Adult-onset vitelliform maculopathy initially presents in adulthood as bilateral subtle and often asymptomatic macular lesions, which are often misdiagnosed as early age-related macular degeneration (AMD). Although the prognosis is generally good with this condition, in some cases, retinal findings may progress to visually debilitating and blinding retinal pigment epithelium (RPE) disruption. So, in advanced cases, standard of care management should include a referral to a low vision specialist, who can improve visual function and increase quality of life for affected patients.
This case report offers a comprehensive overview of the medical intervention and low vision management of an elderly white male with complaints of a gradual and asymmetrical decrease in vision who was subsequently diagnosed with adult-onset vitelliform maculopathy.

A large, well-demarcated vitelliform lesion with underlying cuticular drusen was visible, along with sub- and intra-retinal pigment epithelium fluid accumulation, in the right eye.
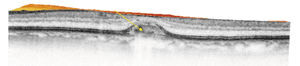
The vitelliform lesion in the left eye is smaller than that in the right and is without fluid accumulation. Cuticular drusen is visible within the area of RPE disruption.
History
An 80-year-old white male presented to our eye clinic describing a gradual decrease in vision in both eyes. He had been followed for 11 years for primary open-angle glaucoma in his right eye only, as well as AMD, pseudophakia, dry eye syndrome and chronic allergic conjunctivitis in both eyes. Previous examinations were negative for diabetic or hypertensive retinopathy.
Current ocular medications included dorzolamide hydrochloride b.i.d., timolol maleate q.d. and brimonidine tartrate b.i.d., all O.D. only for treatment of his glaucoma, as well as preservative-free artificial tears q.i.d. O.U. and Zaditor (ketotifen fumarate, Alcon) b.i.d. O.U. for treatment of dry eyes and allergic conjunctivitis.
His systemic history was positive for insulin-dependent diabetes mellitus, hypertension and hypercholesterolemia with carotid artery disease.
Systemic medications included low-dose aspirin (81mg), clonidine hydrochloride, simvastatin, Lantus (insulin glargine, Sanofi-Aventis) and Humalog (insulin lispro, Eli Lilly) injections, lisinopril, metoprolol succinate, and nitroglycerin. The patient had no known drug allergies.
Diagnostic Data
The patient initially presented in May 2000 because he had lost his habitual spectacles. Uncorrected visual acuity was 20/60 O.D. and 20/50 O.S. Best-corrected visual acuity improved to 20/20-2 O.D. and O.S. with a thorough refraction. Dilated fundus evaluation was unremarkable except for the presence of increased macular pigmentation and fine drusen, which were diagnosed as early dry AMD.
Subsequent evaluations revealed progressive RPE dropout, clumping and drusen. The patient experienced a corresponding vision loss, O.D. greater than O.S.
Nine years later, the patient’s best-corrected visual acuity had progressed to 20/50-1 O.D. and O.S. External examination, including extraocular motilities and confrontation fields, was normal with no afferent pupillary defect. Biomicroscopy found normal and healthy anterior segment structures. Goldmann applanation pressures measured 20mm Hg O.D. and 18mm Hg O.S. Dilated fundus evaluation revealed large, bilateral choroidal lesions with overlying pigment epithelial detachments, O.D. greater than O.S.
Optical coherence tomography (OCT) scans revealed bilateral, dense choroidal elevations with questionable fluid accumulation in the right eye, irregular reflectivity in the left eye, and possible choroidal neovascular membranes in both eyes.
We referred the patient to a retinal specialist for further evaluation. At that visit, Spectralis OCT (Heidelberg Engineering) scans of the macula revealed vitelliform lesions in both eyes, with subretinal fluid only in the right eye.
Diagnosis
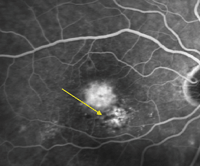
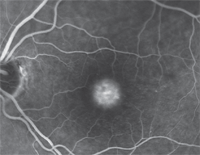
Fluorescein angiography revealed central staining O.U., with an occult choroidal neovascular membrane inferior to the macula in the right eye and no leakage or membrane noted in the left.
