We present here the technology of the Argus II retinal implant (Second Sight) from the team that made it a reality—and is now hard at work on future enhancements.
 |
| Here, an Argus II recipient is wearing the device. |
The Argus I & II
Named for the mythical Greek giant with 100 eyes, the Argus is an epiretinal prosthesis that combines internally fixated electronics, with an external camera mounted on a pair of sunglasses.The first-generation device, the Argus I, used cochlear implant principles and some components for an FDA Phase I clinical trial in 2002 during which the safety of long-term stimulation was demonstrated, making way for the more advanced Argus II.
Argus I was implanted monocularly in six participants blinded by retinitis pigmentosa (RP).1-4 All participants perceived light once the device was activated and were able to perform visuospatial and motion tasks after brief training. Safety was observed in all instances of implantation during the study period; one participant had the device explanted for unrelated health reasons, and no evidence of tissue damage or electrode corrosion existed post-implantation at the five-year follow-up.1-4
Prior to receiving FDA approval as a humanitarian device in 2013, the Argus II obtained European Union approval and since then has been implanted 150 times to date globally; this number is anticipated to increase in the coming years. The device has been used primarily in patients with profound vision loss from RP in the United States, and, to a lesser extent, choroideremia and other retinal dystrophies.5
Function. The Argus II retinal prosthesis system is intended to function as a surrogate for the degenerated outer retinal layers and photoreceptors, interfacing with and stimulating the intact inner retinal neurons. Studies conducted in postmortem eyes concomitant with the engineering of the first Argus device showed approximately 80% survival of the inner nuclear layer and 30% of the ganglion cell layer in the maculae of patients suffering from RP.6 Nearly 90% preservation of these layers was seen in both the neovascular and atrophic forms of age-related macular degeneration.7,8
Device components. The Argus II consists of both implanted and external components. The external portion of the system is comprised of a pair of glasses with a small camera mounted into the frame, which is connected via cable to a video processing unit worn on the belt or on a shoulder strap. The visual information collected by the camera (when the system is turned on) is received, processed and converted into a brightness map in real time by the video processing unit. Power and data are sent via a radio frequency link from an external coil on the glasses to a receiving coil, which is contained within a hermetically sealed enclosure secured to the eye with a scleral band and sutures.
Subsequently, an inbuilt application-specific integrated circuit (ASIC) in the electronics unit generates stimulus-appropriate electrical pulses, which are relayed via a metallized polymer connecting cable to a 60-channel microelectrode epiretinal array. The array comes into contact with the retinal surface, where it is placed over the macula and tacked to the retina with a custom-made, spring-tension, metallic tack (Second Sight), allowing transmission of the electronic signal originating from the external portion of the system.
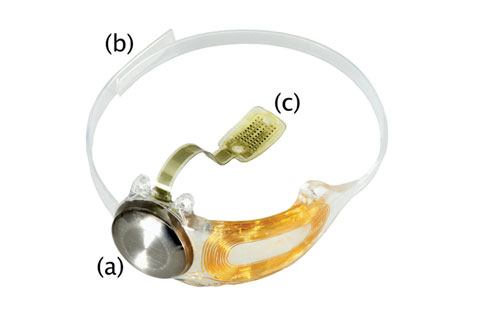 |
| The implanted components of the Argus II Retinal Prosthesis System can be seen here: The hermetically sealed coil and electronics enclosure (a) is attached to the scleral band (b), as well as via a metallized cable to the electrode array (c). Click image to enlarge. |
Surgical Procedure
Here is a concise summary of the key steps of the surgical implantation of the device:1. A 360-degree limbal conjunctival peritomy is performed.
2. The receiving coil is inserted under the lateral rectus muscle and extended into the inferotemporal quadrant; the electronics case is placed into the superotemporal quadrant.
3. The scleral band, to which the receiving coil and electronics case are attached, is passed under the inferior, medial and superior rectus muscles.
4. Suture tabs on the implant allow fixation to the sclera, and a Watzke sleeve (Labtician Ophthalmics) and mattress sutures or scleral tunneling secure the scleral band in the nasal quadrants.
