Retinal angiomatous proliferation (RAP) is a somewhat rare finding that can lead to a form of end-stage, age-related macular degeneration (AMD) in response to chronic surrounding tissue ischemia. RAP represents the development and formation of a distinctive shunt vasculature originating from the outer retina potentially communicating with deeper choroidal neovascularization.
The presence or development of RAP can indicate an impending, severe visual consequence; therefore, proper recognition of the disorder can allow aggressive therapy to be initiated, if warranted. The development of RAP or lesions can have a potentially devastating effect on central visual acuity and could even lead to an irreversible relative or absolute central scotoma.
History
An 87-year-old white male presented as a new patient in January 2007 with a chief complaint of “worsening hazy vision” in his left eye that he’d first noticed approximately two months prior to his visit.
He reported having uneventful bilateral cataract surgery in 2002; otherwise, he had an unremarkable ocular history. He had recently been prescribed Antivert (meclizine, Pfizer) for symptoms of vertigo without relief, and he was concerned about his vision relative to his vertigo. Otherwise, he had no other remarkable family, systemic or ocular history.
Diagnostic Data
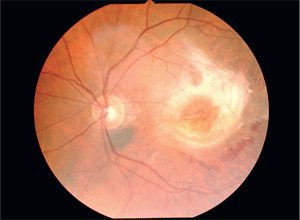
1. Both small- and large-diameter vessels were visible in the center of the lesion. This signified retinal-retinal and retinal-choroidal neovascular shunting protruding directly through the fovea.
Best-corrected visual acuity was 20/30 O.D. and hand motion O.S. Stereopsis was not present with Randot testing. Ishihara color testing was normal O.D., but unreliable O.S.
Pupils were equal, round and responsive to light and accommodation. An afferent pupillary defect was not elicited in the left eye. Extraocular muscles were smooth, with good alignment and no subjective diplopia. There was mild dermatochalasis with slight associated ptosis in each eye; however, levator function was intact in both eyes. Confrontation visual field was full without defect in the right eye, but a distinct central scotomatous defect was evident in the left, with the remaining peripheral field full. Biomicroscopic examination revealed a well-placed, posterior chamber intraocular lens (PC-IOL) in each eye, with deep and quiet anterior chambers. Angles were grade IV by Van Herick’s method O.U.
There was no indication of concurrent or previous ocular inflammatory disease in either eye. The corneas appeared clear, with no evidence of dystrophic or degenerative changes in either eye. Applanation intraocular pressure measured 19mm Hg O.D. and 18mm Hg O.S. Dilated 20D and 90D ophthalmoscopic evaluation of the right eye demonstrated a posterior vitreous detachment (PVD), with an otherwise unremarkable periphery, posterior pole, vessels and optic disc. A cup-to-disc ratio of 0.45 x 0.45 was estimated, and rim tissue appeared healthy.
The macula exhibited only minimal, dry degenerative changes in the form of trace retinal pigment epithelial (RPE) hyperplasia and early drusen formation. Dilated 20D and 90D ophthalmoscopic evaluation of the left eye also revealed the presence of a PVD along with an unremarkable periphery. A cup-to-disc ratio of 0.45 x 0.45 with healthy rim tissue was observed in the left eye
as well.
An underlying choroidal nevus measuring approximately 0.50 x 1.0 disc diameters (DD) was found at the inferior disc margin and was of no consequence. Under 90D biomicroscopy, the macula displayed a large, raised central fibrotic lesion with 360º fibrous proliferation that extended anteriorly into the vitreous. Intraretinal hemorrhages were also present just infero-temporal to the edge of the proliferation. Within the center of the lesion appeared both small- and large-diameter vessels, that represented retinal-retinal and retinal-choroidal neovascular shunting protruding directly through the fovea (figure 1).
Diagnosis
Based on the above findings, we diagnosed the patient with atypical end-stage neovascular AMD. Specifically, we suspected retinal angiomatous proliferation.
