An estimated 25% of patients in general eye clinics report symptoms of dry eye syndrome (DES).1 More than nine million Americans suffer from a moderate to severe form of it. Management of the condition costs United States health care $55.4 billion annually.2-4 Given the breadth of the condition’s impact, the goals of clinicians, researchers, policymakers and industry align: provide relief when needed, and if possible, a lasting cure. This article recaps many successful modalities currently in use by practitioners and looks just over the horizon to some emerging options anticipated for the near future.
Artificial Tears and Ointments
Initial treatment for DES includes artificial tears and ointments. Preservative-free formulations are recommended, especially if dosing more than four times per day is necessary. Preservatives, particularly benzalkonium chloride, are considered cytotoxic to ocular surface epithelia—including conjunctival goblet cells. Phosphate-containing eye drops should be used cautiously as they can promote corneal calcifications especially in the cases of corneal epithelial defects (e.g., corneal erosions or ulcers).9 Artificial tears are simple in composition and have a low residue time on the ocular surface, limiting their efficacy.
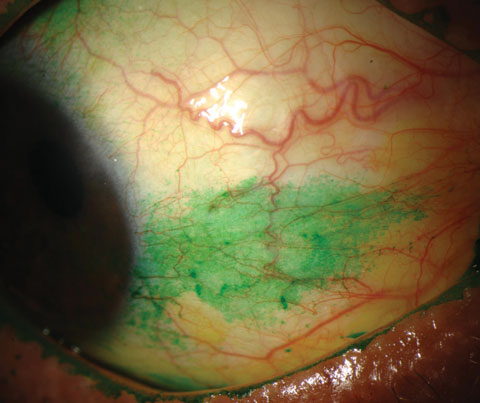 |
| Conjunctival staining with lissamine green in a dry eye patient. Photo: Michelle Hessen, OD. |
Emulsions containing liquid lipids are also available, and aim to stabilize and better reproduce the tear film. These take the form of charged or uncharged microemulsions and liposomes. Charged microemulsions, such as the cationic Cationorm (carboxymethylcellulose sodium 0.5% ophthalmic solution, Santen Pharmaceutical)—marketed in the United States as Retaine MGD (Ocusoft)—and the anionic Systane Balance (propylene glycol 0.6% ophthalmic solution, Alcon), show improvements in symptoms and corneal staining when compared with aqueous eye drops.10,11 In a study, 79 patients were randomized to four drops a day of either Cationorm or a polyvinyl alcohol/povidone eye drop.12 At 28 days, both treatment arms showed improvement in symptoms, and tear-film stability—indicated by tear break-up time (TBUT)—relative to baseline, but Cationorm demonstrated significant improvement in TBUT compared with the aqueous eye drops.10 Similarly, a trial with 49 participants receiving Systane Balance or saline eye drops for 28 days showed a significantly higher improvement in TBUT with Systane Balance.11 A liposomal eye spray, Tears Again Advanced Eyelid Spray (OcuSoft), showed greater improvement in inflammation of the lid margin and TBUT compared with hyaluronate artificial tears at four and 12 weeks in a trial of 216 patients.13
Uncharged microemulsions like Refresh Optive Advanced (carboxymethylcellulose sodium 0.5%, glycerin 1.0%, polysorbate 80 0.5%, Allergan) contain castor oil as the main lipid component. Treating 191 patients with lipid emulsion eye drops resulted in improvement in symptoms, Ocular Surface Disease Index (OSDI) score and TBUT when compared with baseline at 90 days in patients with mild to moderate DES.14 The research identified no treatment arm with aqueous tear drops. However, another study that treated 208 patients with either aqueous tear drops (carboxymethylcellulose sodium 0.5%, glycerin 0.9%) or lipid microemulsion eye drops (Refresh Optive Advanced) shows improvement in OSDI and TBUT at 30 days when compared with baseline, and shows no difference between the aqueous or lipid emulsion eye drops.15
| Defining DES Recently, the Definition and Classification Subcommittee of the International Dry Eye Workshop redefined DES as “a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability with potential damage to the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface.”5 The etiopathogenetic classification suggested by the Dry Eye Workshop distinguishes two main types of dry eye: an aqueous deficiency state and an evaporative state. |
Artificial tears and emulsions are not as effective as ointments containing semisolid lipids, which—though highly effective—cause blurred vision and have limited use during the daytime. Researchers have created a nanoscale dispersed eye ointment (NDEO) that retains the advantages of ointment in liquid form, and could be used as a drug delivery system for lipophilic drugs. In mice, NDEO treatment reduces corneal fluorescein staining and increases TBUT at day three compared with baseline and a polymer-based artificial tear. Improvement of TBUT started earlier (day three) with NDEO and lasted longer—until day 14—compared with polymer-based artificial tears.16
Reducing Tear Drainage
Minimizing tear drainage via placement of punctal plugs or thermal cauterization may contribute to the healing of the ocular surface by extending available lubrication provided by natural and artificial tears. Even punctal plugs alone may improve tear film stability and elongate the TBUT without the need for repeated administration of artificial tears; however, recurrent loss of punctal plugs can be problematic, often limiting the effectiveness of this treatment.17 Ocular surface inflammation should be addressed with topical medications prior to considering punctal occlusion.