5. Core and peripheral vitrecomies are performed.
6. A temporal sclerotomy of approximately 5mm is made to allow the introduction of the electrode array into the eye, after which the array is secured to the retina using a retinal tack. The sclerotomy is then sealed with sutures, and all other sclerotomies are closed.
7. An allograft consisting of either processed pericardium or aponeurosis (manufactured in France), donor cornea or autologous fascia lata is sutured over the implant to reduce the risk of irritation or erosion, and Tenon’s capsule and conjunctiva are subsequently closed.9
Surgical time generally falls between one and a half and four hours.
 |
| The external components of the Argus II: (a) The camera mounted on a pair of sunglasses, connected by cable to the battery pack (b) and video processing unit. Click image to enlarge. |
Safety, Reliability and Complications
Between 2007 and 2009, 30 participants (29 with RP and one with choroideremia) received the Argus II implant in the United States and Europe.10 The Argus II was implanted in the worse-seeing eye of each patient. At five years post-implantation, two Argus II implants had failed, both because of progressive loss of the ability to maintain the radiofrequency link between the external antenna on the glasses and the receiving antenna implanted on the eye.9 Both failed at approximately four years after implantation, but remained implanted to continue collecting long-term safety data.Also at five years following implantation, recently published data indicate that 60% of patients—18 out of 30—had experienced no device or surgery-related serious adverse events (SAEs). Of the described SAEs (e.g., conjunctival erosion, hypotony, conjunctival dehiscence and presumed endophthalmitis), all were manageable with standard ophthalmic treatment approaches and no eyes were lost during the course of the study.
Between years three and five post-implantation, only one additional SAE was reported: One patient with a rhegmatogenous retinal detachment who remained stable for one year eventually underwent pars plana vitrectomy, epiretinal membrane peel, fluid-air exchange and injection of silicone oil once neovascular glaucoma was noted. The intraocular pressure returned to normal shortly after surgery and the retinal detachment resolved. One patient died at six years after implantation due to causes unrelated to the Argus II.9
During the first five years of follow up, a total of three explantations were performed. One implant was removed at 14 months due to recurrent conjunctival erosion.11 An additional two patients requested explantation at three-and-a-half and four-and-one-third years post-implantation, respectively. The first request was due to recurrent conjunctival erosion, despite the absence of other device-related issues. The second patient experienced chronic hypotony and ptosis in the operated eye and chose explantation for cosmetic reasons. During each of the partial explantations, the cable was cut mid-vitreous, the sclerotomy sutured closed, and the extraocular portion of the device and attached portion of the cable were removed, leaving the electrode array tacked to the retina. No adverse events were noted following explanation.
Another recently published study reported the outcome of six patients who received the implant in a single center, by a single surgeon. At 12 months follow up, no SAEs requiring further surgery, such as wound dehiscence, endophthalmitis or retinal detachment, were noted.12
More complete data on adverse events experienced by the 100+ patients implanted with the Argus II device since approvals in Europe and the United States are awaiting publication.
