Ongoing advances in topical ocular therapeutics have given the eye care provider more options in the treatment of ocular inflammation than ever. In this article, we review the various topical corticosteroid and nonsteroidal anti-inflammatory drug (NSAID) options available. A closer look at when and how to implement these medications into your treatment regimen will help you care for all of your patients who present with ocular inflammation. Additionally, a succinct review of immunomodulators for the dry eye patient will hone in on this common cause of inflammation.
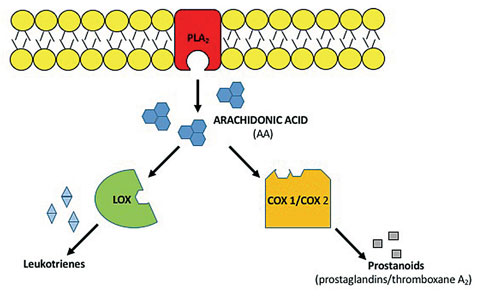 |
| Fig. 1. This illustration demonstrates the inflammatory pathway of the innate immune system: PLA2, LOX and COX-1/COX-2. |
The Inflammatory Pathway
The successful treatment of ocular inflammation requires a solid foundation and understanding of the inflammatory pathway (Figure 1). Inflammation is the body’s response to repair tissue to normal structure and physiologic function. In the presence of tissue damage or an offending agent, neutrophils and monocytes recruit macrophages to remove the offending stimulus.1 This recruitment initiates the healing process and manifests the cardinal signs of inflammation: redness, swelling, heat and pain.1
Tissue injury causes degradation and lysis of the cell membrane via the phospholipase A2 (PLA2) enzyme, resulting in arachidonic acid (AA) formation. AA mediates the inflammatory cascade and is metabolized by 5-lipoxygenase (LOX) and cyclooxygenase isoenzymes (COX-1/COX-2).1 When AA is metabolized via LOX, the generated leukotrienes recruit white blood cells to the damaged area.1 Similarly, when AA is metabolized via the COX-1/COX-2 isoenzymes, prostanoids (prostaglandins and thromboxane A2) are produced.1 Prostanoid formation increases arterial dilation and vascular permeability, which account for the redness, edema and pain associated with inflammation.1
The two major classes of anti-inflammatories—corticosteroids and NSAIDs—each cause different pathway inhibition (Figure 2).
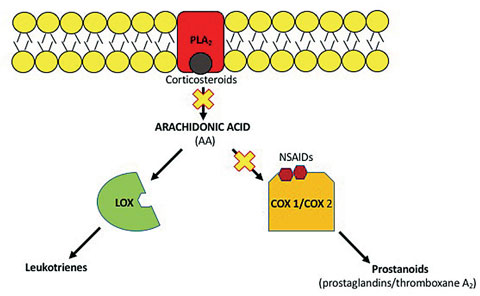 |
| Fig. 2. The two classes of anti-inflammatory drugs each inhibit the inflammatory pathway differently. |
Corticosteroids
The anti-inflammatory properties of corticosteroids are mediated at the genomic level.2 When corticosteroids bind to receptors in the cytoplasm, the bound complex migrates into the nucleus and upregulates the expression of anti-inflammatory proteins and downregulates the expression of proinflammatory proteins.2 More specifically, lipocortins, which are produced after corticosteroid cellular modulation, inhibit PLA2 and histamine synthesis in mast cells.3 Corticosteroids also have non-genomic modulating effects mediated by the corticosteroid-receptor binding, which include inhibition of vasodilation, vascular permeability, decrease in scar formation and the stabilization of intracellular and extracellular membranes.3 Because corticosteroids inhibit the inflammatory cascade earlier than NSAIDs, corticosteroids are more effective anti-inflammatory agents.4
In my personal clinical experience, employing topical corticosteroids for many inflammatory (i.e. uveitic, ocular surface, atopic) conditions has yielded positive results. Although the condition may not be completely resolved at the first follow up, I usually see a dramatic decrease in symptoms.
If there is no improvement in patient symptoms or clinical signs, an astute clinician should investigate medication compliance and instillation. As many of the corticosteroid options are in suspension form, failing to shake the bottle prior to instillation tends to be the culprit of delayed resolution. Additionally, the varying strengths of topical corticosteroids allow clinicians the option to increase the medication strength when little improvement is observed.