Treatment and Follow-Up
Considering the extent of involvement through the fovea as well as the degree of vision loss, the odds for a positive visual outcome resulting from therapeutic intervention were slim. We explained the poor prognosis to the patient, and we recommended observation. A second opinion was subsequently obtained, and a retinal specialist confirmed the diagnosis. Unfortunately, the specialist also agreed that treatment would be ineffective in this case.
In view of the early dry AMD changes present in the fellow eye, along with the associated slight reduction in acuity, careful monitoring was advised for that eye, as well as follow-up appointments every four to six months.
Macular Heidelberg Retinal Tomography (HRT, Heidelberg Engineering), stereo digital photography, ophthalmoscopy, central visual field testing, visual acuity, color vision and home Amsler grid testing are important elements in the prospective management plan. Eventually, ICG angiography may also be necessary if progression ensues.
We prescribed antioxidant vitamin therapy and polycarbonate spectacle lenses with UV protection.
Discussion
The clinical presentation of RAP can be quite dramatic in its ophthalmoscopic findings. As we saw in the case above, advanced progression can reveal a deep, central clustering of retinal neovascular shunt vessels and/or choroidal neovascularization, pigmentary epithelial detachment (PED), serous macular detachment, macular and paramacular fibrosis and adjacent intraretinal hemorrhages. In addition to ischemia, the clinicopathological findings of progressive RAP implicate an inflammatory mechanism in causing this distinctive neovascular pathology.
But, drusen, macrophage migration, vascular endothelial growth factor (VEGF)-positive retinal tissue and hypoxia-inducible factors have also been histologically isolated, pointing to a dual etiology.1
Posterior retinal and choroidal anastomoses have been confirmed utilizing indocyanine green angiography (ICG-A), a superior method for detecting retinal angiomatous proliferation vs. traditional fluorescein angiography (FA).2 The molecular properties of indocyanine green dye prevent it from leaking into subretinal or sub-RPE spaces.
Retinal angiomatous proliferation is a unique neovascular manifestation that is occasionally found in progressive AMD with its own defining characteristics. In contrast to the posterior-anterior choroidal neovascular membrane formation that typifies common wet AMD, RAP first begins in the deep or outer retinal vasculature. Neovascular shunt vessels may advance into the subretinal space, breaking through retinal pigment epithelium and Bruch’s membrane. They eventually find their way into the choroid and may create occasional anastomoses. As exemplified in the case report, central acuity loss with relative or central scotoma can often be the end result.
Studies report varying degrees of RAP prevalence, ranging from 8% to 13%.3-5 AMD is normally a bilateral condition, so the possibility of RAP development and progression in both eyes is a concern. In patients diagnosed with unilateral RAP lesions, the form of neovascularization that develops in the fellow eye is virtually always RAP. In addition, the risk of neovascularization in the fellow eye is higher in patients with RAP than in those with other forms of neovascular AMD.6 With the continued advancement in imaging technology and evaluation methods, perhaps an even greater incidence and prevalence will be found as more clinicians develop awareness and learn to recognize the diagnostic features of RAP.
The pathophysiology of choroidal neovascular membrane (CNVM) formation is not completely understood. Pathologic conditions that involve RPE or damage to Bruch’s membrane have been known to induce neovascular membrane formation by stimulating the release of vascular endothelial growth factor from the RPE. VEGF targets vascular endothelial receptor cells and activates signal transduction pathways to form neovascular vessels.
An additional protein also derived from the RPE was recently discovered, which is now known as pigment epithelium derived factor (PEDF).7 PEDF was found to have an inhibitory effect on ocular neovascularization, whereas VEGF is a known ocular angiogenic stimulator. A balance between PEDF and VEGF may need to be maintained for proper retinal vasculature to exist.7 So, an imbalance in these factors may determine the development of retinal or choroidal neovascularization.7
Considering that traumatic, mechanical or AMD-related effects on the RPE and Bruch’s membrane have been primarily implicated in the formation of ocular neovascularization, specifically classic or occult CNVM, the pathophysiologic mechanism behind intra-retinal neovascularization and RAP may be even more difficult to isolate.