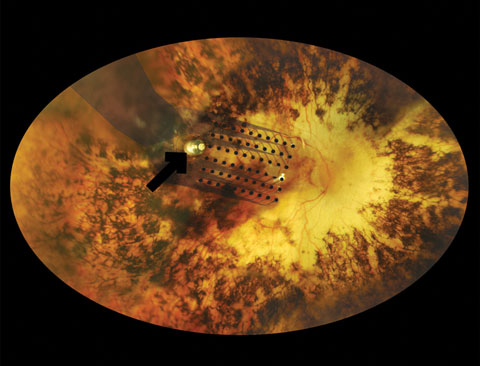 |
| This fundus photograph shows the electrode array component of the Argus II, which has been fastened to the recipient’s retina via the metallic tack (black arrow). Click image to enlarge. |
Functional and Psychophysical Assessment
The vision afforded by today’s retinal prostheses is quite limited. While some individuals are capable of significant feats of object and shape recognition, Argus II recipients experience visual percepts in a manner described most accurately as seeing “moving shadows.”The difficulty in quantifying visual improvement in these patients has presented issues with market approval by regulatory bodies. For instance, FDA guidelines have typically equated efficacy with measurable benefits, at least according to standardized outcomes (e.g., letter visual acuity, contrast sensitivity, visual field testing). While Argus II users have not shown these kinds of measurable improvements, the visual benefits become clearer when compared with patients with ultra-low vision—defined as hand movements, light projection or light perception—in whose cases a list of 760 distinct activities that can benefit has previously been elicited.13
The testing modalities used in both the Argus II feasibility study and the post-approval study include: (1) target localization, (2) motion direction discrimination and (3) grating visual acuity.14-15 Further, researchers have shown a significantly improved ability to identify high-contrast shapes in 11 participants with the implant.16
Target localization was evaluated by assessing the ability of participants to scan a visual scene and point to a white square that appeared in random locations on a black screen. At three years post-implantation, 89% of the 28 participants were able to localize the target more accurately with the system turned on than with the system off.10
In testing motion direction discrimination, participants were asked to identify the direction of movement of a white bar across a black screen at a random angle, and 56% of the 27 participants were better able to do so with the system on than with the system off.10
Grating visual acuity, originally developed for a clinical trial of a subretinal photodiode array, measures the ability of an individual to differentiate the orientation of black and white gratings of varying spatial frequencies.17 And while no participant was able to perform this test in any measurable capacity with the system turned off, 48% at one year and 33% at three years post-implantation scored 2.9 logMAR or more with the system on.
Future Developments
In the last decade, we have seen several different treatment modalities for degenerative retinal disease emerging in the form of clinical trials. Trials with gene therapy, stem cell transplantation and electronic neural prostheses implanted within different locations in the eye are all underway.18-22 However, retinal prostheses are currently the only market-ready modality to date.Only two retinal prostheses—the Argus II and the Alpha AMS (Retina Implant AG)—are approved in Europe, the US or both. However, modifications to currently available image-processing software will likely help existing prostheses garner greater benefits for patients. As it stands, image-processing software modifications have already been applied to improve the vision obtainable with the Argus II device. Various modifications have allowed improved edge-detection and image enhancement, which took place during early studies on shape and object recognition. In 2013, researchers presented new software that uses image magnification and minimization, as well as image enhancement features, to achieve resolution that seems to exceed the electrodes’ limit. It allowed one Argus II patient to achieve visual acuity equivalent to 20/200 (logMAR 1.0) on grating acuity testing.23
Future devices are likely to have more and smaller electrodes. However, the potential additional benefit is still uncertain. The makers of Argus II have alluded to a next-generation device with 240 electrodes, with the possibility of the addition of peripheral electrodes.24
The ultimate goal: design devices with electrodes similar in size to the cell bodies of individual retinal ganglion cells, allowing individual cell activation.
Although more progress is needed, the ability to offer functional vision in those who otherwise would have had none is a remarkable feat in itself. The Argus II retinal prosthesis is the end result of years of hard work on the part of innumerable investigators who contributed their time, ideas and efforts. Further, were it not for the altruism of dozens of volunteers during the trials of both the Argus I and Argus II, this work would not have been possible.