Therapeutic Contact Lenses
Silicone hydrogel soft contact lenses provide high oxygen permeability, making the extended wear modality an acceptable option for assisting with ocular surface repair. Therapeutic bandage contact lenses provide corneal protection from the eyelids’ mechanical friction, as well as exposure to the external environment and evaporation of the tear film. Rigid gas permeable scleral lenses create a tear reservoir for constant lubrication of the corneal surface while maintaining necessary oxygen supply. Sclerals also mask surface irregularities, which also contribute to improvement in vision. Results of a retrospective chart review of 49 consecutive patients with ocular surface disease at the setting of various systemic conditions, including Sjögren’s syndrome (SS), showed improvement in visual acuity, healing of epithelial defects, reduction in photophobia and improvement in quality of life from scleral lens use.18
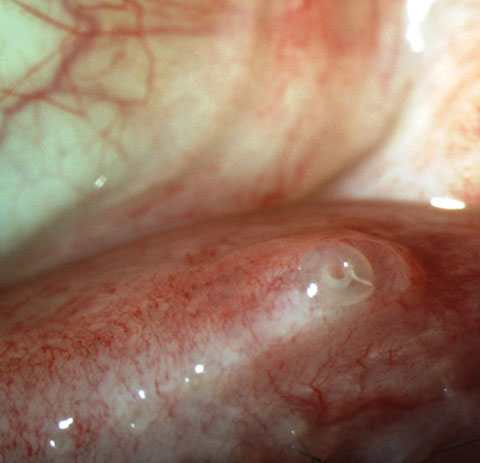 |
| Punctal plugs help to increase contact time between the tears and ocular surface, but inflammation must be addressed before considering this option. Photo: Alan G. Kabat, OD. |
Secretagogues
Acetylcholine, acting through muscarinic receptors, controls exocrine gland secretion, including lacrimation. Cevimeline and pilocarpine are oral cholinergic agonists that can be used off-label to treat DES. In randomized, prospective, double-blinded trials, oral cevimeline treatment show improvements in tear biometrics (as measured by Schirmer testing, rose bengal and fluorescein staining) and TBUT, as well as subjective patient symptom reporting.19-21 In a multicenter, placebo-controlled, fixed-dose trial, 373 patients with primary or secondary Sjögren’s syndrome and clinically significant dry eyes were randomized to receive either pilocarpine or placebo tablets for 12 weeks.22 Those receiving 20mg pilocarpine per day experienced significant improvement in dry eye symptoms compared with the placebo group. The drawback—side effects like nausea, abdominal pain, excessive sweating, headache, dizziness and cardiac arrhythmias—limit the use of these drugs.19,21,22
Improving Meibum
Oral doxycycline, minocycline and azithromycin are effective in treating DES caused by meibomian gland dysfunction (MGD), likely due to a combination of their ability to reduce inflammation in addition to their role as antibiotics.23 In a comparative study, a five-day course of oral azithromycin suggested it is more effective than a one-month course of doxycycline in relieving the signs and symptoms of MGD, while causing significantly fewer side effects.24 Topical azithromycin is also effective in treating evaporative DES and improving the quality of meibomian gland secretions.25
Procedures such as Lipiflow and intense pulsed light therapy are also capable of reducing tear evaporation by improving meibum secretions.26
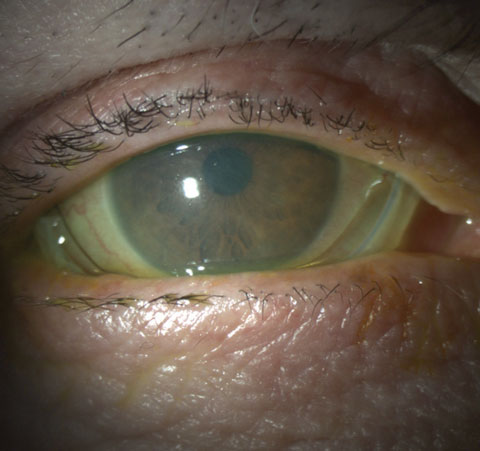 |
| Prokera plus in a patient with severe keratoconjunctivitis sicca. Photo: Alan G. Kabat, OD. |
Anti-inflammatory Therapy
It is widely recognized that inflammation plays a significant role in the pathogenesis of dry eye, promoting ocular surface disruption and symptoms of irritation. A number of anti-inflammatory treatments are currently used to treat DES. These may prove exceptionally useful in your treatment of the condition.27
• Topical NSAIDs. These versatile agents are used in the management of many ocular conditions, including allergic conjunctivitis, postoperative ocular pain, cystoid macular edema status post-cataract surgery and DES. NSAIDs treat inflammation by inhibiting different forms of the cyclooxygenase enzyme, thus reducing prostaglandin production—the mediators of inflammation.28