Interestingly, other neovascular biochemical mediators from the vitreous, retina and even the choroid have been associated with new vessel proliferation in the retina and may be contributors to the etiology of RAP. These mediators may also alter capillary permeability to produce leakage, hemorrhages, exudates and the retinal vascular changes characterizing RAP. In that regard, there is predominantly a vertical extension of the deep, intra-retinal neovascularization representing proliferating capillaries extending to the retina’s anterior and posterior boundaries.8
Stages of Progression
Stages of progression of RAP are categorized by the extent of retinal and choroidal involvement.8
• Stage I. Intraretinal neovascularization can be recognized on slit lamp examination as a small nodular mass of angiomatous tissue in the middle and inner retina, accompanied by intraretinal hemorrhages and intraretinal edema. A small cluster of intraretinal dilated capillaries is the earliest clinically recognized manifestation in RAP. Retinal-retinal anastomoses (RRA) can be seen in 30% of cases.
• Stage II. A localized neurosensory retinal detachment and an increase in intraretinal edema are seen. Pre-, intra- and subretinal hemorrhages are present. Associated serous PED can be seen in 94% of eyes as the subretinal neovascularization reached or fused with the RPE. Nearly 50% of patients are diagnosed in this stage, as the neovascularization progresses more rapidly, becomes associated with an increase in the exudative changes, and induces alterations in vision. RRA is seen in 39% of patients.
• Stage III. Clinical and angiographic examinations can clearly demonstrate the presence of CNVM, but the condition is usually confirmed in just 7% of the cases. CNVM in the subretinal space, or the presence of a vascularized PED, is best determined with ICG-A. Indocyanine green angiography has been found to be the most effective in resolving findings in all stages.
Ocular coherence tomography is useful in evaluating and documenting RAP. OCT can reveal the typical pattern of structural change in RAP demonstrating increased foveal thickness, cystoid macular edema (CME), serous retinal detachment, reflective intraretinal neovascular vessels, and RPE elevation and detachment. These findings correlate well with ICG angiography, suggesting that utilization of both methods may provide a more complete diagnostic picture.9
Management Strategies
Therapeutic options include periocular sub-Tenon’s Kenalog (triamcinolone, Bristol-Myers Squibb) injections, anti-VEGF therapy, conventional focal argon laser photocoagulation and intravitreal Kenalog in conjunction with PDT and Visudyne (verteporfin, QLT PhotoTherapeutics), which have been found effective in reducing or eliminating retinal edema, causing regression of neovascularization, and stabilizing or improving visual acuity.10 These modalities have been used separately as primary mono-therapies, as well as in combination or succession. They exhibit various degrees of success. In patients with evidence of stage III RAP with CNVM, one recent study demonstrated a sub-Tenon’s injection of Kenalog delayed moderate to severe visual loss after a period of 10 to 11 months.11
Two three-month trials of Avastin (bevacizumab, Genentech) have also produced encouraging outcomes. Reduction of leakage, diminished intra- and subretinal fluid, and an increase in visual acuity was apparent in just four weeks after only one injection.12,13 After three months, another trial found that a significant decrease in macular thickness and improvement or stabilization of visual acuity was demonstrated as well.14
Direct focal laser argon photocoagulation of RAP lesions seems to be a practical and safe method of managing neovascular leakage; however, results are shown to be better for patients with early-stage disease. Focal laser treatment alone applied purely to the intraretinal component of the lesion may be adequate to reduce leakage and may impede the angiomatous process resulting in stabilization of the pathology and visual acuity.15-17
In cases of considerable macular edema in stage I disease, direct laser photocoagulation combined with prior intravitreal triamcinolone injection is also considered a viable treatment option that has yielded improved visual acuity.17 Not surprisingly, a “multiple approach” strategy of focal laser photocoagulation, PDT with verteporfin and intravitreal Kenalog was found to be most efficacious in stage I disease.15 Of course, long-term stabilization or advancement may require additional laser treatments or adjunctive measures.