After a thorough review of past macular OCT scans and fluorescein angiography studies, it appeared that bilateral and well-demarcated vitelliform lesions had been present as far back as December 2010.
The patient was formally diagnosed with adult-onset vitelliform maculopathy (AOVM) in both eyes, with an active choroidal neovascular membrane in the right eye only.
Treatment and Follow-up
Due to the choroidal neovascular membrane in the right eye, the patient received approximately 20 monthly intravitreal injections of either Avastin (bevacizumab, Genentech/Roche) or Lucentis (ranibizumab, Genentech/Roche) over the course of two years. Residual RPE disruption resulted in decreased visual acuity, central scotomas and metamorphopsias in both eyes, which affected the patient’s quality of life. At this point, the patient’s visual acuity was 20/100-2 O.D. and 20/50-2 O.S. with no improvement on pinhole. This prompted a referral from the retina clinic to the low vision specialist.
Low Vision Evaluation
The patient’s entering complaint for the low vision examination was decreased vision at near, with the right eye affected more than the left. Goals for the patient were to increase his ability to read newspaper print and to be able to read words on his 32-inch television screen.
At this visit, the patient’s best-corrected distance visual acuity through trial frame refraction was 20/100 O.D. with eccentric viewing at 3 o’clock, and 20/30 centrally O.S. without eccentric viewing. The patient was able to read 0.6m optotype at 26cm through a +3.00D add over the manifest without rivalry. But, binocular arc perimetry visual field testing revealed a paracentral 18-degree scotoma as well as decreased contrast sensitivity in both eyes.
After a detailed examination, the patient was prescribed near spectacles with a safety frame, polycarbonate lenses and anti-reflective coatings. Yellow slip-in filters were given to the patient for use when reading to increase contrast. The patient was also given a pocket typoscope to decrease lateral masking, and new spectacles were ordered. Also, amber-filtered lenses were ordered for outdoor use to lessen glare and photophobia. MaxTV telescopic glasses (Eschenbach Optik) enhanced the patient’s distance acuity; these were recommended for use when the patient was stationary and for viewing distant objects such as the television. No further referral for orientation and mobility training was recommended.
The devices were mailed to the patient’s home, and he was instructed to return to the low vision clinic as needed. He was also scheduled to continue care with the retinal specialist for ongoing medical management.
Discussion
Adult-onset vitelliform maculopathy, also known as adult foveo-macular vitelliform dystrophy or late-onset Best’s disease, is a rare condition with onset between the fourth and fifth decades of life, and mostly affecting females.1-3 The patient classically presents with bilateral, round and discrete yellow lesions that are in or near the macula, which result in disturbances in central vision.1,2 The disease rarely progresses to development of choroidal neovascular membranes or areas of geographic atrophy and concurrent cuticular drusen.1
AOVM has been controversially theorized to be a variant of Best’s disease and is often categorized as a pattern dystrophy.4,5 Several hypotheses exist for the origin of the characteristic yellow lesions, including the theory that they represent melanin and lipofuscin-laden macrophages or are the result of a proliferation of RPE cells or subretinal fibrin.6
In AOVM, fluorescein angiography (FA) results vary with disease progression. Early changes appear as central hyper-fluorescent staining due to RPE disruption.7 More developed disease shows a “corona sign” or central hypo-fluorescent atrophic lesion with surrounding hyperfluorescent halos due to loss of photoreceptor outer segments.7 Vitelliform lesions tend to stain late.1 Other diagnostic findings include an electro-oculogram (EOG) and electroretinogram (ERG) that are relatively normal and mild tritan color vision defects.1,3
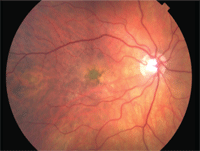
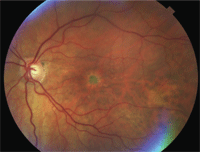
Fundus evaluation reveals subtle and irregular vitelliform lesions after three years of intravitreal injections in the right eye (on left) and observation in the left (on right).