Short-term use of NSAIDs can be useful in ameliorating symptoms of ocular discomfort in dry eye; however, they should be used with caution and under close monitoring, and treatment discontinued if the corneal epithelium becomes damaged. NSAIDs—specifically diclofenac—have been shown to reduce corneal sensitivity.29 This may cause an additional insult to the disrupted epithelium in DES patients. In the literature, several cases of corneal melt associated with topical NSAID use—including diclofenac, ketorolac, nepafenac and bromfenac—show preexisting epitheliopathy.30-32 Though the exact relationship between corneal melt and topical NSAID use is still not clear, various suggested mechanisms include activation of matrix metalloproteinases, impairment of wound healing and neurotrophic effect resulting from the analgesic action of these drugs.33
• Topical Corticosteroids. More potent than NSAIDs, these agents help reduce ocular inflammation through several mechanisms of action.34 Topical administration of methylprednisolone 1% ophthalmic solution for several weeks provides moderate to complete relief of DES symptoms and reduces corneal fluorescein staining in patients with SS-related DES, suggests research.35 Pulse treatment with methylprednisolone for two weeks followed by a taper led to improvement in symptoms starting at two weeks, followed by improved TBUT and Schirmer test scores by the end of taper. After the first pulse treatment, mean drug-free remission time was 56.6 weeks; after the second, it increased to 72.4 weeks. No serious complications, including IOP elevation and cataract formation, occurred during the entire follow-up period.36
Loteprednol etabonate (Lotemax, Bausch + Lomb) is a safer topical corticosteroid designed to rapidly break down into inactive metabolites to ensure that it does not circulate systemically at detectable levels. This negates any significant effect on IOP.37 Recent research following 133 patients who were treated with either loteprednol etabonate 0.1% with hyaluronate artificial tears or fluorometholone 0.1% with hyaluronate artificial tears for two years showed significant improvement in symptoms, Schirmer test scores and fluorescein staining compared with baseline after six months in both groups. The research also showed TBUT improvement in both groups compared with baseline at 12 months. Neither group displayed significant elevation in average IOP at any point in the study.38
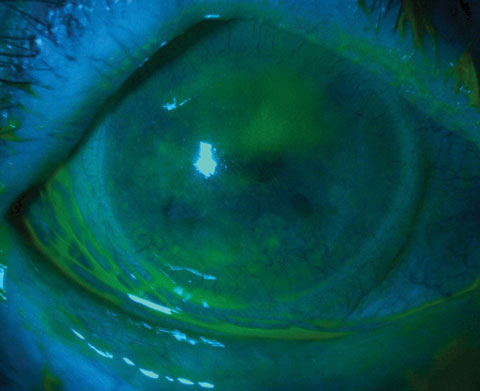 |
| This patient with severe keratoconjunctivitis sicca was ultimately treated with topical cyclosporin, autologous serum and amniotic membrane therapy. Photo: Alan G. Kabat, OD. |
Topical corticosteroids are also commonly used in combination with Restasis to reduce side effects and improve response to therapy by reducing inflammatory mediators. Patients treated with topical corticosteroids should be monitored closely for known cataract formation risks—glaucoma, corneal thinning and infectious keratitis.39
• Sutureless Amniotic Membrane. In addition to physically protecting the corneal epithelium, amniotic membrane grafts hydrate and oxygenate the regenerating tissue, promote epithelial healing, reduce inflammation and display anti-angiogenic effects.40 AmbioDisk (IOP Ophthalmics) is a dehydrated, sutureless amniotic membrane graft with no support structure; it is generally applied underneath a bandage contact lens. ProKera (Biotissue) is a sutureless biological bandage consisting of a cryopreserved amniotic membrane attached to a stabilizing polycarbonate ring; it can be used in cases of severe DES to promote healing and reduce inflammation.41
• Autologous Serum. The patient’s own serum contains several anti-inflammatory factors that ultimately inhibit the ocular surface inflammatory cascade responsible for DES. Clinical trials show autologous serum drops improve ocular irritation symptoms, and improve conjunctival and corneal dye staining seen in SS.42-44 Conversely, greater risk of microbial growth is associated with autologous serum drops, which contain high protein content and are generally nonpreserved.45
• Nutritional Supplements. Omega-3 fatty acids are known to have anti-inflammatory effects.46 Recent reports find that oral omega-3 supplementation shows promise in DES treatment.47-49 One trial randomized 259 patients to receive 1g/day of omega-3s or placebo for three months. At the end of this period, the group treated with omega-3s showed significant improvement in symptoms and TBUT.48 In other research, 496 female contact lens users who complained of dry eyes received 600mg/day of omega-3s or a placebo for six months. The research suggests that the omega-3 treatment improves symptoms, lens comfort levels and TBUT.47 A third trial randomized 105 patients with DES to receive 2g/day of omega-3s or placebo for three months. Once again, omega-3 supplementation resulted in improved TBUT, OSDI and tear osmolarity compared with placebo.49
