| |
Volume 16, Number 3 |
September 2020 |
|
FROM
THE DESK OF THE EDITOR
Nutritional supplementation in macular degeneration continues to be a hotly debated topic. In 2013 Dr. Carl Awh and colleagues were the first to suggest that high-dose zinc may be harmful to some patients with AMD, specifically those with high complement factor H (CFH) risk alleles. The original AREDS investigators have strongly disagreed with this, and the debate has ping-ponged back and forth for years. The studies, supporting either side, have all just taken turns analyzing a similar dataset of patients from the original AREDS.
A new paper on this hot topic was published last month in the Journal of Vitreoretinal Diseases. The authors of the GAIN study are neither affiliated with Dr. Awh nor the AREDS group. They took a much different approach by looking at patients presenting with newly diagnosed CNVM from AMD. Then, an independent agency gathered information about their history of AREDS vitamin use or non-use, as well as obtained genetic testing. If you have any interest in the zinc debate, I highly suggest this as a must-read. You can also read a summary here in the abstract section of the newsletter. The GAIN study supports Dr. Awh’s original argument that high-dose zinc is harmful (increased risk of progression to CNVM) to those with high CFH and beneficial for those with high ARMS2.
Precision medicine has become more common. Just look at cancer treatment. We now have the ability to figure out the best treatment protocol for a cancer patient based on the genetic makeup of a tumor. Applying precision medicine in AMD is an exciting prospect. But will this debate ever be settled? Who knows? At this point, we just have to sift through the information out there and try to do the very best for our AMD patients.
Stay well!
Anna Bedwell, OD, FAAO
Editor-in-Chief
PRESIDENT'S MESSAGE
Nine months ago, as we started the year 2020, most of us expected a wonderful 12 months. What eye doctor wouldn't love 2020? A few months later our world was turned upside down with a global viral pandemic. Hopefully we have seen the worst of it and life will slowly get back to normal. It will truly be an unprecedented medical achievement if a vaccine is available by the end of the year. Even if it's early 2021 that we have a vaccine, that is half of the time that it normally takes to produce a viable vaccine. As I was feeling a bit sorry for myself the other day due to the current state of affairs, I reflected on how much progress we have seen in medicine over the past 25 to 30 years. Treatment of diabetic retinopathy has evolved from watch and pray, to laser treatment, to now anti-VEGF treatment. Eyes that in the past would have been blind are being saved every day. Macular degeneration has likewise seen a massive shift in diagnosis and treatment with AREDS, AREDS2, and current treatment with anti-VEGF as well. Diagnosis has come light years from where we were just two decades ago. Who today can imagine treating retinal disease without an OCT? Did any of us really know that someone had vitreo-macular traction prior to the OCT era? We really have come a long way, baby.
This trip down memory lane gave me great comfort in the knowledge that we WILL, in time, overcome this pandemic and life WILL return to normal. We will assuredly gain even greater knowledge of how to manage these new viruses as they pop up in the future as well. I'm convinced that our knowledge of ocular disease will grow, and cures will be found for many of the conditions that we now manage. Compared to just 40 years ago, we are already far ahead of where we were in treating many eye diseases. For now, we need to continue to be constantly learning to improve patient outcomes on an individual basis. The upcoming ORS Retina Update meeting this December will help all of us to get more up-to-date on current and future diagnostic and management options for vitreo-retinal diseases. I hope that many of you will sign up and attend the first virtual Retina Update meeting and get better prepared to make 2021 a better year for your patients. I hope to see you all there...virtually, of course.
Jeffrey Austin, OD, FAAO
ORS President

YOU
MAKE THE DIAGNOSIS
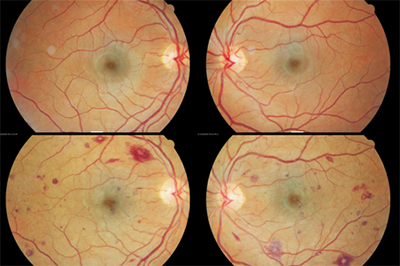
Images from the same patient with baseline (top) and three weeks later (bottom).
Answer appears later in newsletter.

Image Gallery
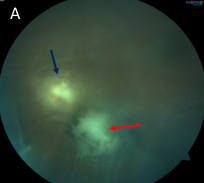
Which of the following images represents active toxoplasmosis?