Luminaries in Artificial VisionWhile significant progress in treating degenerative outer retinal disease has been made in the past several decades, the scientific exploration that ultimately led us to the present day began as early as the mid 18th century. French physician Charles LeRoy created the sensation of light in the blind by passing electrical currents around the head in 1755.25 Foerster discovered in 1929 that it was possible to elicit transient and reproducible visual percepts—phosphenes—with direct electrical stimulation of the visual pathway. In the 1960s, Button and Puttnam implanted four occipital lobe electrodes with percutaneous connections in three blind patients.26 Using handheld photocells, which transmitted light-dependent signals to the occipital lobe electrodes, participants were able to roughly determine the location of illuminated objects.26Other investigators have conducted important work in visual prosthetics, further showing that electrical stimulation of the visual cortex can create spatial visual percepts.27,28 However, due to advances in alternative approaches—namely, retinal artificial vision prostheses as well as the significant associated morbidity—the cortical visual prosthesis has not yet reached medical implant approval status. Twenty-five years of focused work on the epiretinal implant around the globe has led us to the Argus II. The original work leading to the advent of the Argus II implant’s current iteration began in 1987. Dr. Humayun was a Duke medical student beginning his career in ophthalmology and struggling with patients’ loss of vision, including his own grandmother, who suffered from diabetic retinopathy. Eugene de Juan, Jr., MD, a member of the medical staff and vitreoretinal surgeon at the Duke University Eye Center at the time, and Howard Phillips, a nuclear and electrical engineer, along with Dr. Humayun, comprised the three-person team that launched the research that eventually led to the Argus implant. In 1991, Dr. Humayun’s group reported the results of its retinal stimulation experiments in rabbits in which photoreceptor lesions had been chemically induced.29 Shortly thereafter, groups at Harvard, MIT and in Cologne, Germany also began to use implanted electrodes to stimulate the visual pathway.30,31 From 1992 to 1994, Dr. Humayun’s group conducted the first human tests to evaluate epiretinal electrical stimulation. Studies began at the Duke Eye Center and continued later at the Wilmer Eye Institute.32,33 So began an intensive period of animal experiments, development of surgical technique and instruments, intraoperative patient testing and simulation studies of prosthetic vision. While at Wilmer, the intraocular retinal prosthesis laboratory obtained three important new collaborators: Gislin Dagnelie, PhD, for his knowledge of psychophysics and simulation studies, MD-PhD student Robert Greenberg and postdoctoral researcher James Weiland, PhD, from the University of Michigan for his expertise in electrode materials and neural stimulation.34 |
Dr. Humayun is a professor of Ophthalmology, Biomedical Engineering and Cell and Neurobiology, the director of the Institute for Biomedical Therapeutics at the University of Southern California and co-director of the USC Roski Eye Institute.
Dr. Ameri is the director of the USC Retinal Degeneration Center and an assistant professor of Clinical Ophthalmology at the USC Roski Eye Institute.
|
1. de Balthasar C, Patel S, Roy A, et al. Factors affecting perceptual thresholds in epiretinal prostheses. Invest Ophthalmol Vis Sci. 2008;49:2303–14. 2. Horsager A, Greenwald SH, Weiland JD, et al. Predicting visual sensitivity in retinal prosthesis patients. Invest Ophthalmol Vis Sci. 2009;50:1483–91. 3. Mahadevappa M, Weiland JD, Yanai D, et al. Perceptual thresholds and electrode impedance in three retinal prosthesis subjects. IEEE Trans Neural Sys Rehab Eng. 2005;13:201–6. 4. Yanai D, Weiland JD, Mahadevappa M, et al. Visual performance using a retinal prosthesis in three subjects with retinitis pigmentosa. Am J Ophthalmol. 2007;143:820–7. 5. Accessed 20 Jan 2017. Second Sight Medical Products. Argus II Retinal Prosthesis System Dry AMD Feasibility Study Protocol. Available at www.clinicaltrials.gov/ct2/show/NCT02227498. Accessed January 20, 2017. 