There is evidence of complete resolution of angiographic leakage achieved in 83% of patients after 12 months who initially presented with stage I or II RAP progression and underwent simultaneous combined treatment of intravitreal triamcinolone with PDT and verteporfin.10 OCT confirmed this reduction or elimination of retinal edema and regression of neovascularization. Visual acuity improved in 35% and stabilized in 47% of patients10 A small percentage of patients developed recurrent leakage after six to 12 months, but resolved completely after repeated, simultaneous, combined treatment10
A complete remodeling of intraretinal vascular structure has also been exhibited in stage II RAP after successive triamcinolone and PDT with verteporfin treatment.18 A feeding retinal artery that shunted significant blood flow from an arteriole toward an intraretinal neovascular complex before treatment regained a normal appearance after treatment. Blood flow was restored through the original retinal arteriole, and the feeder RAP vessel was no longer visible.18
Another study examined the use of intravitreal triamcinolone followed by PDT with verteporfin in eyes with stage II RAP and found a potential benefit in terms of stabilization or even improvement of vision.19 After follow-up visits at three, six, nine, and 15 months, the greatest improvement in visual acuity was found initially and declined slightly over time; however, there was overall improvement from baseline. Additionally, only 18% of patients lost three or more lines of visual acuity. Retreatment was required in 45% of eyes.19
Of course, the final outcome of therapy hinges on the RAP stage when treatment is initiated. Smaller, stage I lesions will naturally have a greater chance of being successfully treated. Deeper, stage II and III involvement will be most resistant.
Additional management strategies have been proven ineffective. Posterior juxtascleral injection of anecortave acetate, a synthetic anti-angiogenic derived from cortisol, has been found to reduce capillary permeability (and thus exudative material) in patients with RAP; however, progression of neovascularization and significant loss of vision has been the outcome in all patients.20 Surgical ablation combined with PDT has also been attempted, but it was found inadequate due to a high incidence of reperfusion from retinal inflow vessels.21
Retinal angiomatous proliferation is indeed a unique variation of neovascular AMD. Considered a somewhat newly described entity, RAP has undoubtedly always been a potential part of the AMD process. Advancing technology in diagnostic imaging will allow for a better understanding of the pathomorphologic stages. Unfortunately, with the current therapeutic modalities available today, progressive AMD leading to RAP has a poor visual prognosis. However, anti-angiogenesis research continues to be widely studied, and it will unquestionably lead to more definitive and effective therapeutic protocols.
Dr. Martinelli practices in Charleroi and Uniontown, Pa. He is a clinical preceptor at Pennsylvania College of Optometry at Salus University, and he is a consultant for Alcon, Allergan, Abbott Medical Optics and Lumenis.
1. Shimada H, Kawamura A, Mori R, Yuzawa M. Clinicopathological findings of retinal angiomatous proliferation. Graefes Arch Clin Exp Ophthalmol. 2007 Feb;245(2):295-300.
2. Iranmanesh R, Eandi CM, Peiretti E, et al. The nature and frequency of neovascular age-related macular degeneration. Eur J Ophthalmol. 2007 Jan-Feb;17(1):75-83.
3. Kuerzinger GR, Lang GK, Lang GE. Retinal angiomatous proliferation in age-related macular degeneration. Klin Monatsbl Augenheilkd. 2006 Aug;223(8):691-5.
4. Salazar-Diez JL, Iturralde-Errea D, Diaz-de-Durana-Santacoloma E, et al. Results of treatment of retinal angiomatous proliferation with photodynamic therapy. Arch Soc Esp Oftalmol. 2006 Jul;81(7):401-4.
