 Currently, more than 1.75 million Americans have advanced age-related macular degeneration (AMD).1 More alarmingly, however, that number is expected to increase to 2.95 million by the year 2020.1,2
Currently, more than 1.75 million Americans have advanced age-related macular degeneration (AMD).1 More alarmingly, however, that number is expected to increase to 2.95 million by the year 2020.1,2
AMD is a highly complex disease influenced by multiple factors, including age, genetic predisposition, environmental stimulants, and oxidation and inflammation.3
Unfortunately, most eye care clinicians are forced to wait until patients exhibit physical signs of macular damage before advising them how to most effectively prevent disease progression. By then, it may be too late to slow or halt the conversion to neovascular AMD.
What if we could identify those at high risk for progression earlier in the disease process and potentially block the cascade before the onset of significant visual damage? Encouraging advancements in genetic screening, as well as several new and emerging therapies for early AMD, may soon help us achieve this goal.
The Role of Oxidation
The pathogenesis of AMD still remains somewhat unclear; however, oxidative stress is believed to play a significant role in both disease development and progression. Oxidative stress refers to cellular damage caused by reactive oxygen intermediates (ROI), which are byproducts of oxygen metabolism.4
Because the retina consumes significant quantities of oxygen following visible light exposure, it is highly susceptible to oxidation. Additionally, the photoreceptor outer segments are rich in long-chain polyunsaturated fatty acids (PUFAs), which are readily oxidized by ultraviolet light exposure and cigarette smoke.
Macular pigments naturally protect the central retina from oxidative stress. These pigments are also believed to limit retinal damage by absorbing blue light and/or quenching ROIs.4 Further, retinal pigment epithelium (RPE) cells are responsible for phagocytosis of the photoreceptor outer segments.
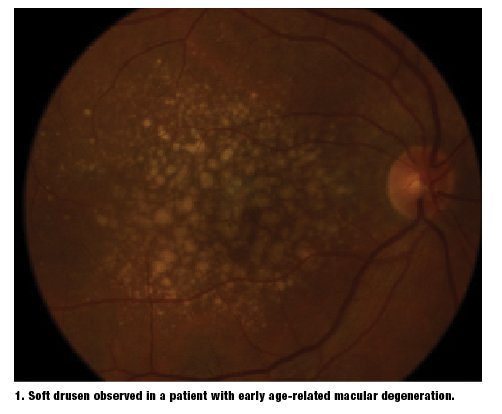
Oxidized PUFAs are not correctly cleaved from RPE lysosomes during phagocytosis. As a result, lipofuscin begins to accumulate and is deposited in Bruch’s membrane. Increased lipofuscin granules accelerate RPE cell death. Additionally, excess lipofuscin accumulation precipitates drusen formation–– the earliest clinical finding associated with AMD (figure 1).5
A key molecular component of lipofuscin is A2E––a phototoxic byproduct of the visual cycle that likely is an underlying cause of dry macular degeneration.6 When A2E is exposed to light, the RPE cells experience further damage, including free radical generation, drusen formation and activation of several complement factors associated with AMD development, such as complement factor H (CFH) and complement factor B. Fortunately, A2E can be detected with fundus autofluorescence (FAF). So, clinicians with access to FAF imaging devices may be able to identify patients at an increased risk for AMD before drusen formation is evident on clinical examination.7
Take note that patients with progressive dry AMD are at an elevated risk for geographic atrophy (GA) formation (figure 2). The build-up of lipofuscin causes RPE stretching and subsequent pigment release. Over time, further RPE cell degeneration and proliferation clinically manifest as areas of pigmentary change located near or around existing drusen. Eventually, the drusen and overlying pigment epithelium disappear, leaving a patch of geographic atrophy.