The primary differential diagnoses for AOVM include Best’s disease and AMD.8 Best’s disease is genetically similar to AOVM, but Best’s disease has a characteristically larger lesion size than seen in AOVM and presents with an earlier age of onset, abnormal EOG and a poorer visual prognosis.6,8,9
AOVM is often misdiagnosed as AMD because macular degeneration occurs much more commonly than AOVM.8 Differentiation is possible due to the later onset of macular degeneration as well as with macular OCT analysis.8 Exudative macular degeneration shows a characteristic and irregular intra- and subretinal serous shadowing with fibrovascular changes.3 Alternatively, AOVM lesions have discrete and well-demarcated “hills” of subfoveal disruption with no optical shadowing effects.3
Current medical management of AOVM has been disappointing. Verteporfin photodynamic therapy (PDT) has been attempted unsuccessfully.3 Monthly intravitreal injections of the anti-vascular endothelial growth factor (anti-VEGF) medications Lucentis and Avastin have been shown to produce short-term visual improvement.3 The FDA recently approved the use of another anti-VEGF intravitreal injection known as Eylea (aflibercept, Regeneron).10 The pivotal aspect of this medication is that it is used only once per month for three months, then once every two months thereafter.10 This requires fewer injections per year than its monthly-injection predecessors, Lucentis and Avastin.10
In milder presentations, visual prognosis for AOVM is typically good because the lesion lies in the RPE and does not involve the sensory retina.1,2 Prognosis worsens as subretinal fluid accumulates from choroidal disruption with progression to exudative detachments.1 Visual prognosis is directly correlated with the magnitude of disconnect between the photoreceptor’s inner and outer segments, as well as that between the RPE and choriocapillaris layers.6,11 Atrophy and thinning of affected retina are concerning because they increase the likelihood for development of a full-thickness macular hole, which can be visually debilitating.4
Low vision consultation is warranted for advanced cases of AOVM when the patient experiences difficulty performing daily tasks and is motivated for intervention. Laying the groundwork for the evaluation requires asking the patient questions to determine his or her functional status:
• “What brings you in to the examination?”
• “Do you live alone or with a spouse?”
• “Do you have a physical impairment or notice any environmental issues that hinder your mobility?”
• “How well do you get around in both familiar and unfamiliar settings?”
In order to determine in which direction the evaluation should go, the examiner should determine the previous devices the patient has used, as well as the level of effectiveness experienced. The patient’s chief complaint often guides the goal-selection portion of the examination.
Visual acuity should be measured with and without correction. Central acuity is required for determining whether legal blindness exists, but eccentric viewing acuities offer a valuable method for gauging the functional capacity of the patient. Be aware that reading speed drastically decreases as the distance of the preferred retinal locus (or eccentric viewing area) increases from central fixation.12
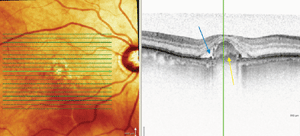
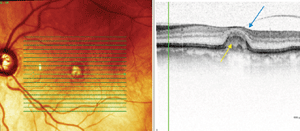
Spectralis OCT scans of the vitelliform lesions: Persistent subretinal fluid can be noted in the right eye (top), with a partial posterior hyaloid detachment appreciated in the left (bottom), and cuticular drusen confirmed in both eyes.