Research suggests topical omega- fatty acids are beneficial in DES. Mice treated with 0.02% omega-3, 0.2% omega-3, hyaluronic acid (HA) or a mixture of HA and omega-3s. The 0.2% omega-3s alone and those treated with 0.2% omega-3s and HA demonstrated significantly improved corneal irregularity scores. Proinflammatory cytokine levels were reduced much more in the group treated with the omega-3s/HA mixture.50
• Cyclosporin A. Topical cyclosporin A (CsA) is frequently used to treat various inflammatory ocular surface disorders.51 It reduces inflammation by inhibiting T-cell activation and downregulating inflammatory cytokines in the conjunctiva and lacrimal gland. Research suggests the reduction in inflammation via these mechanisms enhances tear production.52-56 Topical cyclosporine also increases goblet cell density and decreases epithelial cell apoptosis.57
| Barriers to Topical Treatments The tear film is the first barrier to the absorption of topical drugs, acting to dilute drug concentration, rapidly clear drugs due to renewal of tear film and blinking reflex, rapidly drain drugs through the nasolacrimal ducts, and inhibit penetration through native barriers such as the ocular surface mucin layer. The tear film is comprised of an outer lipid layer, reducing the evaporation of tears, an intermediate aqueous layer and an inner layer called the glycocalyx, which consists of lysozymes and negatively-charged cell surface mucins.6,7 Once these are overcome, the cornea presents another barrier to drug absorption due to its highly organized, hydrophobic epithelium and its hydrophilic corneal stroma.8 |
Lipids are used as drug delivery systems to deliver CsA to the ocular surface and combat the inflammatory component of DES. Lipids have the advantage of increasing corneal contact time, especially in the case of cationic lipids, which are electrostatically attracted to the mucin layer of the tear film.58 Restasis (cyclosporine 0.05% ophthalmic emulsion, Allergan) is a well-known example of an anionic microemulsion CsA delivery vehicle. Recently, a cationic microemulsion, Novasorb (Novagali), was shown to deliver three to four times more CsA to rabbit corneas.58 Novasorb containing 1mg/mL CsA was approved by the European Union under the brand name Ikervis (Santen) and is undergoing Phase III trials in the United States.
In addition to emulsions, other carriers are being investigated as drug delivery systems. Nano-capsules comprised of an oily core surrounded by a polymer shell have been used as delivery systems for CsA. Research shows the nanocapsules delivered a concentration of CsA to rabbit corneas five times greater than with an oily solution of CsA. Corneal CsA levels remained higher for 72 hours following treatment.59 Yet another nanocapsule delivery system delivered CsA to the cornea at a 10 to 15 times higher concentration than CsA in castor oil, and remained higher for 24 hours.60
A CsA-nanocapsule formulation delivered CsA to the cornea at therapeutic concentrations but with less effect on ocular comfort profile.61 Cationic chitosan nanoparticles loaded with CsA targeted therapeutic doses to the cornea and conjunctiva in rabbits, while avoiding delivery to the inner ocular structures, plasma and blood.62
Research suggests polymeric micelles—self-assembling nanocarriers comprised of amphiphilic polymers—are good carriers for CsA delivery to the cornea, leading to a 28-fold increase in CsA levels when compared with an oily solution of CsA.63 Treatment with CsA-carrying liposomes led to improved amounts of CsA in the tear film, increased tear formation and lower irritation in rabbit eyes.64 All these substances show promise for efficient delivery of topical CsA to the cornea.