Indications and Contraindications. Many of the adnexal and anterior segment inflammatory conditions can be successfully treated with topical steroid therapy. Additionally, first-line therapy for post-op retinal cystoid macular edema (CME) is topical corticosteroids and NSAIDs.5 Indicated conditions for corticosteroid use include steroid-responsive inflammation of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe.6
Corticosteroids inhibit the resident immune system to employ its anti-inflammatory properties. This decrease in the immune system increases the risk for opportunistic infections. When you have concern for infectious etiologies, be cautious to initiate isolated corticosteroid therapy.
As red eye presentations can often be difficult cases, consider the following before initiating therapy:
- Take a thorough case history that may preclude corticosteroid use, at least initially
- Instill vital dyes that may reveal an epithelial herpetic etiology
Initiating Therapy. New drug designs have increased corticosteroid potency and metabolism with decreased side effects.7-10 Before initiating corticosteroid therapy, clinicians should consider the following baseline factors:
- Presence of an anterior chamber reaction (cell/flare)
- Significance of hyperemia/injection
- Symptomatology (photophobia, pain, irritation) level
- Targeted location of treatment
- Presumed diagnosis
Intraocular inflammation should be treated with maximal efficacy topical corticosteroids, which are formulated to increase penetration into the eye for more targeted anti-inflammatory action.4 Dermatologic, surface and corneal diagnoses, however, would benefit from lesser-strength corticosteroid options.4
Location-specific therapy decreases the risk of side effects and corticosteroid-related sequelae. Ocular hypertension and a hastening of posterior subcapsular cataract formation are well documented in the literature with prolonged corticosteroid use.2-4,8,9,11,12 Research suggests roughly 33% of the adult population are moderate responders—IOP increases by 6mm Hg to 15mm Hg.3 Alternatively, 4% to 6% of the general population are high responders—IOP elevates above 15mm Hg with therapy.3 Family history of glaucoma, diabetes, myopia and younger age are all risk factors that preclude responders to elevations in IOP.11,12
However, more often than not, the benefits of corticosteroid treatment outweigh the potential side effects. Additionally, visual acuity, IOP assessment and ophthalmoscopic evaluation of the optic nerve head at subsequent visits will ensure clinicians catch any possible side effects before they become vision threatening.
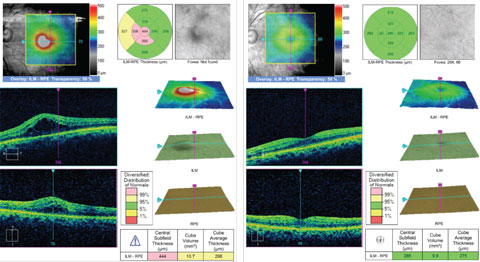 |
| The OCT on the left shows a patient with CME. This was successfully resolved with Durezol BID OS and Ilevro QD OS over a nine-week period, as seen in the right OCT. Click image to enlarge. |
Intraocular Inflammation. Intraocular inflammatory conditions require aggressive corticosteroid therapy to prevent vision-threatening complications and sequelae.4,7 An appropriate corticosteroid penetrates the ocular surface to inhibit inflammation at its source. Durezol (difluprednate 0.05%, Alcon) and Pred Forte (prednisolone acetate ophthalmic suspension 1%, Allergan) are corticosteroid options for endogenous inflammations of the eye.4
Approved by the FDA in 2008, Durezol—a synthetic difluorinated prednisolone derivative—is the most recent corticosteroid addition to the list of drugs approved for the treatment of postoperative inflammation and pain.7 In addition to difluorination and augmentation with butyrate, Durezol has increased corneal penetration due to the substitution of a hydroxyl group with acetate.7 This translates into topical dosing that is half of what is needed with Pred Forte, which may help improve patient compliance. A study that investigated Durezol dosed QID compared with prednisolone acetate 1% dosed at eight times a day found Durezol to be noninferior in a multicenter randomized double-masked trial.7 Additionally, Durezol is formulated as an emulsion, which does not require shaking prior to instillation. In one study, researchers found Durezol had superior dose uniformity compared with Pred Forte and its generic counterpart because of its emulsion formulation.13
Historically, Pred Forte has been the main option for intraocular inflammation treatment.7 Research shows Pred Forte has varying anti-inflammatory benefits compared with the generic formulation. In a comparative analysis of prednisolone acetate suspensions, one study found that Pred Forte exhibited greater homogeneity and bioavailability of the drug between doses compared with EconoPred (Alcon) and generic prednisolone acetate.14 Pred Forte and its generic counterparts are formulated in a suspension and require vigorous shaking before instillation.