Answer appears later in the newsletter.
|

JOURNAL
ABSTRACTS
Genetics and Age-Related Eye Disease Study Formulation Interaction in Neovascular Age-Related Macular Degeneration (GAIN study)
Genetic testing in AMD has been a controversial subject over the last decade. When it comes to nutrition, does one size fit all or is there a potential for harm? More specifically, does high-dose zinc increase the risk for neovascular AMD in those with high complement factor H (CFH)? All prior studies approaching the topic have used the same dataset from the original AREDS study. The GAIN study took a different approach to answer this question.
The study was designed as a case-only study that evaluated patients with a new diagnosis of neovascular AMD. From there, participants were screened for two groups: those that consistently took an AREDS formulation (with at least 40mg zinc) over the past five years vs. those that had never consumed an eye vitamin formulation. Those with an unreliable history or intermittent use were excluded. Genetic testing was performed to separate into genotype groups (GTG). Of particular interest were GTGs 2 (high CFH, low ARMS2; n=47) and 3 (low CHF, high ARMS2; n=119).
If genotype and AREDS formulation have no relationship, then the expectation would be consistency of AREDS formulation use among the GTGs. That was not the case. The authors found a higher proportion of AREDS formulation use to non-use in GTG 2, alluding to a negative interaction between CHF and high-dose zinc. In GTG 3, there was a lower proportion of AREDS formulation use to non-use indicating zinc was protective in decreasing progression to neovascular AMD. The data suggested 4.81-fold greater odds for AREDS formulation use for those in GTG 2 compared to GTG 3. Their recommendation sides with Awh and colleagues. The GAIN researchers supported routine genetic testing on patients with intermediate AMD to identify those 15% falling into GTG 2, for whom zinc is detrimental.
Kaufman SR, Yoganathan P, Small KW, et al. Genetics and age-related eye disease study formulation interaction in neovascular age-related macular degeneration. Journal of VitreoRetinal Diseases. 2020; Aug. 19 [Epub ahead of print].
Ranibizumab for Macular Edema Secondary to Central and Branch Retinal Vein Occlusion in Patients Younger Than 50 Years of Age
It is well-established that anti-VEGF is effective for macula edema from retinal vein occlusion. But what about the young adult population? Those young adults are more likely to have a different mechanism causing a vein occlusion compared to older adults.
This study focused in on the younger adults, under age 50, with a new diagnosis of macular edema from either central or branch vein occlusion. Excluded were patients pregnant or nursing, had history of stroke of coronary artery event in the prior six months, and those with other confounding ocular disease. All patients received an injection of ranibizumab at baseline and then PRN on a monthly basis for 12 months. Thirty-eight eyes (25 CRVO and 13 BRVO) were enrolled for study. The average patient age was 41.7 +/- 8.5 years with a higher proportion of males at 71%. None of the CRVO cases were ischemic but four of the BRVO had ischemic signs.
At completion, the CRVO patients gained 15.1 ± 8 ETDRS letters with 3.6 ± 2.4 injections while the BRVO group gained 7.0 ±7.1 letters with 4.4 ± 2.4 injections. A total of 17 eyes achieved a final VA of 0 LogMAR (20/20 Snellen). Interestingly, none of these young adult patients had coagulation or thrombophilic disorders, but, instead, most had underlying vascular risks factors (HTN, DM, hyperlipidemia) with 60.5% hypertensive.
The literature is sparse with regard to the younger subgroup of vein occlusion patients though natural history studies suggest a milder clinical course compared to older patients. The authors concluded that intravitreal ranibizumab dosed PRN is an effective treatment option in patients younger than 50 suffering from macular edema secondary to vein occlusion.
Battaglia Parodi M, Romano F, Arrigo A, et al. Ranibizumab for macular edema secondary to central and branch retinal vein occlusion in patients younger than 50 years of age. Biomed Res Int. 2020; Jun 17. [Epub ahead of print].
