In 2016, we gained the ability to treat our keratoconus patients suffering from progressive vision loss through the FDA approval of epi-off corneal crosslinking. Historically, keratoconus patients were given glasses or contact lenses to help optimize their vision in the hopes that they would not progress. Thankfully, we can now slow and even halt keratoconus progression, reducing the number of patients who need corneal transplants.
As we continue to perform conventional epi-off corneal crosslinking, outside the United States crosslinking protocols are evolving and researchers are proposing new protocols that aim to offer a faster, more comfortable and effective way to help our patients. This article will look at various crosslinking methods, including accelerated, pulsed light, transepithelial and others that seek to improve on the gold standard, and discuss what these advancements and innovations could mean for us and our patients in 2022 and in the future.
 |
|
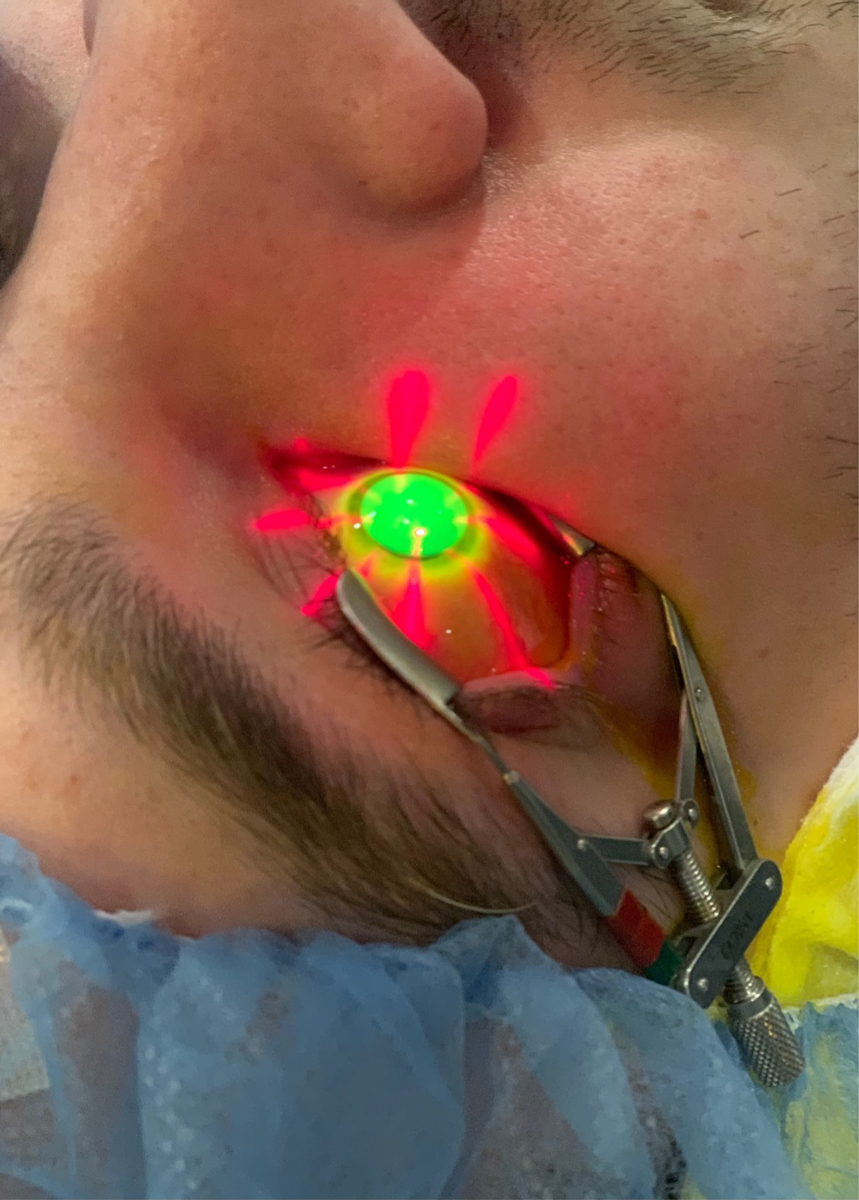
Epi-off corneal crosslinking in a patient with progressive keratoconus. Photo by Alexandra Wiechmann, OD. Click image to enlarge. |
Conventional Epi-off Crosslinking
The current gold standard for corneal crosslinking in patients with progressive keratoconus and other corneal ectasia is the epi-off Dresden protocol created in 2003.1 This process involves the removal of 8mm to 9mm of epithelium from the central cornea, followed by the application of a riboflavin solution every two minutes for 30 minutes. After the cornea is saturated, it is irradiated using 370nm UVA at an intensity of 3mW/cm2 while riboflavin is applied every two minutes for another 30 minutes. A minimum corneal thickness of 400µm is required before UV irradiation or intraoperatively. This is the only FDA-approved crosslinking protocol in the United States, and it has proven to be effective at strengthening the cornea and halting progression in patients with keratoconus.