Other maximally effective corticosteroids for treating intraocular inflammation include Vexol (rimexolone 1%, Alcon) and prednisolone phosophate sodium 1%.
Available in generic formulations, Vexol and prednisolone phosphate sodium provide for cost-conscientious prescribing. Additionally, research found Vexol had equal efficacy in the treatment of anterior uveitis with a decreased chance of increasing IOP compared with Pred Forte.15 This may be a good alternative for known steroid responders or glaucoma patients.
Ocular Surface. Retrometabolic drugs are currently used in the treatment of many systemic conditions.8 Such agents convert the inactive drug metabolite into a structurally modified analogue. After eliciting therapeutic benefit, the analog undergoes rapid degradation and metabolism, thereby decreasing the opportunity for adverse drug reactions.9 Because these synthetic analogs possess the same therapeutic benefit with decreased adverse drug reactions, they are known as soft drugs.8,9
Loteprednol etabonate, for example, is a cortienic acid-based derivative that exhibits highly lipophilic properties, reported to be 10 times greater than dexamethasone, allowing increased penetration into cell membranes.9,10,16 Researchers found C-labelled loteprednol etabonate 0.5% metabolites in highest concentration in the cornea of rabbit eyes and lower metabolite concentrations in the iris/ciliary body and aqueous humor, respectively, indicating a majority of loteprednol etabonate metabolism occurs in the cornea.17,18 Additionally, researchers increased penetration of loteprednol etabonate in the cornea and conjunctiva compared with the aqueous humor levels due to rapid hydrolysis.19
Lotemax (loteprednol etabonate 0.5%, Bausch + Lomb)—available as a gel or ointment—is a useful anti-inflammatory option to have in your arsenal. A 0.2% suspension formulation of loteprednol etabonate is available under the name Alrex, and a combination of loteprednol etabonate 0.5% and tobramycin 0.3% is branded as Zylet.
Both Lotemax 0.5% gel and ointment are FDA-approved for the treatment of postoperative inflammation and pain.20 Alrex is FDA-approved for the treatment of seasonal allergic conjunctivitis.21 Though the literature is divided on its efficacy in treating anterior uveitis compared with prednisolone, there is increased safety with loteprednol etabonate use, evidenced with a lower frequency of IOP elevation.3
Other conditions that respond well to lesser-strength (compared with Pred Forte and Durezol) corticosteroids include keratoconjunctivitis sicca, corneal-involving viral conjunctivitis, allergic and vernal conjunctivitis and to prevent scarring of the cornea.4
Alternative options for the treatment of ocular surface inflammation include FML (fluorometholone ophthalmic suspension 0.25%, 0.1%, Allergan) or prednisolone acetate 0.12%. These alternatives are available in generic formulations and are lower in cost compared with their branded counterparts.
 |
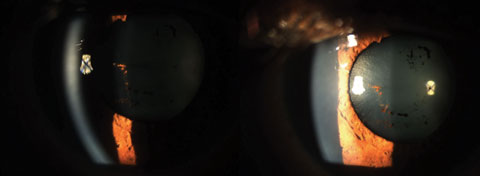 |
| The lower photo shows dramatic improvement over one week of the anterior uveitis (4+cell/4+flare) and posterior synechiae in this patient’s right eye (above) with Durezol six times a day and cyclopentolate BID. Click images to enlarge. Photos: Sulman Hans, OD |
Dermatological Inflammation of the Eyelids/Adnexa. Inflammation of the eyelids and adnexa are common presentations, and a detailed history and thorough ophthalmoscopic evaluation should yield whether it is allergic in nature or of infectious etiology. In cases of moderate to significant dermatologic inflammation, topical corticosteroids are warranted.
Dermatologic presentations can be treated with ointment formulations (an off-label use), such as Lotemax 0.5% and FML 0.1%, as they allow for increased contact time at the site of inflammation.4
If the inflammation is secondary to infectious etiologies, clinicians can use a combination corticosteroid/antibiotic ointment such as TobraDex (tobramycin 0.3%/dexamethasone 0.1%, Alcon), TobraDex ST (tobramycin 0.3%/dexamethasone 0.05%, Alcon), Maxitrol (neomycin 0.35%/dexamethasone 0.1%, Alcon) or Blephamide (sulfacetamide 10%/prednisolone acetate 0.2%, Allergan), to name a few. These provide good gram-positive coverage for the normal flora of the eyelids/skin. Additionally, consider sulfa allergies prior to prescribing some of these alternatives.