Central Retinal Vein Occlusion after Gastric Bypass Surgery
This report highlights a case of asymptomatic CRVO that developed within one month of gastric bypass surgery. A 60-year-old female underwent gastric bypass surgery. Immediately prior to surgery, she underwent ophthalmic examination, which was normal. After surgery, she developed gastrointestinal bleeding, which required blood transfusion. One month after surgery, she again underwent ophthalmic examination. Her right eye was normal, and best-corrected visual acuity was 20/20 in each eye. Her left fundus revealed marked disc swelling, four quadrant retinal hemorrhaging, and tortuous veins. There was no capillary dropout on IVFA and no macular edema. In the course of follow-up over one year, the patient’s vision remained 20/20 at all times, and the fundus findings in the left eye resolved. The authors postulate that the postoperative bleeding associated with the gastric bypass surgery may have been a trigger for the asymptomatic CRVO in the left eye. Interestingly, the clinical findings in this case fit the profile for an entity known as papillophlebitis very well, though it is not mentioned in the report. Some practitioners feel that papillophlebitis is a distinct entity representing an inflammatory variant of CRVO, while others feel that it is not a separate entity and merely represents very mild CRVO. It is characterized by disc swelling, tortuous and dilated veins, and mild-to-moderate four quadrant hemorrhaging. There is typically little to no macular edema, and vision most often remains normal or is only mildly reduced.
Laiginhas R, Esteves-Leandro J, Cardoso P, et al. Central retinal vein occlusion after gastric bypass surgery. Obes Surg. 2020; May 27. [Epub ahead of print].
Papillophlebitis in a COVID-19 Patient: Inflammation and Hypercoaguable State
This report details a case of presumed papillophlebitis in a patient recovering from COVID-19 infection. A 40-year-old Caucasian male presented with a complaint of recent onset of visual disturbance OD. Best-corrected vision was 20/20 in each eye. The right eye was normal, but the left fundus exhibited significant disc swelling, cotton-wool spots, dilated and tortuous veins, and moderate four quadrant hemorrhaging. There was no evidence of ischemia on IVFA, and no macular edema. An extensive medical evaluation failed to reveal any significant underlying conditions, other than a hypercoaguable state believed to be the result of recent COVID-19 infection-related inflammation. Interestingly, one week after the initial diagnosis, the patient’s visual acuity rapidly deteriorated to 20/200 in the left eye due to severe macular edema. This is somewhat atypical for papillophelebitis. An Ozurdex intravitreal dexamethasone implant was utilized, resulting in rapid improvement in the macular edema, with vision recovering to 20/40 two weeks later. The rapid response to the Ozurdex implant was consistent with an underlying inflammatory mechanism, quite possibly the result of COVID-19 infection.
Insausti-García A, Reche-Sainz JA, Ruiz-Arranz C, et al. Papillophlebitis in a COVID-19 patient: Inflammation and hypercoagulable state. Eur J Ophthalmol. 2020; Jul 30. [Epub ahead of print.]
Central Retinal Vein Occlusion in Otherwise Healthy Children and Adolescents: Association with Multigenetic Variants of Thrombophilia
Though the pathogenesis of central retinal vein occlusion (CRVO) is still unknown, several risk factors have been widely agreed upon. The risk factors include age, hypertension, and glaucoma. The bearing thrombophilia-related traits have on the risk of developing CRVO remains unclear, both in adult and pediatric populations. Patients under 40 only represent 10 to 15% of patients who develop CRVO. This case study sought to understand the part thrombophilia plays in young healthy patients that develop CRVO. Each of the four patients in this report underwent extensive bloodwork, brain and orbital magnetic resonance imaging (MRI), electrocardiography, and ultrasound of the supraaortic vessels. Each also received genetic screening for thrombophilia including: Factor XII, ITGB3 (GPIIIa) PlA1/A2 (rs5918), ITGA2 (GPIa/IIa) C807T (rs1126643) and G873A (rs1062535), prothrombin G20210A, Leiden Factor V G1691A, MTHFR C677T, MTHFR A1298C mutations, and plasminogen activator inhibitor (PAI)-1 4G/5G polymorphisms.
The ages of the patients included in this study range from six to 18 years, and each child had an unremarkable medical history. Best-corrected visual acuities at time of diagnosis ranged from 20/40 to 20/400. Fundus examinations were consistent with CRVO with swollen optic nerves, tortuous vessels, and retinal hemorrhages. In all cases blood work and imaging were normal. Most notably, in all four cases, genetic testing showed the patient was a carrier for, or had a mutation in, one of the genes mentioned above. All patients were prescribed prophylactic low-does aspirin to decrease the risk of additional thrombotic episodes. Two of the four cases had five and eight years of follow-up, and neither patient has had a thrombotic episode since diagnosis with CRVO. Ultimately, this small case study suggested that mutations and variation in genes that code for coagulation pathways are likely a risk factor in young, healthy patients who present with CRVO. In these cases, an exhaustive genetic work up should be performed. It is also important to note the potential success of low-dose aspirin in preventing other thrombotic events.