• Lifitegrast. A novel integrin antagonist that prevents the binding of lymphocyte function-associated antigen-1 to the intercellular adhesion molecule-1 expressed on inflamed epithelium, lifitegrast disrupts T-cell mediated inflammation seen in DES by preventing T-cell adhesion, migration, activation and cytokine release.65
In the first Phase III trial of this agent (OPUS-1), 588 patients with DES received topical lifitegrast 0.5% or placebo twice daily for 84 days. The study met the primary objective endpoint, but not the subjective one. The lifitegrast arm shows significantly improved inferior corneal staining score, but did not show a significant improvement in the visual function subscale score of OSDI, even though patients reported significant improvements in eye dryness and ocular discomfort.65
The study design in its next Phase III trial, OPUS-2, was similar to OPUS-1 but was modified to require a minimal level of symptom severity at baseline, and the primary symptom endpoint was changed to eye dryness. Thus, OPUS-2 enrolled patients with moderate to severe DES, while OPUS-1 enrolled patients with mild to moderate DES. This study met its primary symptomatic endpoint but failed to meet its primary objective endpoint, since there was no difference in improvement in inferior corneal staining between the two groups.66
Lifitegrast recently concluded a third Phase III efficacy and safety study called OPUS-3, and is now under evaluation by the FDA. The OPUS-3 study, conducted on 711 patients, reveals that lifitegrast significantly improves the symptoms of DES when compared with placebo starting from day 14 and retains improvement until day 84.67
Lacrimal Gland Therapy
Researchers explored transcytosis as a targeted-delivery option from the serum to the lacrimal gland lumen in rabbits and mice. Elastin-like protein polymers were genetically engineered and successfully targeted—transcytosed—to lacrimal glands via the coxsackievirus and adenovirus receptor, a receptor expressed at one of the highest levels in the body by the lacrimal gland.68 These nanoparticles can be fused with proteins with drug-binding abilities.69 This is an attractive approach, as it can reduce systemic side effects of the medications and increase treatment compliance and efficacy by creating a drug reservoir in the lacrimal gland.
Gene Therapy
Once the barriers to getting the drug to the ocular surface are overcome, gene therapy can be a viable option to treat dry eye, and delivery systems are crucial to accomplishing the feat. Using well-tolerated nanoparticles, researchers loaded genes coding for MUC5AC—a glycoprotein that plays a key role in tear homeostasis, and which is implicated in several ocular diseases—onto them. Researchers successfully increased expression of MUC5AC, which reduced CD4+ T-cell density, increased goblet cell density, restored tear production and normalized the mucin layer in the inflamed eye.70
Conclusion
DES is a common disorder that causes significant discomfort. Treatments aim to replenish and stabilize the tear film and reduce inflammation. Clinicians and patients already have experienced and/or can expect advances on many fronts ranging from improved artificial tears and ointments, light therapy and topical gene therapy via eye drops to restore the mucin layer of the tear film. Targeting inflammation has led to the use of omega-3 fatty acids, ever-improving carrier systems for the delivery of CsA and novel anti-inflammatory compounds.
Treatments comparable to sci-fi are in the offing—soon to be mainstream. With these exciting advancements, we can look forward to the relief they will bring to our patients, and ultimately, millions of dry eye suffers. And, in the meantime, we can use what we have—which themselves are wonderful treatments—to elicit the best outcomes possible for our patients. n
Mr. Vahedi is a medical student at Johns Hopkins School of Medicine in Baltimore.
Dr. Hessen is a clinical instructor at the Wilmer Eye Institute’s Ocular Surface Diseases and Dry Eye Clinic at Johns Hopkins School of Medicine, where she specializes in ocular surface disease, including autoimmune disorders.