Ending Treatment. All corticosteroid therapy should be tapered to prevent any bouts of repeat inflammation. Unfortunately, tapering schedules are more of an art than a science. There is no universally accepted tapering regimen in the literature other than defined improvement by the Standard Uveitic Nomenclature (SUN) group.22 With improvement, and thereby response to the steroid regimen, the taper can be initiated. Ultimately, the clinician is responsible for changing dosing schedules and the start of the taper based on clinical improvement and intuition. When a clinician inevitability encounters a reoccurrence of inflammation because a steroid is tapered too quickly, initial dosing schedule of the steroid is required.
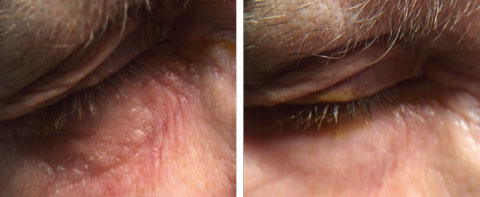 |
| This patient’s neomycin ointment hypersensitivity of the right eyelid (left) was resolved (right) with topical FML 0.1% ointment applied BID OU for four weeks. Click image to enlarge. |
NSAIDs
Topical nonsteroidal anti-inflammatory therapy is used sparingly in the primary care setting, mostly to prevent and treat postoperative inflammation. A vast majority of the NSAID options are FDA-approved for the treatment of pain and inflammation associated with cataract surgery.4
Additionally, NSAID therapy can be used to maintain mydriasis during cataract surgery and decrease pain in photorefractive keratectomy patients.18
Indications. In addition to the synergistic effects NSAID and corticosteroids have on postoperative CME formation and treatment, NSAID use primarily ameliorates pain on the ocular surface. Anecdotally, research suggests NSAIDs can be used temporarily to treat pain and irritation related to mechanical or surgical irritation of the conjunctiva and cornea, pre-post 5% betadine wash for viral epidemic keratoconjunctivitis or in combination with a strong corticosteroid for recalcitrant cases of uveitis.4 Most indications for NSAID use are off-label therapy options.
Generally, topical NSAIDs will be used in combination with maximal efficacy corticosteroids in the treatment of postoperative CME, typically in concert with the cataract surgeon.
Today’s NSAID options are dosed anywhere from QID to QD. Clinicians should follow the recommended dosing to limit risk for side effects related to prolonged NSAID use. As with corticosteroid use, duration of use should be limited (roughly one month). Chronic use retards corneal epithelial healing and corneal melting and perforation, although extremely rare, are reported in the literature.23
Newest to the topical NSAID drug market are Ilevro (nepafenac 0.3%, Alcon) and Prolensa (bromfenac 0.07%, Bausch + Lomb). Both Ilevro and Prolensa are dosed QD and are FDA-approved for postoperative inflammation and pain.24,25 Ilevro, a prodrug, is highly permeable to the cornea and is rapidly hydrolyzed to amfenac in the aqueous.26,27 Amfenac is a potent inhibitor of COX-1/COX-2 isoenzymes.27 Once-a-day dosing is possible due to the increased nepafenac concentration from 0.1% to 0.3%. Almost structurally identical to amfenac, Prolensa contains a bromine atom that makes Prolensa highly lipophilic, increasing corneal penetration and duration of action.27 As NSAIDs are intrinsically acidic, Prolensa has been buffered to a pH of 8.3 for additional comfort with instillation.27
An NSAID’s formulation is key to ensuring the patient is obtaining a therapeutic concentration of drug with each instillation. NSAID formulations in suspension require shaking to increase the drug’s bioavailability.
Other NSAID options available include: Acular LS and Acuvail (ketorolac tromethamine, Allergan), Bromsite (bromfenac ophthalmic solution, Sun Pharmaceuticals) and Voltaren (diclofenac sodium, Endo Pharmaceuticals). These options may be dictated by the patient’s insurance coverage or budget. They range in dosing from BID to QID and may sacrifice medication compliance, given the increase in dosing compared to their QD counterparts.