Notations such as “hand motion” and “counting fingers” to describe visual acuity should be avoided due to limited legal justification and variable reproducibility.13
For patients with visual impairment, the examination is limited by the use of the Snellen distance acuity chart.13 The Feinbloom chart has been found to be more beneficial than the Snellen chart due to varying optotypes and letter graduations, as well as its ability to measure up to 20/2800 distance visual acuity.13,14 The MNRead chart has been favored at near due to its logarithmic scale, which results in the ability to determine an estimated add power.13
Contrast sensitivity measurement is essential for patients with central visual impairment. Amplifying print contrast has been found to surpass enlarging font size as a technique for increasing reading efficiency in patients with visual impairment.15 The Pelli-Robson chart is favored for calculating impaired contrast due to its logarithmic contrast progression.13
Visual field testing is necessary for determining functional visual status. Central scotomas and metamorphopsias may be evaluated with use of the Amsler grid.16 Home Amsler grid testing also provides a proactive way for patients to monitor their own monocular visual changes.16
The optical foundation of the examination is laid by obtaining an accurate manifest refraction. Due to the patient’s decreased visibility through the phoropter, individuals with central visual field loss should always receive a trial frame refraction.14 Retinoscopy and keratometry data are also helpful tools for guiding challenging refractions.13
When prescribing spectacles for patients with visual impairment, a safety frame with polycarbonate lenses provides additional impact resistance, while anti-reflective coatings provide added comfort from glare. For reading glasses, as the power of the lenses increases, base-in prism should be added to the prescription to increase binocularity.17 High-powered lenses may be prescribed up to +10.00D for visually impaired patients; beyond this amount, consider electronic aids.17
Handheld and stand magnifiers as well as visors are viable options for patients at near. Note that increasing a device’s magnification decreases the visual span (or field) for reading. So, increasing magnification at near may not be the most effective way of increasing reading efficiency.18 Reading efficiency has been found to improve with use of a typoscope, which aids in decreasing the lateral masking effect of surrounding letters and words.18 This lateral masking effect refers to the negative effect adjacent words or letters may have on reading efficiency.18
Mounted and handheld telescopes are ideal for spotting objects at a distance.19,20 Although dynamic visual acuity may improve with adaptation, these devices are typically recommended for use only when the patient is stationary, due to an increased risk of falling due to disorientation.19,20
A few electronic alternatives for patients with advanced central vision loss include closed-circuit televisions, tablets and portable electronic magnifiers.21 Electronic talking or large-face wristwatches are also routinely considered for patients with central vision loss.21
AOVM is a condition that is often misdiagnosed as AMD. It is important to note the appearance on macular OCT and FA of the well-demarcated vitelliform lesions. These findings will guide the practitioner to the diagnosis of AOVM.
Once diagnosed, management involves the collaboration of care for visually impaired patients by low vision specialists and retinal specialists. Due to the increased life expectancy and concerns for improving quality of life for affected patients, demand for low vision rehabilitation is growing.22
Currently, our patient continues to be followed by a retinal specialist and is undergoing treatment with Eylea for persistent subretinal fluid in the right eye. His vision has stabilized at 20/150 O.D. and 20/50- O.S., and he is scheduled to return to the low vision clinic as needed.
Dr. Hodges is currently completing her residency at the Lake City VA Medical Center in Florida. She will be joining a private practice in Winter Haven, Fla., following the completion of her residency in June 2012. Dr. McGonigal is the Associate Chief of Optometry and Optometry Residency Coordinator at the Lake City VA Medical Center.
1. Finger RP, Charbel Issa P, Kellner U, et al. Spectral domain optical coherence tomography in adult-onset vitelliform macular dystrophy with cuticular drusen. Retina. 2010 Oct; 30(9): 1445-64.
2. Friedman NJ, Kaiser PK, Pineda R. Massachusetts Eye and Ear Infirmary Illustrated Manual of Ophthalmology. 3rd ed. Amsterdam: Saunders Elsevier; 2009:169-71, 448.
3. Gallego-Pinazo R, Dolz-Marco R, Pardo-López D, et al. Primary intravitreal ranibizumab for adult-onset foveomacular vitelliform dystrophy. Graefes Arch Clin Exp Ophthalmol. 2011 Mar;249(3):455-8.
