 |
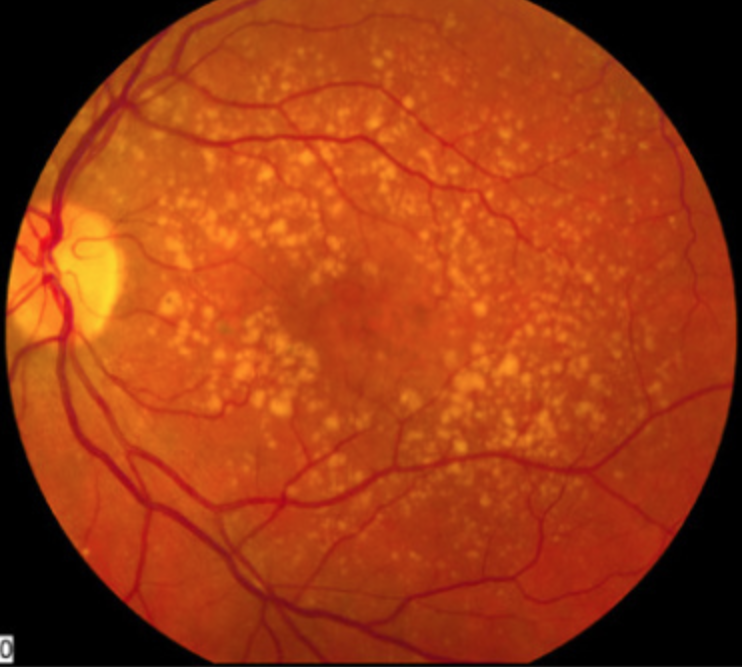
| Autoantibodies against inflammatory proteins were downregulated in patients with AMD. Photo: NEI. Click image to enlarge. |
Anti-retinal autoantibodies are assumed to be associated with age-related macular degeneration (AMD). To investigate this further, researchers evaluated autoantibodies in bloodwork of participants with different stages of AMD in a large population-based, observational cohort study in Germany and found that autoantibodies against inflammatory proteins were downregulated in patients with AMD, but it’s unclear whether autoantibodies play a role during the pathogenesis of the condition. The researchers believe this is the first study of its kind.
A total of 15,010 participants between the ages of 35 and 74 from the Gutenberg Health Study were included. Non-mydriatic fundus photography was performed, and images of the first 5,000 participants were graded based on the Rotterdam Eye Study classification. Blood samples of participants with AMD (n=541) and of age-matched participants without AMD (n=490) were analyzed by antigen-microarrays. Besides descriptive statistics, autoantibody levels were compared, and the association of autoantibody level with AMD was calculated by logistic regression analysis. Possible associations of the autoantibodies and both clinical and laboratory parameters on AMD subjects were analyzed.
Autoantibodies against mitogen-activated protein kinase 3, glutathione peroxidase 4, clusterin, lysozyme, transferrin and protein kinase C substrate 80K-H were downregulated in AMD 1. Autoantibodies against transferrin were significantly downregulated in participants with early AMD and soft, distinct drusen (≥63μm) or pigmentary abnormalities. Autoantibodies against heat shock 70kDa protein 1A and insulin were upregulated in AMD 1.
Autoantibodies against MAPK3 were downregulated in AMD 1. “Hypothetically, the downregulation in the AMD 1 group might indicate a loss of the protective function and an enhancement of the pro-apoptotic activation with resulting retinal pigment epithelium (RPE) degeneration,” the authors explained in their paper.
Diabetes may lead to the accumulation of advanced glycation end products (AGEs), such as in the RPE cell layers or photoreceptors. AGEs and receptors for AGEs were also found in the RPE and the photoreceptors in the maculae of human donor retina from patients with AMD, indicating to some extent similar biological pathways in AMD and diabetes, the authors explained. Antibodies against insulin were significantly upregulated in AMD patients.
In general, it remains unclear whether the described autoantibodies play a causative role during the pathogenesis of AMD or appear as secondary effects during the disease’s progression, the authors concluded.
“Although the role of anti-retinal autoantibodies in the induction or progress of retinal degeneration presently is not well defined, it has been shown that anti-retinal autoantibodies can develop on average three to 15 years prior to the first clinical signs,” they wrote. “In order to clarify the question whether the anti-retinal autoantibodies play a causative role, future longitudinal studies, possibly with additional mass spectrometry to detect associated proteins, could be desirable.”
Korb CA, Lackner KJ, Wolters D, et al. Association of autoantibody levels with different stages of age-related macular degeneration (AMD): results from the population-based Gutenberg Health Study (GHS). Graf Arch Clin Exp Ophthalmol. May 9, 2023. [Epub ahead of print]. |