Advancement in drug delivery systems and drug formulations continue to equip eye care providers for successful treatment of many inflammatory conditions. The ever-changing field promises future advancements to help patients heal quicker and decrease vision-threatening conditions. With proper selection, evaluation and education, eye care providers have a large repertoire of treatment regimens for the successful treatment of their patients.
Dry Eye Disease |
| Topical anti-inflammatory drugs provide eye care providers a long-term treatment strategy for dry eye disease (DED) with almost no risk of systemic ADR when dosed as approved.28,29 Both Restasis (cyclosporin A 0.05% emulsion, Allergan) and Xiidra (lifitegrast 5% solution, Shire) act on T-cells in the tear film, conjunctiva and cornea to decrease tissue destruction and further inflammation related to DED.30-33 Restasis was FDA-approved in 2003 to increase the eye’s ability to produce tears in patients with inflammation related to keratoconjunctivitis sicca.34 Dosed BID, Restasis is a calcineurin inhibitor that prevents interlukin-2 (IL-2) formulation.30 IL-2 is secreted by T-helper cells and stimulates the proliferation of cytotoxic T-cells and additional T-helper cells. Restasis halts the propagation of additional T-cells, decreasing further damage to the ocular surface. Clinically, the effects of Restasis will not manifest immediately because of activated T-cells on the ocular surface prior to its use. To combat this, clinicians should consider using topical corticosteroids in conjunction with Restasis for a few weeks.32 The corticosteroid will help target the inflammation, while Restasis will maintain long-term anti-inflammatory effects. Xiidra, FDA-approved in 2016 to treat the signs and symptoms of dry eye, is the second and newest addition to topical immunomodulatory drugs for the treatment of dry eye.35 Xiidra also inhibits the T-cell mediated inflammatory pathway by preventing the recruitment and activation of T-cells to the ocular surface. Xiidra does this by blocking the adhesion of lymphocyte function-associated antigen-1 (LFA-1) to intracellular adhesion molecule-1 (ICAM-1).31 By blocking this interaction, T-cells do not migrate out of the blood vessel and decrease the interaction with antigen presenting cells.31 Furthermore, there is a decrease in cytokine release at inflammation sites. Formulated in a solution, Xiidra is dosed BID and is preservative-free. Future studies of this new agent will help to educate doctors on its clinical performance and role in the treatment regimen. A topical soft steroid such as Lotemax may also be considered at initiation of Xiidra, given the pre-existing inflammation likely present on the ocular surface. Having two therapeutic options available provides perceptive practitioners an alternative drug choice if a patient has failed on prior therapies, thus increasing the likelihood of success in the management of this complicated disease process. |
Dr. Higa graduated in 2014 from the Illinois College of Optometry and completed a one-year post-graduate residency in primary care at The Eye Institute. He currently is an instructor at Salus University-PCO where he works with interns and residents. He has a special interest in ocular surface disease and anterior segment inflammation. Dr. Higa has received honoraria from Allergan but has no direct financial disclosures for the products mentioned.
|
1. Ricciotti E, FitzGerald GA. Prostaglandins and inflammation. Arteriosclerosis, thrombosis, and Vascular Biology. 2011;31(5):986-1000. 2. Comstock TL, DeCory HH. Advances in corticosteroid therapy for ocular inflammation: loteprednol etabonate. International Journal of Inflammation. 2012 Mar 28;2012. 3. Sheppard JD, Comstock TL, Cavet ME. Impact of the topical ophthalmic corticosteroid loteprednol etabonate on intraocular pressure. Advances in Therapy. 2016;33(4):532-52. 4. Thomas R, Melton R. 2016 Clinical guide to ophthalmic drugs. Rev Optom. 2016;153(5): 20-5. 5. Yonekawa Y, Kim IK. Pseudophakic cystoid macular edema. Curr Opin Ophthalmol. 