6. Santos A, Humayun MS, de Juan E Jr, et al. Preservation of the inner retina in retinitis pigmentosa. A morphometric analysis. Arch Ophthalmol. 1997;115:511–5. 7. Kim SY, Sadda S, Humayun MS, et al. Morphometric analysis of the macula in eyes with geographic atrophy due to age-related macular degeneration. Retina. 2002;22:464–70. 8. Kim SY, Sadda S, Pearlman J, et al. Morphometric analysis of the macula in eyes with disciform age-related macular degeneration. Retina. 2002;22:471–7. 9. Da Cruz L, Dorn JD, Humayun MS, et al. Five-year safety and performance results from the Argus II retinal prosthesis system clinical trial. Ophthalmology. 2016;123(10):2248-54. 10. Ho AC, Humayun MS, Dorn JD, et al. Long-term results from an epiretinal prosthesis to restore sight to the blind. Ophthalmology. 2015;122:1547–54. 11. Humayun MS, Dorn JD, da Cruz L, et al. Interim results from the international trial of Second Sight’s visual prosthesis. Ophthalmology. 2012;119:779–88. 12. Rizzo S, Belting C, Cinelli L, et al. The Argus II retinal prosthesis: 12-month outcomes from a single-study center. Am. J. Ophthalmol. 2014;157:1282-90. 13. Dagnelie G, Jeter P, Dalvin L, et al. Invest Ophthalmol Vis Sci. 2013;54:ARVO E-Abstract 2784. 14. Second Sight Medical Products. Argus II Retinal Stimulation System Feasibility Protocol. Available at www.clinicaltrials.gov/ct2/show/NCT00407602. Accessed January 20, 2017. 15. Second Sight Medical Products. New Enrollment Post-Approval Study of the Argus® II Retinal Prosthesis System Available at www.clinicaltrials.gov/ct2/show/NCT00407602. Accessed January 20, 2017. 16. Luo YHL, Zhong JJ, da Cruz L. The use of Argus II retinal prosthesis by blind subjects to achieve localisation and pre- hension of objects in 3-dimensional space. Graefes Arch Clin Exp Ophthalmol. 2014;253:1907–14. 17. Zhou DD, Dorn JD, Greenberg RJ. The Argus II retinal prosthesis system: an overview. IEEE Int Conf Multimedia Expo Workshops (ICMEW). 2013;1–6. 18. Petrs-Silva H, Linden R. Advances in gene therapy technologies to treat retinitis pigmentosa. Clin Ophthalmol. 2014;8:127–36. 19. Al-Shamekh S, Goldberg JL. Retinal repair with induced pluripotent stem cells. Transl Res. 2014;163:377–86. 20. Stingl K, Bartz-Schmidt KU, Besch D, et al. Subretinal visual implant alpha IMS - Clinical trial interim report. Vision Res. 2015;111(Pt B):149–60. 21. Kitiratschky VB, Stingl K, Wilhelm B, et al. Safety evaluation of “retina implant alpha IMS” prospective clinical trial. Graefes Arch Clin Exp Ophthalmol. 2015;253: 381–7. 22. Ayton LN, Blamey PJ, Guymer RH, et al. First-in-human trial of a novel suprachoroidal retinal prosthesis. PLoS One. 2014;9:1–26. 23. Sahel J, Mohand-Said S, Stanga P, Caspi A, Greenberg R, Argus II Study Group, 2013. AcuboostTM: enhancing the maximum acuity of the Argus II Retinal Prosthesis System. In: ARVO Meeting Abstracts 2013;(54):1389. 24. Stronks HC, Dagnelie GG. The functional performance of the Argus II retinal prosthesis. Expert Rev Med Dev. 2014;11:23e30. 25. LeRoy C. Où l’on rend compte de quelques tentatives que l’on a faites pour guérir plusieurs maladies par l’électricité Hist Acad Roy Sciences (Paris), Mémoire Math Phys. 1755;60:87–95. 26. Button J, Puttnam T. Visual responses to cortical stimulation in the blind. J Iowa Med Soc. 1962;17–21. 27. Brindley GS, Lewin WS. The sensations produced by electrical stimulation of the visual cortex. J Physiol (Lond). 1968;196:479–93. 28. Troyk P, Bak M, Berg J, et al. A model for intracortical visual prosthesis research. Artf Org. 2003;27:1005–15. 29. Humayun MS, Probst RH, Hickingbotham D, et al. Invest Ophthalmol Visual Sci. 1991;32:ARVO Abstract 2747. 30. Rizzo JF, Socha M, Edell D, et al. Development of a silicon retinal implant: Surgical methods and mechanical design. Invest Ophthalmol Vis Sci. 1994;4(35):1535. 31. Szurmann P. Invest Ophthalmol Vis Sci. 1997;38:ARVO Abstract 185. 32. Humayun MS, de Juan E Jr, Dagnelie G, et al. Visual perception elicited by electrical stimulation of retina in blind humans. Arch Ophthalmol. 1996;114:40–6. 33. Humayun MS, de Juan E Jr, Weiland JD, et al. Pattern electrical stimulation of the human retina. Vision Res. 1999;39:2569–76. 34. Humayun MS, de Juan E Jr, Dagnelie G. A Quarter Century of Retinal Prosthesis Research and Development.Ophthalmology. 2016;123:S90-S97. 1. |