|
1. Doughty MJ, Fonn D, Richter D, et al. A patient questionnaire approach to estimating the prevalence of dry eye symptoms in patients presenting to optometric practices across Canada. Optometry & Vision Science 1997;74(8):624-31. 2. Schaumberg DA, Sullivan DA, Buring JE, et al. Prevalence of dry eye syndrome among US women. American journal of ophthalmology 2003;136(2):318-26. 3. Schaumberg DA, Sullivan DA, Dana MR. Epidemiology of dry eye syndrome. Lacrimal Gland, Tear Film, and Dry Eye Syndromes 3: Springer; 2002:989-98. 4. Yu J, Asche CV, Fairchild CJ. The economic burden of dry eye disease in the United States: a decision tree analysis. Cornea 2011;30(4):379-87. 5. Lemp MA, Foulks GN. The definition and classification of dry eye disease. Ocul Surf 2007;5(2):75-92. 6. Nichols B, Chiappino M, Dawson C. Demonstration of the mucous layer of the tear film by electron microscopy. Investigative ophthalmology & visual science 1985;26(4):464-73. 7. Yañez-Soto B, Mannis MJ, Schwab IR, et al. Interfacial phenomena and the ocular surface. The ocular surface 2014;12(3):178-201. 8. Wilson CG, Tan L. Nanostructures Overcoming the Ocular Barrier: Physiological Considerations and Mechanistic Issues. Nanostructures Overcoming Ocul Barrier Physiol Considerations Mech Issues 2012:173-89. 9. Bernauer W, Thiel M, Kurrer M, et al. Corneal calcification following intensified treatment with sodium hyaluronate artificial tears. British journal of ophthalmology 2006;90(3):285-8. 10. Amrane M, Creuzot-Garcher C, Robert P-Y, et al. Ocular tolerability and efficacy of a cationic emulsion in patients with mild to moderate dry eye disease–A randomised comparative study. Journal Français d’Ophtalmologie 2014;37(8):589-98. 11. Aguilar AJ, Marquez MI, Albera PA, et al. Effects of systane® Balance on noninvasive tear film break-up time in patients with lipid-deficient dry eye. Clinical ophthalmology (Auckland, NZ) 2014;8:2365. 12. Ousler G, Devries DK, Karpecki PM, et al. An evaluation of Retaine™ ophthalmic emulsion in the management of tear film stability and ocular surface staining in patients diagnosed with dry eye. Clinical Ophthalmology (Auckland, NZ) 2015;9:235-43. 13. Khaireddin R, Schmidt K. [Comparative investigation of treatments for evaporative dry eye]. Klinische Monatsblatter fur Augenheilkunde 2010;227(2):128-34. 14. Simmons PA, Carlisle-Wilcox C, Chen R, et al. Efficacy, Safety, and Acceptability of a Lipid-Based Artificial Tear Formulation: A Randomized, Controlled, Multicenter Clinical Trial. Clinical therapeutics 2015;37(4):858-68. 15. Simmons PA, Carlisle-Wilcox C, Vehige JG. Comparison of novel lipid-based eye drops with aqueous eye drops for dry eye: a multicenter, randomized controlled trial. Clinical ophthalmology (Auckland, NZ) 2015;9:657. 16. Zhang W, Wang Y, Lee BTK, et al. A novel nanoscale-dispersed eye ointment for the treatment of dry eye disease. Nanotechnology 2014;25(12):125101. 17. Ferrara JL, Levine JE, Reddy P, et al. Graft-versus-host disease. The Lancet 2009;373(9674):1550-61. 18. Romero-Rangel T, Stavrou P, Cotter J, et al. Gas-permeable scleral contact lens therapy in ocular surface disease. American journal of ophthalmology 2000;130(1):25-32. 19. Fife RS, Chase WF, Dore RK, et al. Cevimeline for the treatment of xerostomia in patients with Sjögren syndrome: a randomized trial. Archives of internal medicine 2002;162(11):1293-300. 20. Ono M, Takamura E, Shinozaki K, et al. Therapeutic effect of cevimeline on dry eye in patients with Sjögren’s syndrome: a randomized, double-blind clinical study. American journal of ophthalmology 2004;138(1):6-17. 21. Petrone D, Condemi JJ, Fife R, et al. A double‐blind, randomized, placebo‐controlled study of cevimeline in Sjögren’s syndrome patients with xerostomia and keratoconjunctivitis sicca. Arthritis & Rheumatism 2002;46(3):748-54. 22. Vivino FB, Al-Hashimi I, Khan Z, et al. Pilocarpine tablets for the treatment of dry mouth and dry eye symptoms in patients with Sjögren syndrome: a randomized, placebo-controlled, fixed-dose, multicenter trial. Archives of internal medicine 1999;159(2):174-81. 23. Liu Y, Kam WR, Ding J, et al. Can tetracycline antibiotics duplicate the ability of azithromycin to stimulate human meibomian gland epithelial cell differentiation? Cornea 2015;34(3):342-6. 