2012;23(1):26-32. 6. Allergan. Pred Forte package insert. Irvine, CA, 2013. 7. Jamal KN, Callanan DG. The role of difluprednate ophthalmic emulsion in clinical practice. Clinical Ophthalmology. 2009;3:381. 8. Bodor N, Buchwald P. Soft drug design: general principles and recent applications. Medicinal Research Reviews. 2000;20(1):58-101. 9. Bodor N, Buchwald P. Ophthalmic drug design based on the metabolic activity of the eye: soft drugs and chemical delivery systems. The AAPS Journal. 2005;7(4):E820-33. 10. Yellepeddi VK, Palakurthi S. Recent advances in topical ocular drug delivery. Journal of Ocular Pharmacology and Therapeutics. 2016;32(2):67-82. 11. Chang DF, Tan JJ, Tripodis Y. Risk factors for steroid response among cataract patients. J Cataract Refract Surg. 2011;37(4):675-81. 12. Kersey JP, Broadway DC. Corticosteroid-induced glaucoma: a review of the literature. Eye. 2006;20(4):407-16. 13. Stringer W, Bryant R. Dose uniformity of topical corticosteroid preparations: difluprednate ophthalmic emulsion 0.05% versus branded and generic prednisolone acetate ophthalmic suspension 1%. Clin Ophthalmol. 2010 Oct 5;4:1119-24. 14. Roberts CW, Nelson PL. Comparative analysis of prednisolone acetate suspensions. Journal of Ocular Pharmacology and Therapeutics. 2007;23(2):182-7. 15. Foster CS, Alter G, DeBarge LR, et al. Efficacy and safety of rimexolone 1% ophthalmic suspension vs 1% prednisolone acetate in the treatment of uveitis. Am J ophthalmol. 1996;122(2):171-82. 16. Alberth M, Wu WM, Winwood D, Bodor N. Lipophilicity, solubility and permeability of loteprednol etabonate: a novel, soft anti-inflammatory steroid. J Biopharm Sci. 1991;2(2):115-25. 17. Druzgala P, Wu WM, Bodor N. Ocular absorption and distribution of loteprednol etabonate, a soft steroid, in rabbit eyes. Current Eye Research. 1991;10(10):933-7. 18. Schalnus R. Topical nonsteroidal anti-inflammatory therapy in ophthalmology. Ophthalmologica. 2003;217(2):89-98. 19. Glogowski S, Proksch JW. Ocular pharmacokinetics of loteprednol etabonate following ocular administration of a novel ointment formulation or a suspension (Lotemax) in rabbits with corneal inflammation. Invest Ophthalmol Vis Sci. 2010;51(13):1980. 20. Bausch+Lomb. Lotemax package insert. Bridgewater, NJ, 2016. 21. Bausch+Lomb. Alrex package insert. Tampa, FL, 2013. 22. Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol. 2005;140(3):509-16. 23. Lin JC, Rapuano CJ, Laibson PR, et al. Corneal melting associated with use of topical nonsteroidal anti-inflammatory drugs after ocular surgery. Arch Ophthalmol. 2000;118(8):1129-32. 24. Alcon. Ilevro package insert. Ft. Worth, TX, 2013. 25. Bausch+Lomb. Prolensa package insert. Tampa, FL, 2013. 26. Chastain JE, Sanders ME, Curtis MA, et al. Distribution of topical ocular nepafenac and its active metabolite amfenac to the posterior segment of the eye. Experimental Eye Research. 2016 Apr;145:58-67. 27. Ahuja M, Dhake AS, Sharma SK, Majumdar DK. Topical ocular delivery of NSAIDs. The AAPS Journal. 2008;10(2):229-41. 28. Gire AI, Karakus S, Ingrodi SM, Akpek EK. Frequent dosing of topical cyclosporine A for severe ocular surface disease. Journal of Ocular Pharmacology and Therapeutics. 2016;32(3):150-4. 29. Donnenfeld ED, Karpecki PM, Majmudar PA, et al. Safety of Lifitegrast ophthalmic solution 5.0% in patients with dry eye disease: a 1-year, multicenter, randomized, placebo-controlled study. Cornea. 2016;35(6):741. 30. Matsuda S, Koyasu S. Mechanisms of action of cyclosporine. Immunopharmacology. 2000;47(2):119-25. 31. Perez VL, Pflugfelder SC, Zhang S, et al. Lifitegrast, a novel integrin antagonist for treatment of dry eye disease. The Ocular Surface. 2016;14(2):207-15. 32. Pflugfelder SC. Anti-inflammatory therapy for dry eye. Am J Ophthalmol. 2004;137:337-42. 33. Donnenfeld E, Pflugfelder SC. Topical ophthalmic cyclosporine: pharmacology and clinical uses. Surv Ophthalmol. 2009;54(3):321-38. 34. Allergan. Restasis package insert. Irvine, CA, 2013. 35. Shire. Xiidra package insert. Lexington, MA, 2016. |