5. Cohen SY, Dubois L, Tadayoni R, et al. Prevalence of reticular pseudodrusen in age-related macular degeneration with newly diagnosed choroidal neovascularisation. Br J Ophthalmol. 2007 Mar;91(3):354-9.
6. Gross NE, Aizman A, Brucker A, et al. Nature and risk of neovascularization in the fellow eye of patients with unilateral retinal angiomatous proliferation. Retina. 2005 Sep;25(6):713-8.
7. Wu L. Choroidal Neovascularization. Available at: www.emedicine.com/oph/topic534.htm (Accessed June 2007).
8. Van de Moere A, Sandhu SS, Talks SJ. Retinal Angiomatous Proliferation: Diagnosis and Treatment Options. Ophthalmology. 2003 Aug;110(8):1517-25.
9. Politoa A, Napolitano MC, Bandello F, Chiodini RG. The role of optical coherence tomography (OCT) in the diagnosis and management of retinal angiomatous proliferation (RAP) in patients with age-related macular degeneration. Ann Acad Med Singapore. 2006 Jun;35(6):420-4.
10. Van de Moere A, Kak R, Sandhu SS, Talks SJ. Anatomical and visual outcome of retinal angiomatous proliferation treated with photodynamic therapy and intravitreal triamcinolone. Am J Ophthalmol. 2007 Apr;143(4):701-4.
11. Rutishauser-Arnold Y, Tholen AM. Periocular sub-tenon triamcinolone acetonide injections for the treatment of retinal angiomatous proliferation (RAP) and occult choroidal neovascularization. Klin Monatsbl Augenheilkd. 2007 Apr;224(4):269-73.
12. Pedersen R, Soliman W, Lund-Andersen H, Larsen M. Treatment of choroidal neovascularization using intravitreal bevacizumab. Acta Ophthalmol Scand. 2007 Aug;85(5):526-33.
13. Joeres S, Heussen FM, Treziak T, et al. Bevacizumab (Avastin) treatment in patients with retinal angiomatous proliferation. Graefes Arch Clin Exp Ophthalmol. 2007 Nov;245911):1597-602.
14. Meyerle CB, Freund KB, Iturralde D, et al. Intravitreal bevacizumab (Avastin) for retinal angiomatous proliferation. Retina. 2007 Apr-May;27(4):451-7.
15. Olea JL, Sastre M, Aragon JA, et al. Treatment of retinal angiomatous proliferation (RAP). A retrospective study. Arch Soc Esp Oftalmol. 2007 Jan;82(1):27-35.
16. Johnson TM, Glaser BM. Focal laser ablation of retinal angiomatous proliferation. Retina. 2006 Sep;26(7):765-72.
17. Krieglstein TR, Kampik A, Ulbig M. Intravitreal triamcinolone and laser photocoagulation for retinal angiomatous proliferation. Br J Ophthalmol. 2006 Nov;90(11):1357-60.
18. Bottoni F, Romano M, Massacesi A, Bergamini F. Remodeling of the vascular channels in retinal angiomatous proliferations treated with intravitreal triamcinolone acetonide and photodynamic therapy. Graefes Arch Clin Exp Ophthalmol. 2006 Nov;244(11):1528-33.
19. Mantel I, Ambresin A, Zografos L. Retinal angiomatous proliferation treated with a combination of intravitreal triamcinolone acetonide and photodynamic therapy with verteporfin. Eur J Ophthalmol. 2006 Sep-Oct;16(5):705-10.
20. Klais CM, Eandi CM, Ober MD, et al. Anecortave acetate treatment for retinal angiomatous proliferation: a pilot study. Retina. 2006 Sep;26(7):773-9.
21. Nakata M, Yuzawa M, Kawamura A, Shimada H. Combining surgical ablation of retinal inflow and outflow vessels with photodynamic therapy for retinal angiomatous proliferation. Am J Ophthalmol. 2006 May;141(5):968-70.