4. Kanski J. Clinical Ophthalmology: A Systematic Approach. 6th ed. Edinburgh: Elsevier Butterworth-Heinemann; 2007: 676-9.
5. Seddon JM, Afshari MA, Sharma S, et al. Assessment of mutations in the best macular dystrophy (VMD2) gene in patients with adult-onset foveomacular vitelliform dystrophy, age-related maculopathy, and bull’s eye maculopathy. Ophthalmology. 2001 Nov; 108(11): 2060-7.
6. Querques G, Forte R, Querques L, et al. Natural course of adult-onset foveomacular vitelliform dystrophy: a spectral-domain optical coherence tomography analysis. Am J Ophthalmol. 2011 Aug;152(2):304-13.
7. Jarc-Vidmar M, Kraut A, Hawlina M. Fundus autofluorescence imaging in Best’s vitelliform dystrophy. Klin Monbl Augenheilkd. 2003 Dec;220(12):861-7.
8. Kandula S, Zweifel S, Freund KB. Adult-onset vitelliform detachment unresponsive to monthly intravitreal ranibizumab. Ophthalmic Surg Lasers Imaging. 2010 Nov-Dec;41 Suppl:S81-4.
9. Ehlers J, Shah CP (eds). The Wills Eye Manual Office and Emergency Room Diagnosis and Treatment of Eye Disease. 5th ed. Boston: Lippincott Williams & Wilkins; 2008: 326-27.
10. Pollack A. Success Long in Coming for Eylea, a Vision Treatment. New York Times. 21 Nov. 2011. B3. Print.
11. Puche N, Querques G, Benhamou N, et al. High-resolution spectral domain optical coherence tomography features in adult-onset foveomacular vitelliform dystrophy. Br J Ophthalmol. 2010 Sep;94(9):1190-6.
12. Battista J, Kalloniatis M, Metha A. Visual function: The problem with eccentricity. Clin Exp Optom. 2005 Sep;88(5):313-21.
13. Brown B. The Low Vision Handbook for Eyecare Professionals. 2nd ed. Thorofare, NJ: Slack; 2007: 80-8.
14. Sunness JS, El Annan J. Improvement of visual acuity by refraction in a low-vision population. Ophthalmology. 2010 Jul;117(7):1442-6.
15. Hazel CA, Petre KL, Armstrong RA, et al. Visual function and subjective quality of life compared in subjects with acquired macular disease. Invest Ophthalmol Vis Sci. 2000 May;41(6):1309-15.
16. Harvey PT. Common eye diseases of elderly people: Identifying and treating causes of vision loss. Gerontology. 2003 Jan-Feb;49(1):1-11.
17. Christoforidis JB, Tecce N, Dell’Omo R, et al. Age related macular degeneration and visual disability. Curr Drug Targets. 2011 Feb;12(2):221-33.
18. Calabrèse A, Bernard JB, Hoffart L, et al. Small effect of interline spacing on maximal reading speed in low-vision patients with central field loss irrespective of scotoma size. Invest Ophthalmol Vis Sci. 2010 Feb;51(2):1247-54.
19. Demer JL, Porter FI, Goldberg J, et al. Dynamic visual acuity with telescopic spectacles: improvement with adaptation Invest. Invest Ophthalmol Vis Sci. 1988 Jul;29(7):1184-9.
20. Demer JL, Porter FI, Goldberg J, et al. Predictors of functional success in telescopic spectacle use by low vision patients. Invest Ophthalmol Vis Sci. 1989 Jul;30(7):1652-65.
21. Minto H, Butt IA. Low vision devices and training. Community Eye Health. 2004;17(49):6-7.
22. Vingolo EM, Salvatore S, Cavarretta S. Low-vision rehabilitation by means of MP-1 feedback examination in patients with different macular disease: A pilot study. Appl Psychophysiol Biofeedback. 2009 Jun;34(2):127-33.

