Eleven patients completed 6 months of follow-up, and nine of those patients had maintained or improved best corrected visual acuity compared with baseline. Central subfield thickness decreased in 10 patients. BCVA improved an average of 7.1 letters in the 11 patients, with an average decrease in central subfield thickness of 300.6 µm, according to the release.
Patients will be followed out to 12 months, and IOP increases and cataract progression will also be monitored. |
 |
Novel visible-light OCT may improve retinal disease diagnosis, assessment
A collaborative team from Bascom Palmer Eye Institute and Florida International University has developed a novel visible-light OCT technology for imaging rhodopsin, according to a Bascom Palmer press release.
A study, supported by the NIH, reported that VIS-OCT technology accurately measured rhodopsin distribution and functionally imaged rod photoreceptors in the retinas of rats.
In using VIS-OCT to image rhodopsin, the light-sensing molecule that converts light signals to neuronal signals sent to the brain, clinicians would be able to diagnose and assess disease progression in retinal degenerative disorders, including hereditary retinal degeneration, while also monitoring progression of dry age-related macular degeneration.
The group is working to make the imaging equipment more “patient-friendly” and to move it into the clinical setting, according to the press release. |

MEET
THE FELLOWS
In each issue, a Fellow of the Optometric Retinal Society is highlighted. In this issue, Dr. Ava Bittner, a new ORS fellow, will be highlighted
Dr. Ava Bittner received her Optometry degree from the Pennsylvania College of Optometry in 2001 and earned a PhD in clinical investigation from the Johns Hopkins School of Public Health in 2011. Dr. Bittner completed a clinical research post-doctoral fellowship at the Johns Hopkins Wilmer Eye Institute’s Lions Low Vision Research and Rehabilitation Center from 2002-2007, and then joined the faculty as an Assistant Professor at the Wilmer Eye Institute. Dr. Bittner was a proud recipient of the AAO Ezell Fellowship Award in 2010-2011, and was inducted into the Phi Beta Kappa academic Honorary Society in 2011 for excellence in her thesis work. In 2014, she joined the faculty at the Nova Southeastern University, College of Optometry and currently holds the rank of Associate Professor. Dr. Bittner's research interests include the psychophysical assessment of the healthy and diseased visual system, both to increase our understanding of vision loss and to validate tools to monitor retinal disease and therapeutic outcomes. Dr. Bittner has received NIH funding (K23 and R21 awards) from the National Eye Institute, and is currently the principal investigator of a randomized controlled trial evaluating electro-acupuncture and transcorneal electrical stimulation as potential interventions to improve ocular/retinal blood flow and visual function in retinitis pigmentosa (RP). Dr. Bittner's previous research has examined the relationships between psychosocial factors (e.g. mood states) and day to day variations in vision or photopsias in RP patients. She has served as a co-investigator or consultant on multi-center clinical trials for patients with retinal degenerations; i.e., Visioncare’s Implantable Miniature Telescope for AMD, Optobionics’ Artificial Silicon Retina® for RP, Second Sight’s Argus II retinal prosthesis for RP, and QLT Inc.'s oral synthetic retinoid for patients with inherited retinal disease due to RPE65 or LRAT mutations. Dr. Bittner (formerly Dr. Kiser) currently has 22 peer-reviewed publications in scientific journals.
This past June, Dr. Bittner and her husband had their first child, a healthy baby girl, Annora Corinne. While still in the womb, her daughter had joined Dr. Bittner while she continued her 10 year practice of Bikram (hot) yoga, as well as her passion for travel planning (with a trip to Nepal during her first trimester) and skiing at her favorite destination, Snowmass, Colorado. It seems that the regular yoga practice during pregnancy has paid off, as her daughter's daycare often comments that she is a really calm, peaceful, attentive and happy baby! Nowadays with the demands of a thriving research career and new baby, there is less time than usual for yoga, while the many positive aspects in her life keep her motivated.
|
Why do most retinitis pigmentosa patients lose their blue color vision first?