While effective, the Dresden protocol has its disadvantages. Removal of the epithelium has a higher risk of pain and increased discomfort. It is greatest in the first three days postoperatively and often requires the use of topical or oral pain medications.2 There is a chance that the patient will develop reduced visual acuity, but per the data submitted to the FDA, only 1% to 2% of patients had corneal opacity or scarring at the 12-month follow-up.3
Typical postoperative protocol includes application of a bandage contact lens immediately after treatment and frequent use of topical antibiotics during the first three to five days as the epithelium heals. Upon removal of the bandage contact lens, the patient is often switched to topical steroids to decrease inflammation and reduce the risk of persistent corneal haze. This is different than the postoperative haze typically seen after crosslinking between months one through six, which is not visually significant and does not require steroids for resolution.4 Adding topical steroids before the corneal epithelium has fully healed has been linked to an increased risk of microbial keratitis.5
Epi-off Accelerated Crosslinking
Conventional crosslinking lasts a minimum of one hour and is one of the longest procedures performed in ophthalmology clinics today. To make the process more efficient and less time-consuming, researchers and clinicians have been trying out new accelerated protocols with higher UVA irradiation employed for a shorter amount of time. This is based on the Busen-Roscoe law of reciprocity: “a response is determined by the product of the intensity and the duration of the stimulus.”6 This means that for an accelerated protocol to achieve similar results as the Dresden protocol, it would need to maintain the same or have a cumulative energy of 5.4J/cm2.
In a 2021 study, the seven-year data comparing epi-off accelerated crosslinking and conventional epi-off crosslinking in progressive keratoconus was shared.7 The accelerated protocol used UVA irradiation of 9mW/cm2 for a duration of 10 minutes instead of the typical 30 minutes.7 There was no significant difference between protocols.7
Conversely, another study included multiple accelerated protocols, including three minutes at 30mW/cm2, five minutes at 18mW/cm2 and 10 minutes at 9mW/cm2, and found that each accelerated protocol was less effective at flattening the cornea when compared with conventional crosslinking.8 While there is some promise of achieving successful crosslinking in shorter duration, more research is needed to find the ideal energy and duration.
The Importance of Oxygen
Not only is the energy and duration important but we now know that oxygen is also key in the crosslinking process. For corneal crosslinking to occur, UVA must interact with both riboflavin and oxygen in the stroma to create oxygen free-radicals that kick-start the formation of covalent bonds between collagen molecules.
To illustrate the importance of oxygen in crosslinking, ex vivo epi-off crosslinking was performed on porcine corneas in both a regular oxygen environment (21%) and a low-oxygen environment (<0.1%).9 The corneas that were crosslinked in the normal oxygen environment showed improved biomechanical stability, while the corneas in the low-oxygen environment had results similar to the non-treated controls.9 This ultimately raised concerns over the effectiveness of transepithelial crosslinking (epi-on) where the corneal epithelium limits the amount of oxygen diffusion into the stroma.7
In an effort to make epi-off crosslinking even more effective, it was determined that the level of oxygen in the stroma depletes within seconds after exposure to UVA irradiation and takes three to four minutes to replenish to normal levels.10 From this came a crosslinking method called pulsed light accelerated crosslinking, in which UVA irradiation is applied on and off repeatedly to allow for oxygen to diffuse back into the stroma. When compared with conventional crosslinking, this method alone was not as effective because it did not penetrate as far down into the cornea as conventional crosslinking but may prove to be a key component in future crosslinking protocols.11
To determine how much of the cornea is crosslinked, researchers measure what is called the stromal demarcation line, which represents a transition zone between crosslinked and non-crosslinked stromal tissue. It has been found to occur in histopathological studies as early as 24 hours and has been seen as early as two weeks on slit lamp biomicroscopy.12 The presence and depth of a stromal demarcation line has been considered an indirect measurement of treatment effectiveness. When comparing epi-off and transepithelial protocols, the demarcation line was significantly deeper with the former (275.05±41.80µm vs. 132.60±22.14µm).13 The absence or shallow location of the demarcation line with transepithelial crosslinking is likely secondary to the shielding effect of the intact epithelium to riboflavin and UVA irradiation.
 |
|
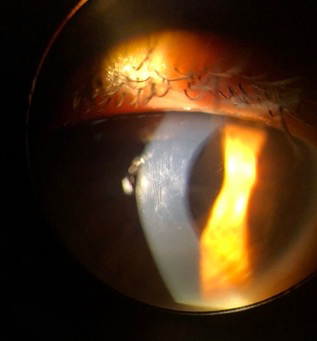
Vogt’s striae in a patient with advanced keratoconus before crosslinking. Photo by Thomas Nettleton, OD. Click image to enlarge. |
Crosslinking Thin Corneas
As the epithelium can interfere with successful crosslinking, so too can the corneal thickness. Within the Dresden protocol, patients with thin pachymetry measurements can still undergo crosslinking if their corneas can be swelled to 400µm with a hypo-osmolar riboflavin solution. Unfortunately, there are many patients with moderate to advanced cases of keratoconus whose corneas will never swell to 400µm safely but who, arguably, have a greater need for crosslinking to avoid penetrating keratoplasty. To fill this need, there have been multiple attempts to successfully crosslink thinner corneas.