|
Vita Risk: Personalized Supplement Recommendations
The Age Related Eye Disease Study (AREDS) indicated that high-dose antioxidant supplementation plus zinc reduced the overall risk of progression to advanced AMD in patients with moderate to severe disease in one eye.19 A pharmacogenetic analysis of the AREDS dataset indicated that certain components of the formulation may be beneficial for some patients but actually harmful to others, depending on individual variations in the CFH and ARMS2 genes.20 The researchers estimated that 49% of the AREDS study population would have achieved a greater clinical benefit from nutrients not supplied in the AREDS formulation.20 Further, they determined that genotype-dictated supplement formulations would have more than doubled subjects’ rate of progression reduction.20 As this research has been the source of much debate among retina subspecialists, its clinical implications are not yet fully understood. Interestingly, Macula Risk PGx incorporates a metric called the Vita Risk Pharmacogenetic Result into its analysis; it is believed to identify the most appropriate nutritional supplement(s) for reducing the individual’s likelihood for AMD development and progression. This technology could potentially help clinicians customize nutritional supplement recommendations for patients who are genetically predisposed to AMD. |
Genetic Influences on AMD
Most common medical conditions—such as hypertension, type 2 diabetes and breast cancer—are complex, multifactorial diseases that result secondary to a combination of genetic components and environmental factors.8 The same can be said of age-related macular degeneration. Within the last decade, researchers have uncovered several gene-gene and gene-environment interactions that directly influence the pathogenesis, progression rate and potential severity of age-related macular degeneration.8,9
The clinical presentation of AMD is highly variable. For example, although drusen formation and RPE atrophy are considered hallmarks of the disease, their presence alone is not a reliable prognostic indicator.9 Large and soft drusen are known to be strong risk factors for the development of more advanced forms of AMD, including central GA and secondary choroidal neovascularization.9 Although these clinical findings are associated with a greater overall “risk,” not all patients experience comparable rates of disease development and progression. This is where genetic testing is becoming an invaluable source of diagnostic and prognostic information––it can potentially identify specific individuals who truly are at the highest risk for severe visual compromise.
Mutations of the CFH and ARMS2 genes have been definitively associated with an elevated risk of AMD development and progression.8 Further, several other genes, such as complement factors B, C2 and C3, could potentially be linked to macular degeneration.8 Fortunately, clinicians are now able to detect the presence of these gene variations via genetic testing.
Currently, there are two commercially available tests for AMD––Macula Risk PGx (ArcticDx) and RetnaGene (Sequenom Laboratories/Nicox). Both testing kits require the clinician to capture an in-office DNA sample via buccal swab.
Then, the sample is sent to a laboratory for analysis. Finally, the results are sent back to the ordering clinician in two to three weeks. The testing data can be instrumental in both patient education and risk assessment.
Dry AMD Therapies on the Horizon
Anti-VEGF therapies, effective though they may be, intervene late in the course of AMD development and are fraught with limitations (e.g., infection risk, high cost and the need for lifelong, ongoing therapy to maintain visual acuity gains).
Far better would be therapies that intervene upstream in the pathophysiology of AMD, before neovascular membrane formation. Below are snapshots of some of the most promising agents under study.
 Visual Cycle Modulation
Visual Cycle Modulation
The process of regenerating 11-cis-retinol during the visual cycle produces toxic byproducts, including A2E.7 As previously mentioned, when A2E is exposed to light, it generates phototoxic compounds that damage the RPE cell membrane, inhibit proper RPE lysosome activity, and promote the development of early AMD.
• Emixustat (formerly ACU-4429) is intended to modulate visual cycle overactivity in patients with degenerative retinal diseases by decreasing production of the toxic byproducts (including A2E) that promote early AMD development. Developed by Acucela, Emixustat is a once-daily oral tablet intended to treat clinical symptoms associated with dry AMD. Currently, the drug is in Phase IIb/III testing for the treatment of dry AMD and geographic atrophy.10
Neuroprotection
Photoreceptor death occurs when the cells are separated from the underlying RPE. The underlying cause of photoreceptor cell death secondary to retinal disease is complex; however, there are two widely cited processes: apoptosis and necrosis.11 Investigations of neuroprotective agents have not been fruitful to date, and current research suggests that the failure is due to a concentration on monotherapy––when, in fact, a multidrug approach likely is necessary.11
There are several neuroprotective therapeutic agents in clinical trials; however, the most promising agents include:
• Renexus (formerly NT-501), an intraocular implant developed by Neurotech, recently completed Phase II testing for the treatment of geographic atrophy secondary to dry AMD progression.12 The delivery module contains genetically modified cells that have been engineered to secrete ciliary neurotrophic factor and inhibit photoreceptor apoptosis. It is worth noting that Renexus currently is being evaluated for the treatment of several other visually devastating conditions, including retinitis pigmentosa, macular telangiectasia and achromatopsia.
• Brimonidine tartrate intravitreal implant (Allergan), the active agent in Alphagan, is a selective alpha 2-adrenoceptor agonist currently under investigation for dry AMD. Alpha-2 adrenergic receptor stimulation appears to inhibit signals that otherwise lead to retinal ganglion cell death in macular degeneration patients.13 The drug implant completed Phase II testing in March 2013.