Ava K. Bittner, OD, PhD, FAAO
Nova Southeastern University; College of Optometry
Fort Lauderdale, Florida
Background: Over 20 years ago, previous studies reported that some retinitis pigmentosa (RP) patients initially lose short wavelength cones (i.e., s-cones for blue-violet colors) prior to longer wavelength cone function. [1,2] At that time, it was hypothesized that a mechanism might affect a shared biochemical pathway vulnerability found in both rods and s-cones selectively, but not other cones. However, no further research on the relationship between loss of s-cone function and rod-mediated night vision has been published. It is known that some RP patients present early in life with normal rod and cone function, while others have loss of both, and some have only cone function at a young age.[3] We conducted a research study that explored whether history of vision loss or current visual function status may predict which RP patients are susceptible to s-cone loss.
Hypotheses: Why are cones lost in RP, which is due to a rod-specific mutation?
A long standing question in the field of RP research is why cones depend on rods for survival. Listed below are a few of the hypotheses that may explain secondary cone loss in a disease that primarily results in widespread rod death: [4,5,6]
• Toxic agents released with rod death: either directly from rods or from activated retinal microglia that migrate to the photoreceptor layer.
• Rods produce a neurotrophic factor that is required by cones for survival.
• Oxidative damage from rod loss may cause death of cones.
• Reduced activity in the mTOR pathway (affects nutrition, results in starvation of cones).
• Loss of structural support (due to rod loss, causes reduced contact between the cones and retinal pigment epithelium (RPE)) may result in a decrease in glucose supply and the starvation of cones.
Hypotheses: Why is there a greater loss of s-cones versus l- and m-cones in RP?
• S-cones may be more susceptible to changes occurring at the level of the RPE (retinal pigment epithelium) due to morphologic differences when compared to l- and m-cones.
S-cones (blue) have a longer / larger inner segment and deeper innervation projecting further into the sub-retinal space towards the RPE (i.e., closer to the RPE).
• S-cones are the least numerous, representing only represent 10% of all cones, and are located para-foveally (i.e., outside of central vision), thus a loss of a few cells could have a greater impact on visual function.
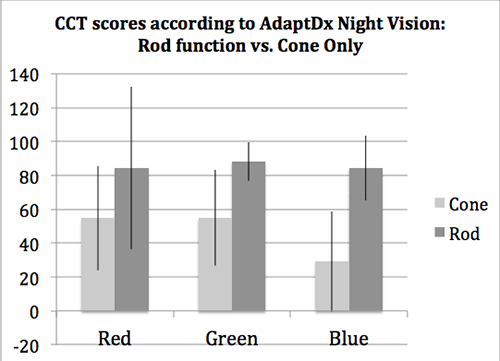
Methods: At two visits within a month, 22 RP subjects with visual acuity (VA) better than 20/400 completed the PC-based Innova Rabin Cone Contrast Test (CCT), ETDRS VA, Humphrey and Goldmann visual fields (VF), and AdaptDx dark-adaptation in each eye.
Results: Of 22 RP patients tested, 15 (68%) had measurable CCT results and they had VA better than 0.5 logMAR. All of the subjects who did not have any measurable rod function with the AdaptDx (n=10) had the greatest CCT loss for s-cone sensitivity (mean score 23 across subjects) compared to l- and m-cones (mean scores 55 and 51, respectively), while those with rod function (n=5) had either normal s-cone function or relatively equal reductions for all three cones (mean scores 87, 88 and 84 for l-, m- and s-cones, respectively). Duration of VF loss (p=0.37) and night vision loss (p=0.14) were not statistically significantly related to s-cone loss across subjects. Statistically significant predictors of reduced CCT sensitivity for each of the three cone types were reduced VA, decreased Goldmann V4e VF log retinal area, and inability to see stars as a child (all p<0.05), as well as no current rod function (p≤0.001).