24. Kashkouli MB, Fazel AJ, Kiavash V, et al. Oral azithromycin versus doxycycline in meibomian gland dysfunction: a randomised double-masked open-label clinical trial. Br J Ophthalmol 2015;99(2):199-204. 25. Utine CA. Update and critical appraisal of the use of topical azithromycin ophthalmic 1% (AzaSite(®)) solution in the treatment of ocular infections. Clinical Ophthalmology (Auckland, NZ) 2011;5:801-9. 26. Vora GK, Gupta PK. Intense pulsed light therapy for the treatment of evaporative dry eye disease. Curr Opin Ophthalmol 2015;26(4):314-8. 27. Behrens A, Doyle JJ, Stern L, et al. Dysfunctional tear syndrome: a Delphi approach to treatment recommendations. Cornea 2006;25(8):900-7. 28. Vane JR, Bakhle YS, Botting RM. Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol 1998;38(1):97-120. 29. Aragona P, Tripodi G, Spinella R, et al. The effects of the topical administration of non-steroidal anti-inflammatory drugs on corneal epithelium and corneal sensitivity in normal subjects. Eye 2000;14(2):206-10. 30. Asai T, Nakagami T, Mochizuki M, et al. Three cases of corneal melting after instillation of a new nonsteroidal anti-inflammatory drug. Cornea 2006;25(2):224-7. 31. Isawi H, Dhaliwal DK. Corneal melting and perforation in Stevens Johnson syndrome following topical bromfenac use. Journal of Cataract & Refractive Surgery 2007;33(9):1644-6. 32. Prasher P. Acute corneal melt associated with topical bromfenac use. Eye & contact lens 2012;38(4):260-2. 33. Gokhale NS, Vemuganti GK. Diclofenac-induced acute corneal melt after collagen crosslinking for keratoconus. Cornea 2010;29(1):117-9. 34. Hessen M, Akpek EK. Dry Eye: an Inflammatory Ocular Disease. Journal of Ophthalmic & Vision Research 2014;9(2):240-50. 35. Marsh P, Pflugfelder SC. Topical nonpreserved methylprednisolone therapy for keratoconjunctivitis sicca in Sjögren syndrome. Ophthalmology 1999;106(4):811-6. 36. Hong S, Kim T, Chung S-H, et al. Recurrence after topical nonpreserved methylprednisolone therapy for keratoconjunctivitis sicca in Sjögren’s syndrome. Journal of ocular pharmacology and therapeutics 2007;23(1):78-82. 37. Bodor N, Buchwald P. Ophthalmic drug design based on the metabolic activity of the eye: Soft drugs and chemical delivery systems. The AAPS Journal 2005;7(4):E820-E33. 38. Jung HH, Ji YS, Sung MS, et al. Long-Term Outcome of Treatment with Topical Corticosteroids for Severe Dry Eye Associated with Sjögren’s Syndrome. Chonnam Medical Journal 2015;51(1):26-32. 39. McGhee CN, Dean S, Danesh-Meyer H. Locally administered ocular corticosteroids. Drug Safety 2002;25(1):33-55. 40. Tosi GM, Massaro‐Giordano M, Caporossi A, et al. Amniotic membrane transplantation in ocular surface disorders. Journal of cellular physiology 2005;202(3):849-51. 41. Suri K, Kosker M, Raber IM, et al. Sutureless amniotic membrane ProKera for ocular surface disorders: short-term results. Eye & contact lens 2013;39(5):341-7. 42. Fox RI, Chan R, Michelson JB, et al. Beneficial effect of artificial tears made with autologous serum in patients with keratoconjunctivitis sicca. Arthritis & Rheumatism 1984;27(4):459-61. 43. Kono I, Kono K, Narushima K, et al. Beneficial effect of the local application of plasma fibronectin and autologous serum in patients with the keratoconjunctivitis sicca of Sjögren’s syndrome. Ryūmachi[Rheumatism] 1986;26(5):339. 44. Tsubota K, Goto E, Fujita H, et al. Treatment of dry eye by autologous serum application in Sjögren’s syndrome. British Journal of Ophthalmology 1999;83(4):390-5. 45. Tananuvat N, Daniell M, Sullivan LJ, et al. Controlled study of the use of autologous serum in dry eye patients. Cornea 2001;20(8):802-6. 46. James MJ, Gibson RA, Cleland LG. Dietary polyunsaturated fatty acids and inflammatory mediator production. The American journal of clinical nutrition 2000;71(1):343s-8s. 47. Bhargava R, Kumar P. Oral Omega-3 Fatty Acid Treatment for Dry Eye in Contact Lens Wearers. Cornea 2015;34(4):413-20. 48. Bhargava R, Kumar P, Kumar M, et al. A randomized controlled trial of omega-3 fatty acids in dry eye syndrome. International journal of ophthalmology 2013;6(6):811. 