Bremond-Gignac D, Daruich A, Gallet M, et al. Central retinal vein occlusion in otherwise healthy children and adolescents: association with multigenetic variants of thrombophilia. Retina. 2020;40(7):1339-43.
Cystoid Macular Edema Related to Cataract Surgery and Topical Prostaglandin Analogs: Mechanism, Diagnosis, and Management
This literature review explored the complicated mechanisms in which prostaglandin analogs (PGA) contribute to the formation of cystoid macular edema (CME) after cataract surgery, and how to manage CME in pseudophakic eyes. CME is a retinal thickening caused by leaking of the perifoveal capillaries of the inner nuclear and outer plexiform layers. This happens as a result of the breakdown of the blood retinal barrier related to inflammatory molecules. The link between CME and PGA use after cataract surgery has been contested, but, ultimately, consensus has been reached that PGA use is in fact a risk factor for CME. The hypothesis is that PGAs accumulate in the aqueous humor, a space where there is already increased inflammation post-surgery, and potentially prolong the release of endogenous proinflammatory mediators. This process is particularly consequential in pseudophakic eyes.
Previously, CME has been detected when patients become symptomatic, and report a change or decrease in vision. Imaging technology such as FA, OCT and OCTA can be used to detect CME before symptoms develop. Early detection becomes important in high-risk eyes; for example, those with chronic inflammation, diabetic retinopathy, and previous vein occlusions, and in patients who are taking PGAs. For patients taking PGAs, management includes discontinuing PGAs, keeping intraocular pressure (IOP) low, and reducing intraocular inflammation. Nonsteroidal anti-inflammatory (NSAID) and corticosteroids are both used to manage CME. NSAIDs are preferred in patients taking PGAs because they do not impact intraocular pressure. Nepafenac has been shown to be the most effective. There is no FDA approved prophylactic treatment for postoperative CME; however, some surgeons use corticosteroids, NSAIDS, or a combination therein preoperatively to decrease the risk.
Holló G, Aung T, Cantor LB, et al. Cystoid macular edema related to cataract surgery and topical prostaglandin analogs: Mechanism, diagnosis, and management. Surv Ophthalmol. 2020;65(5):496-512.

|
ANSWER
TO "YOU MAKE THE DIAGNOSIS"
Case Report: Acute Myeloid Leukemia
The Patient: A 37-year-old female presented for her annual diabetic eye examination without visual complaint. Pertinent history included type 1 diabetes x 26 years under good control with A1Cs consistently close to 7%. Current medications included NPH and Humalog insulin, folic acid, and omega-3 supplementation. Blood pressure was consistently normal without medication. Best-corrected VA was 20/20 OU; IOPs and anterior segment were normal OU; and the retinal exam revealed occasional Mas OU consistent with mild NPDR.
The patient presented to her endocrinologist located within my practice three weeks later for her quarterly diabetes exam. Her A1C was found to be uncharacteristically high at 8.1%. She indicated that she had been sick for the last couple of weeks with a fever and an upper respiratory infection. Her symptoms included chest congestion, sore throat, sore/bleeding gums for which she had been prescribed azithromycin three days prior by her PCP. She also was experiencing a marked increase in photophobia during her illness, and I was asked by her endocrinologist to see her again for further investigation into her photophobia complaint.
Best-corrected VA was still 20/20 OU, and she was, indeed, photophobic during pupillary testing, which showed no abnormality. The anterior segment surface was normal, but there were a trace amount of cells in the anterior chamber OU with normal IOPs. The retina showed a drastically different appearance than her last exam three weeks prior with both retinae showing a moderate to severe increase in blot/NFL hemorrhages with a preponderance of Roth spots in all quadrants extending out to the mid-periphery.
Discussion: The above case describes a rapid progression of retinopathy in a patient with well-controlled type 1 diabetes. The likelihood of diabetic etiology was unlikely given the degree of progression over a one-month period, especially in a patient under good control. The preponderance of Roth spots in the setting of fast progression suggested an alternative, and more severe, underlying etiology. These findings were shared with her endocrinologist inside our practice, and his examination revealed the presence of widespread petechial hemorrhages on her arms and legs. Subsequent laboratory testing revealed results consistent with acute myeloid leukemia.
|

IN THE
NEWS
 |
TissueBlue™ to the Rescue
Vitreoretinal surgeons now have access to the recently FDA-approved Brilliant Blue G ophthalmic solution, TissueBlue™ 0.025%. Used for staining the internal limiting membrane (ILM), it has been used outside of the United States since 2010 and is marketed under the brand ILM-Blue. Being FDA-approved means the solution meets pharmaceutical standards and manufacturing consistency versus a compounded product.