Discussion and Conclusions: In the absence of functional rods, there was a greater loss of s-cone sensitivity than l- and m-cones in RP patients. Some of these patients never had rod function (i.e., never seen stars), while others lost rod vision with disease progression. RP patients with normally functioning rods had normal CCT scores for all 3 cone types; then as rod function was slightly decreased (i.e., delayed dark-adaption rate), we noted reductions in sensitivity for all 3 cone types. These various patterns of rod and cone function loss in RP patients represent different stages of disease progression and/or different subtypes of RP,[3] which is genetically and phenotypically heterogeneous. The greater loss of sensitivity for s-cones than l- and m-cones in the absence of functional rods may be likely explained by a combination of multiple mechanisms studied in animal models of RP (e.g., structural, toxic, nutritional, trophic, and/or oxidative factors). While it is not currently possible to measure these factors in the retina of human subjects, we hope that future clinical trials will explore potential therapeutic approaches related to these mechanisms, and measure the responsiveness of specific cone types to therapy, in order to help yield further valuable insight into the mechanisms involved in s-cone loss, thus furthering our knowledge of RP pathophysiology.
 |
References:
[1] Swanson, W., Birch, D., Anderson, J. S-Cone Function in Patients With Retinitis Pigmentosa. Invest Ophthalmol Vis Sci 1993; 33(11) 3045-3055.
[2] Greenstein, V., Hood, D., Ritch, R., Steinberger, D., & Carr, R. S (blue) cone pathway vulnerability in retinitis pigmentosa, diabetes and glaucoma. Invest Ophthalmol Vis Sci 1989; 30(8): 1732-1737.
[3] Jacobson, S.G., Roman, A.J., Aleman, T.S., Sumaroka, A., Herrera, W., Windsor, E.A.M. , Atkinson, L.A., Schwartz, S.B., Steinberg, J.D., Cideciyan, A.V. "Normal Central Retinal Function and Structure Preserved in Retinitis Pigmentosa." Invest Ophthalmol Vis Sci 2010; 51(2): 1079-1085.
[4] Bovolenta, P., Cisneros E. "Retinitis Pigmentosa: Cone Photoreceptors Starving to Death." Nature Neuroscience 2009; 12(1): 5-6.
[5] Koenekoop, R.K. "Why Do Cone Photoreceptors Die in Rod-Specific Forms of Retinal Degenerations?" Ophthalmic Genetics 2009; 30(3): 152-154.
[6] Ramachandran, P., Song, J., & Bennett, J. Exploiting metabolic and antioxidant pathways to maintain vision in blinding disease. The Journal of Clinical Investigation 2015; 125(4): 1390-1392.
Acknowledgements: NIH funding to AKB: R21 award EY023720
|
WHY
BECOME AN ORS FELLOW?
By Bill Denton, O.D., F.A.A.O.
Chair, Membership Committee
At some point in your career, you realize you just may be coasting. Your knowledge has been limited to the journals you receive and attempt to read, and the conferences that may not be as fulfilling as they once were. You simply need a challenge that will add an extra dimension to your professional learning.
Fellowship in the Optometric Retina Society (ORS) can provide several benefits in addition to the initial challenge of qualifying for this honor. Plenty of perks accompany your induction, but the coolest part is being associated with a body of knowledge and resources which can help you in many other ways. It is not uncommon to receive weekly thought-provoking emails about challenging cases and treatment dilemmas. Some fellows like to share their awesome cases they have diagnosed, while others post their cases with hopes that other Fellows will suggest an alternative differential diagnosis. At times it is like a round-table of brainstorming, but through the use of modern technology. Fellowship has little obligation with a huge opportunity for professional growth.
If you are up to the challenge of becoming a Fellow of the ORS, feel free to peruse the details and application at www.optometricretinasociety.org. Advice can be given to assist you in your quest. Feel free to contact us. |

SPONSOR NEWS

Editor
in Chief
Steven Ferrucci, OD, FAAO
Co-Editor
Mark T. Dunbar, OD, FAAO |
Journal
Reviewers
Tahrim Rahman, OD
Kristie Draskovic, OD
Savannah Brunt, OD
Nicholas Zagorianos, OD
Senior Graphic Designer
Matt Egger
|
Review of Optometry® is published by the Review Group, a Division of Jobson Medical Information LLC (JMI), 11 Campus Boulevard, Newtown Square, PA 19073.
To subscribe to other JMI newsletters or to manage your subscription, click here.
To change your email address, reply to this email. Write "change of address" in the subject line. Make sure to provide us with your old and new address.
To ensure delivery, please be sure to add revoptom@lists.jobsonmail.com to your address book or safe senders list.
Click here if you do not want to receive future emails from Review of Optometry. |
|