• AL-8309B (tandospirone, Alcon) is a selective serotonin 1A agonist that has demonstrated neuroprotive effects in animal testing. Studies have shown that tandospirone may protect both RPE and photoreceptors cells from severe photo-oxidative stress.14 A Phase III clinical trial, the Geographic Atrophy Treatment Evaluation (GATE), completed recruitment in 2013––although, results have not yet been made public.
|
Topical Anti-inflammatory Therapy
During the last decade, we’ve learned that inflammation plays an important role in the pathogenesis of macular degeneration. Therefore, it seems reasonable to suspect that the anti-inflammatory effects of topical steroids and NSAIDs could potentially slow or halt AMD progression. Corticosteroids were one of the first anti-inflammatory drug classes evaluated for the treatment of choroidal neovascular membrane formation in neovascular AMD. When combined with anti-VEGF agents and photodynamic therapy, both triamcinolone and dexamethasone have been helpful as adjunctive treatments––however, neither of these topical steroidal agents have been successful in retarding dry AMD progression when used alone. NSAIDs now demonstrate better potency, clinical efficacy and corneal penetration than ever before. During the last decade, several research groups have evaluated the effect of topical NSAIDs on a variety of inflammatory and neovascular diseases of the retina and choroid, including cystoid macular edema, diabetic retinopathy and advanced AMD.21 With regard to wet AMD, topical bromfenac, diclofenac and nepafenac have been used to slow the progression of neovascularization in several monotherapy trials; however, the results generally have been underwhelming.21 Currently, an ongoing Phase II clinical trial is evaluating the therapeutic efficacy of combination intravitreal ranibizimab injection and topical bromfenac for wet AMD patients.17 |
There is substantial published evidence of an association between AMD and a multitude of key proteins in the complement cascade. For example, patients with one specific variation of the CFH gene are nearly five times more likely to develop AMD than those without the mutation.15
Emerging complement inhibitors are promising and may play a role in both the prevention and management of AMD in the near future. Currently, several agents in development target various components of the complement cascade. One such drug is being tested off-label for the treatment of dry AMD:
• Eculizumab (Soliris, Alexion Pharmaceuticals) is the only FDA-approved monoclonal antibody (against complement C5) that targets the complement system. (The drug currently is indicated for the treatment of a rare disease called paroxysmal nocturnal hemoglobinuria, and is dosed intravenously at 900mg every two weeks.15)
The COMPLETE (Complement Inhibition of Systemic Eculizumab for the Treatment of Non-Exudative Age-Related Macular Degeneration) study at Bascom Palmer Eye Institute in Miami was the first trial designed to evaluate the clinical efficacy of systemic complement inhibition for the treatment of dry AMD.15 The COMPLETE researchers enrolled 30 dry AMD patients, of which 20 received oral eculizumab. Unfortunately, those treated with eculizumab did not exhibit fewer drusen or a lower incidence of GA than patients in the control group. However, none of the patients in the treatment group progressed to wet AMD during the 12-month trial period, compared to 20% of controls.15
The researchers concluded that systemic complement inhibition may not be a successful strategy for slowing short-term GA progression. However, they suggested that intravitreal eculizumab administration may yield more promising results.15
As an aside, an ongoing Phase II trial currently is evaluating the effect of intravenous eculizumab on dry AMD and GA progression.16,17
Inflammation Suppression
It is well known that many retinal conditions are inflammatory in nature, including macular degeneration. Because corticosteroids are widely used in the management of ocular inflammation, intravitreal steroidal treatment likely holds great promise in curtailing dry AMD progression.
• Iluvien (Alimera Sciences) is a sustained-release intravitreal implant that contains 180ug flucinolone acetonide. The device is implanted into the vitreous cavity via a 25-gauge intravitreal injector. Flucinolone suppresses VEGF, and chronic intravitreal infusion has been shown to preserve retinal structure and function.18 Iluvien currently is being evaluated for the treatment of dry AMD and GA in a Phase III study, and final data collection is anticipated in December 2014.
We have recently entered into an era of amazing biotechnology––one which will see the development and expansion of individualized medicine. In the short-term, optometrists will make the largest contribution via genetic screening for retinal disease. In not-too-distant future, however, ODs likely will become more directly involved in the therapeutic management of certain retinal diseases––particularly if an oral or topical medication secures FDA approval for the treatment of dry AMD.
Dr. Haynie is the executive clinical director at Retina & Macula Specialists, with office locations in Tacoma, Renton, Olympia and Kennewick, Wash., and is a member of Thrombogenics’ advisory board. He has no direct financial interests in any of the products mentioned.