The TissueBlue™ patented mixture contains 4% polyethylene glycol (PEG), which gives it a higher specific gravity. By doing so, the stain maintains a cohesive ball as it makes its way to the posterior pole and the targeted ILM tissue versus dispersing into the periphery. It is available in a 2.25mL syringe pre-filled with 0.5mL of solution.
|
 |
The Mask MADE Me Do It
Wearing masks has certainly become a part of everyone’s daily routine. In some cases, this can result in ophthalmic symptoms. Workers at the Centre for Ocular Research & Education (CORE) are informing eye care providers about mask-associated dry eye (MADE).
The mechanism is simple, they say. Obstructed outflow of air redirects upward toward the ocular surface and accelerates tear film evaporation. Contact lens wearers, using masks for extended periods and at digital screens, and those already suffering from dry eye may be further at risk.
The first thing to do is ensure a properly fitting mask, though no specific recommendations were made other than to consider taping the top edge of the mask to the skin. Other recommendations were topical lubricants, limiting time in air-conditioned environments, and taking breaks from digital devices.
|
 |
A Rare Disease Gets a New Treatment
The FDA recently approved Enspryng™ for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adult patients who are anti-aquaporin-4 (AQP4) antibody positive. The Genentech drug is administered subcutaneously by the patient after proper training.
Enspryng is a humanized monoclonal antibody that blocks interleukin-6 (IL-6), which researchers believe is the main inflammatory component for NMOSD. NMOSD is a rare, autoimmune central nervous system disease that targets the optic nerve and spinal cord, and may be misdiagnosed as multiple sclerosis. The two can be distinguished by the presences of the AQP4 antibody.
Two clinical trials supported its approval: SAkuraStar and SAkuraSky. In the former study, 76.5% of treated patients were relapse-free at 86 weeks compared to just over 40% in the placebo group. In the latter study, Enspryng was used alongside baseline immunosuppressant therapy, and over 90% of the treated group were relapse-free after 96 weeks versus 56.8% of the placebo group.
Enspryng is dosed every four weeks, and neutrophil counts should be monitored after the first or second injection, and then at regular clinically determined intervals.
|
 |
First Autonomous Diabetic Retinopathy Screening Device Receives FDA Approval
Artificial intelligence (AI) is everywhere, and now it makes its way into the primary care setting in the form of diabetic retinopathy (DR) screening. Eyenuk recently announced the FDA approval of EyeArt—the first cleared autonomous device that can detect both more than mild DR (mtmDR) and vision-threatening DR (vtDR), and has diagnostic outputs for each eye.
In testing the machine, 380 patients enrolled at seven primary care sites, and 275 patients enrolled at four ophthalmology sites. Two-field photographs were taken followed by a dilated four widefield stereo set. Disease detection results were provided for 97% of those tested, and culminated with a 96% sensitivity and 88% specificity for detecting mtmDR; 92% sensitivity and 94% specificity for detecting vtDR. Additionally, all eyes with ETDRS level 43 or higher were correctly identified as mtmDR positive.
The company expects third-party payer coverage by 2021. A CPT code, 9225X, created in May 2019 allows for “imaging of retina for detection or monitoring of disease; with point-of-care automated analysis with diagnostic report; unilateral or bilateral.”
|
 |
Bausch + Lomb Eyes New Company Division
Bausch & Lomb recently announced a company split that will result in an eye-only, publicly traded division. Bausch + Lomb – NewCo is expected to launch in the first quarter of 2021. The other branch will serve as a diversified pharmaceutical company with leading positions in gastroenterology, aesthetics/dermatology, neurology, and international pharmaceuticals, according to August 6, 2020 company press release. The release stated the benefits of separating the disparate business include improved strategic focus and enhanced financial transparency to better enable stakeholders to value each business independently.
|
 |
Medical Optometry America to Open First Practice Soon
Medical Optometry America (MOA) announced the site of its first location: Shrewsbury, Pa. Set to open in late 2020, MOA is a practice management system and franchise model that stands for exceptional medical eye care as the demand for those services increases.
