 A 67-year-old Hispanic male presented with a history of visual distortion in his left eye. His best-corrected visual acuity measured 20/20 OD and 20/30 OS. Amsler grid testing was positive for metamorphopsia OS.
A 67-year-old Hispanic male presented with a history of visual distortion in his left eye. His best-corrected visual acuity measured 20/20 OD and 20/30 OS. Amsler grid testing was positive for metamorphopsia OS.
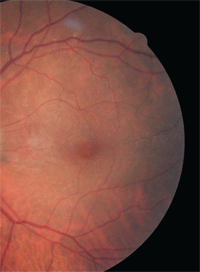
Dilated fundus exam revealed wrinkling within the left macula (figure 1). A spectral-domain optical coherence tomography (SD-OCT) scan of the left eye revealed the presence of vitreomacular traction (VMT) syndrome (figure 2). What are his management options?
PVD and VMT
A posterior vitreous detachment (PVD) is a normal physiologic process that invariably occurs with advanced age. Age-related biochemical changes result in vitreal liquefaction and weakening of the vitreoretinal interface. This process contributes to the development of a PVD. Such detachments often are associated with a complete separation of the vitreous cortex from the internal limiting membrane (ILM). Sometimes, however, a partial PVD may result. In these instances, the remaining adherence to an area of firm vitreoretinal attachment may yield a number of distinct conditions––including VMT syndrome.
VMT is a macular entity associated with an incomplete PVD. The partial vitreomacular adhesion induces tractional forces resulting in both structural and functional damage. There are an array of variable symptoms, which may include metamorphopsia, decreased visual acuity and photopsia. The capacity of OCT to provide high-resolution images of the vitreal-retinal interface has fostered a better understanding of VMT.
VMT represents a broad spectrum of retinal diseases, including cystoid macular edema (CME), epiretinal membranes (ERMs) and macular hole (MH) formation.1,2 The natural disease course is variable. Depending on the individual case, patients may remain stable; experience a spontaneous, complete PVD; or continue to progress.3,4
Traditional Intervention
Although many cases simply are monitored, disease progression and associated visual decline often make pars plana vitrectomy (PPV) a necessity. Progression may lead to visual distortion, vision loss and/or further deterioration of the retinal structures––as well as associated complications.
1, 2. Dilated fundus exam (left) and OCT scan (bottom) of our patient’s left eye.
Should he be monitored for VMT progression, or is he a potential candidate for intravitreal Jetrea?

PPV has been shown to provide a dramatic normalization of the macular contour and a subsequent improvement in symptoms.5 Just like any surgical procedure, however, PPV carries inherent risks––including cataracts, retinal detachment, residual vision loss and infection. Further, the postoperative recovery period can be both taxing and woefully inconvenient for the patient.
Intravitreal Microplasmin
Previous studies have shown that autologous plasmin enzyme had lysing capabilities, which could help induce a PVD.6 Microplasmin is a recombinant protease plasmin with proteolytic properties against fibronectin and lamin (components of the vitreoretinal interference).
It functions as a thrombolytic agent and causes a pharmacological vitreolysis. The introduction of microplasmin into the vitreous would, in essence, induce a nonsurgical PVD. The enzymatic agents alter the biochemistry of vitreous, yielding both vitreous liquefaction and a separation between the vitreous cortex and ILM.
Two multicenter Phase III clinical studies (the MIVI-TRUST trials) evaluated the safety and efficacy of a single 0.125mg intravitreal injection of microplasmin for the treatment of VMT.7 Inclusion criteria included observation of VMT via OCT and associated visual acuity of 20/25 or worse.
Approximately 650 individuals with various maculopathies, including VMT, ERMs and MHs, were evaluated. Patients were randomly assigned to receive either microplasmin or a placebo injection, and were evaluated during the subsequent six months. VMT resolution was noted in 26.5% of those in the treatment group, compared to 10% in the placebo group.7 Additionally, MH closure was documented in 40.6% of the treated group vs. just 10.6% of the placebo group.7
Ocular side effects associated with microplasmin injection were minimal and transient. (In fact, most side effects were local and associated with the intravitreal injection itself.) The most common adverse effects included floaters, mild pain and conjuctival hemorrhage. Serious adverse effects, such as retinal detachment, only were observed in 2% of the treated group vs. 4% in the placebo group.7
The trial data collected by the MIVI-TRUST researchers facilitated FDA approval of Jetrea in October 2012, which launched commercially January 2013. For the first time ever, our patients now have more than one potential treatment option for symptomatic VMT. But will Jetrea, in fact, change the standard of care for those with VMT syndrome? Only time will tell.
Drs. Shechtman and Karpecki have no direct financial interest in any of the products mentioned.
1. Smiddy WE. Macular hole update 2006. Retinal Physician. July 2006. Available at:
www.retinalphysician.com/articleviewer.aspx?articleid=100221. Accessed April 15, 2013.
2. Smiddy WE, Michels RG, Green WR. Morphology, pathology and surgery of idiopathic vitreoretinal macular disorders. Retina. 1990;10(4):288-96.
3. Hikichi T, Yoshida A, Trempe CL. Course of vitreomacular traction syndrome. Am J Ophthalmol. 1995 Jan;119(1):55-61.
4. Sulkes DJ, IP MS, Baumal CR, et al. Spontaneous resolution of vitreomacular traction documented by optical coherence tomography. Arch Ophthalmol. 2000 Feb;118(2):286-7.
5. Machemer R, Parel JM, Norton EW. Vitrectomy: a pars plana approach. Technical improvements and further results. Trans Am Acad Ophthalmol Otolaryngol. 1972 Mar-Apr;76(2):462-6.
6. Trese MT. Enzymatic vitreous surgery. Semin Ophthalmol. 2000 Jun;15(2):116-21.
7. Stalmans, P, Benz MS, Gandorfer A. Enzymatic vitreolysis with ocriplasmin for vitreomacular traction and macular holes. N Engl J Med. 2012 Aug 16;367(7):606-15.