1. Friedman DS, O’Colmain BJ, Munoz B, et al. Prevelance of age-related macular degeneration in the United States. Arch Ophthalmol. 2004 Apr;122(4):564-72.
2. Klein R, Klein BE, Knudston MD, et al. Fifteen-year cumulative incidence of age-related macular degeneration: the Beaver Dam Eye Study. Ophthalmology. 2007 Feb;114(2):253-62.
3. Klein R, Cruickshanks KJ, Nash SD, et al. The prevelance of age-related macular degeneration and associated risk factors. Arch Ophthalmol. 2010 Jun;128(6):750-8.
4. Beatty S, Koh H, Phil M, et al. The role of oxidative stress in the pathogeneisis of age-related macular degeneration. Surv Opthalmol. 2000 Sept-Oct;45(2):115-34.
5. Janik-Papis K, Ulińska M, Krzyzanowska A, et al. Role of oxidative mechanisms in the pathogenesis of age-related macular degeneration. Klin Oczna. 2009;111(4-6):168-73.
6. Pauleikhoff D, van Kuijk FJ, Bird AC. Macular pigment and age-related macular degeneration. Ophthalmologe. 2001 Jun;98(6):511-9.
7. Ben-Shabet S, Parish CA, Vollmer HR, et al. Biosynthetic studies of A2e, a major fluorophore of retinal pigment epithelial lipofuscin. J Biol Chem. 2002 Mar 1;277(9):7183-90.
8. Swaroop A, Branham KE, Chen W, Abecasis G. Genetic susceptibility to age-related macular degeneration: a paradigm for dissecting complex disease traits. Hum Mol Genet. 2007 Oct 15;16 Spec No. 2:R174-82.
9. Klein R, Klein BE, Jensen SC. The five-year incidence and progression of age-related maculopathy: the Beaver Dam Eye Study. Ophthalmology. 1997 Jan;104(1):7-21.
10. Leung E, Landa G. Update on current and future novel therapies for dry age-related macular degeneration. Expert Rev Clin Pharmacol. 2013 Sep;6(5):565-79
11. Demetrios J, Vavvas G. Neuroprotection in age-related macular degeneration. Retina Today. 2012 Mar;64-5.
12. Tao W. A study of an encapsulated cell technology (ECT) implant for patients with atrophic macular degeneration. ClinicalTrials.gov identifier: NCT00447954. Available at: www.clinicaltrials.gov/ct2/show/NCT00447954?term=NCT00447954&rank=1. Accessed May 27, 2014.
13. Aung T, Oen FT, Wong HT, et al. Randomized control trial comparing the effect of brimonidine and timolol on visual field loss after acute primary angle closure. Br J Ophthalmol. 2004 Jan;88(1):88-94.
14. Collier RJ, Wang Y, Smith SS, et al. Complement deposition and microglial activation in the outer retina in light-induced retinopathy: Inhibition by a 5-HT1A agonist. Invest Ophthalmol Vis Sci. 2011 Oct 11;52(11):8108-16.
15. Rosenfeld PJ. Complement inhibition with eculizumab for the treatment of non-exudative macular degeneration (COMPLETE). ClinicalTrials.gov identifier: NCT00935883. Available at www.clinicaltrial.gov/ct2/show/NCT00935883. Accessed May 12, 2014.
16. Klein RJ, Zeiss C, Chew EY, et al. Complement factor H polymorphism in age-related macular degeneration. Science. 2005 Apr 15;308(5720):385-9.
17. Duker J. The COMPLETE trial for dry AMD: Results. Rev Ophthalmol. 2012 Sept;19(9):56-8.
18. Roe R, Boyer D. The pipeline for dry AMD: Novel approaches include antioxidant therapy, protecting photoreceptors and RPE and suppressing inflammation. Rev Ophthamol. 2011 Dec;18(12):41-6.
19. Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, betacarotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001 Oct;119(10):1417-36.
20. Awh CC, Lane AM, Hawken S, et al. CFH and ARMS2 genetic polymorphisms predict response to antioxidants and zinc in patients with age-related macular degeneration. Ophthalmology. 2013 Nov;120(11):2317-23.
21. Francis PJ. Combination ranibizumab and bromfenac for neovascular age-related macular degeneration. ClinicalTrials.gov identifier: NCT00805233. Available at: www.clinicaltrial.gov/ct2/show/NCT00805233. Accessed May 12, 2014.