49. Donnenfeld E. Effect of Oral Re-Esterified Omega-3 Nutritional Supplementation on Dry-Eye Disease: Double-Masked Randomized Placebo-Controlled Study. 2015 ASCRS ASOA Symposium and Congress; 2015: Ascrs. 50. Li Z, Choi J-H, Oh H-J, et al. Effects of Eye Drops Containing a Mixture of Omega-3 Essential Fatty Acids and Hyaluronic Acid on the Ocular Surface in Desiccating Stress-induced Murine Dry Eye. Current eye research 2014;39(9):871-8. 51. Utine CA, Stern M, Akpek EK. Clinical review: topical ophthalmic use of cyclosporin A. Ocular immunology and inflammation 2010;18(5):352-61. 52. Pflugfelder SC, Wilhelmus KR, Osato MS, et al. The autoimmune nature of aqueous tear deficiency. Ophthalmology 1986;93(12):1513-7. 53. Stern ME, Gao J, Siemasko KF, et al. The role of the lacrimal functional unit in the pathophysiology of dry eye. Experimental eye research 2004;78(3):409-16. 54. Sall K, Stevenson OD, Mundorf TK, et al. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. Ophthalmology 2000;107(4):631-9. 55. Stevenson D, Tauber J, Reis BL, et al. Efficacy and safety of cyclosporin A ophthalmic emulsion in the treatment of moderate-to-severe dry eye disease: a dose-ranging, randomized trial. Ophthalmology 2000;107(5):967-74. 56. Laibovitz RA, Solch S, Andriano K, et al. Pilot trial of cyclosporine 1% ophthalmic ointment in the treatment of keratoconjunctivitis sicca. Cornea 1993;12(4):315-23. 57. Kunert KS, Tisdale AS, Gipson IK. Goblet cell numbers and epithelial proliferation in the conjunctiva of patients with dry eye syndrome treated with cyclosporine. Archives of ophthalmology 2002;120(3):330-7. 58. Daull P, Lallemand F, Philips B, et al. Distribution of cyclosporine A in ocular tissues after topical administration of cyclosporine A cationic emulsions to pigmented rabbits. Cornea 2013;32(3):345-54. 59. Calvo P, Sánchez A, Martínez J, et al. Polyester nanocapsules as new topical ocular delivery systems for cyclosporin A. Pharmaceutical research 1996;13(2):311-5. 60. Yenice İ, Mocan MC, Palaska E, et al. Hyaluronic acid coated poly-ɛ-caprolactone nanospheres deliver high concentrations of cyclosporine A into the cornea. Experimental eye research 2008;87(3):162-7. 61. Khan W, Aldouby YH, Avramoff A, et al. Cyclosporin nanosphere formulation for ophthalmic administration. International journal of pharmaceutics 2012;437(1):275-6. 62. De Campos AM, Sánchez A, Alonso MaJ. Chitosan nanoparticles: a new vehicle for the improvement of the delivery of drugs to the ocular surface. Application to cyclosporin A. International Journal of Pharmaceutics 2001;224(1):159-68. 63. Kuwano M, Ibuki H, Morikawa N, et al. Cyclosporine A formulation affects its ocular distribution in rabbits. Pharmaceutical research 2002;19(1):108-11. 64. Karn PR, Do Kim H, Kang H, et al. Supercritical fluid-mediated liposomes containing cyclosporin A for the treatment of dry eye syndrome in a rabbit model: comparative study with the conventional cyclosporin A emulsion. International journal of nanomedicine 2014;9:3791. 65. Sheppard JD, Torkildsen GL, Lonsdale JD, et al. Lifitegrast Ophthalmic Solution 5.0% for Treatment of Dry Eye Disease. Ophthalmology;121(2):475-83. 66. Tauber J, Karpecki P, Latkany R, et al. Lifitegrast Ophthalmic Solution 5.0% versus Placebo for Treatment of Dry Eye Disease: Results of the Randomized Phase III OPUS-2 Study. Ophthalmology 2015;122(12):2423-31. 67. Shire’s OPUS-3 Phase 3 Trial with Lifitegrast Meets Primary and Key Secondary Endpoints, Significantly Reducing Patient-Reported Symptoms for Dry Eye Disease. 2015. Accessed 1/26/2016, 2016. www.shire.com/newsroom/2015/october/shires-opus-3-phase-3-trial-with-lifitegrast-meets-primary-and-key-secondary-endpoints. 68. Xie J, Chiang L, Contreras J, et al. Novel fiber-dependent entry mechanism for adenovirus serotype 5 in lacrimal acini. Journal of Virology 2006;80(23):11833-51. 69. Hsueh P-Y, Edman MC, Sun G, et al. Tear-mediated delivery of nanoparticles through transcytosis of the lacrimal gland. Journal of Controlled Release 2015;208:2-13. 70. Contreras-Ruiz L, Zorzi G, Hileeto D, et al. A nanomedicine to treat ocular surface inflammation: performance on an experimental dry eye murine model. Gene therapy 2013;20(5):467-77. |