In preparing for launch, MOA created, trademarked, and copywrote a set of practice guidelines for ophthalmic disease and surgical comanagement. The group’s model includes the annual eye physical, and patients will receive a personalized “report card” following the exam. MOA has an academic affiliation with New England College of Optometry (NECO). |

|
IMAGE QUIZ ANSWER
The correct answer, image A, shows active toxoplasmosis. The red arrow indicates a chorioretinal scar while the blue arrow shows the reactivated chorioretinitis. There is moderate vitritis present.
Infection with T. gondii can be congenital or acquired from direct ingestion, such as through contaminated meat or vegetables, unpasteurized dairy products or contaminated water. Ocular toxoplasmosis classically presents with a fluffy white/yellow chorioretinitis with overlying vitritis dubbed the “headlight in the fog.” There usually is an adjacent old chorioretinal scar and spillover anterior uveitis. Even though the infection is self-limited in immunocompetent patients, treatment is generally recommended with an oral antiparasitic agent (such as trimethoprim-sulfamethoxazole) and oral steroids.
B: inactive toxoplasmosis scar C: coloboma D: toxocariasis
|
MEET THE FELLOWS
Dr. Leticia Rousso graduated from the University of Florida in 2011 with a bachelor of science degree in health science. She earned a Doctorate of Optometry at Nova Southeastern University in 2015 and was presented with the Volk Award of Excellence
in Ocular Disease. She pursued her interest in this specialty area and completed a one-year residency at Nova Southeastern University focusing on ocular disease. After graduating from the residency program in 2016, she joined the faculty at NSU as a full-time instructor, where she served as interim clinic chief, taught a module conference course, and held weekly conferences with third-year students. Dr. Rousso also developed the curriculum for the NBEO Board Preparatory course that she has offered to third-year students for the past three years.
Dr. Rousso is currently an assistant professor at Nova Southeastern University College of Optometry. She is an attending optometric physician at The Eye Care Institute, where she precepts third- and fourth-year student doctors in primary care. In addition to serving on several committees, Dr. Rousso has authored numerous poster presentations submitted to AOA and SECO, as well as multiple publications on the topics of retina and glaucoma.
Dr. Rousso is a fellow and member of the American Academy of Optometry. In her leisure time, she enjoys being with her family, traveling, playing sports, and staying fit. |
WHY BECOME AN ORS FELLOW?
By Bill Denton, O.D., F.A.A.O.
Chair, Membership Committee
At some point in your career, you realize you just may be coasting. Your knowledge has been limited to the journals you receive and attempt to read, and the conferences that may not be as fulfilling as they once were. You simply need a challenge that will add an extra dimension to your professional learning.
Fellowship in the Optometric Retina Society (ORS) can provide several benefits in addition to the initial challenge of qualifying for this honor. Plenty of perks accompany your induction, but the coolest part is being associated with a body of knowledge and resources which can help you in many other ways. It is not uncommon to receive weekly thought-provoking emails about challenging cases and treatment dilemmas. Some fellows like to share their awesome cases they have diagnosed, while others post their cases with hopes that other Fellows will suggest an alternative differential diagnosis. At times it is like a round-table of brainstorming, but through the use of modern technology. Fellowship has little obligation with a huge opportunity for professional growth.
If you are up to the challenge of becoming a Fellow of the ORS, feel free to peruse the details and application at www.optometricretinasociety.org. Advice can be given to assist you in your quest. Feel free to contact us.
|
Editor
in Chief
Anna K. Bedwell, OD, FAAO
Co-Editor
Brad Sutton, OD, FAAO |
Journal
Reviewers
Meret Thomas-Huebner, OD
Contributor
Jim Williamson, OD, FAAO
Senior Graphic Designer
Matt Egger
|
Review of Optometry® is published by the Review Group, a Division of Jobson Medical Information LLC (JMI), 11 Campus Boulevard, Newtown Square, PA 19073.
To subscribe to other JMI newsletters or to manage your subscription, click here.
To change your email address, reply to this email. Write "change of address" in the subject line. Make sure to provide us with your old and new address.
To ensure delivery, please be sure to add revoptom@lists.jobsonmail.com to your address book or safe senders list.
Click here if you do not want to receive future emails from Review of Optometry. |
|