One technique called contact lens-assisted crosslinking involves placing a riboflavin-soaked daily disposable contact lens onto the cornea after removing the epithelium. The eye is then irradiated with UVA as performed in conventional crosslinking. This technique was performed in a study that produced unsuccessful results, showing no change in corrected distance visual acuity or Kmax postoperatively.14 This was attributed to the fact that contact lenses act as a barrier by preventing most UVA from reaching the stroma and interfering with oxygen diffusion.15
A new protocol was developed in 2021 to crosslink ultrathin corneas. Referred to as the sub400 protocol, it uses a previously published algorithm to perform customized crosslinking based on each eye’s corneal thickness after epithelial removal.16,17 Each eye receives the same UVA irradiation (3mW/cm2), but the duration varies in order to crosslink without harming the endothelium.17 Study results showed successful crosslinking in corneas ranging in thickness from 214µm to 398µm with keratoconus progression halted in 90% of eyes.17
While many researchers have been trying to effectively crosslink patients with thinner corneas, others are focused on creating a quicker, safer and more comfortable procedure for patients all while leaving the epithelium in place. This new wave in crosslinking is called transepithelial crosslinking, or epi-on crosslinking.
Epi-On Crosslinking
Transepithelial crosslinking leaves the epithelium intact and has significantly less postoperative pain, offers a quicker visual recovery (with vision often returning to preoperative levels within two or three days) and allows patients to resume contact lens wear in one week.18 There is also a decreased risk of postoperative infection and persistent corneal haze infrequently seen with conventional crosslinking. With the quick postoperative recovery associated with transepithelial crosslinking, it raises the possibility of performing bilateral crosslinking on the same day.18
While the benefits may seem to speak for themselves, an intact epithelium is quite literally a barrier to a successful crosslinking procedure. Riboflavin is a hydrophilic, high molecular weight molecule that cannot penetrate through an intact epithelium on its own.19 In a study evaluating the difference in biomechanical effect for both epi-on and epi-off crosslinking in rabbits, it was concluded that epi-on crosslinking produced only one fifth of the biomechanical rigidity in the cornea found after conventional crosslinking.20 When this is translated to human corneas, it would likely equate to a 64% increase in rigidity with transepithelial vs. a 320% increase with conventional crosslinking.20 The authors attributed this disparity to insufficient riboflavin diffusion through an intact epithelium.20
Certain chemicals can be used to enhance riboflavin permeability and often include benzalkonium chloride, gentamicin and ethylenediamine tetra-acetic acid (EDTA).21,22 Transepithelial crosslinking was performed on 26 eyes of patients between the ages of 11 and 26 with a riboflavin solution containing two permeability enhancers, trometamol and EDTA.23 Results showed that both uncorrected and corrected distance acuity improved in the first three to six months before gradually returning to baseline over 24 months.23 A similar trend was found with Kmax values.23 Due to the ineffectiveness of treatment at the 12-month follow-up, the team ultimately retreated 50% of the patients younger than 18 with epi-off crosslinking.23 It was concluded that transepithelial crosslinking was likely not a good option for halting progression in patients younger than 26 who have more aggressive forms of keratoconus.23
 |
|
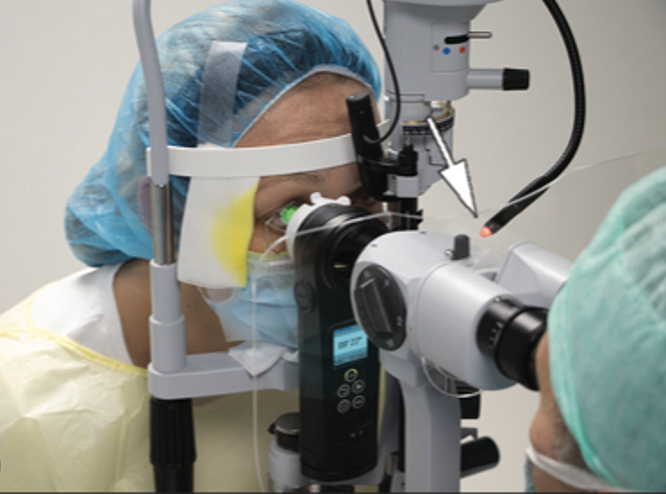
The C-Eye by Emagine AG performs crosslinking on the patient at the slit lamp.25 Click image to enlarge. |
Iontophoresis
Another way that researchers have tried improving riboflavin permeability with transepithelial crosslinking is with iontophoresis, which is the use of an electrical current to enhance molecule penetration into a tissue. In one study, a 1mA electrical current was used to improve riboflavin absorption through an intact epithelium.24 At the two-year follow-up, there was a 20% failure rate in the iontophoresis crosslinking group compared with a 7.5% failure rate with conventional crosslinking.24 The same study also evaluated for the presence of a corneal demarcation line at the two-year follow-up and determined that it was only visible in the iontophoresis corneas 35% of the time compared with 95% in conventional crosslinking cases.24 While an effective way to improve riboflavin penetration through the epithelium, iontophoresis alone does not appear to be the trick to successful epi-on crosslinking.
With the continuous effort and the multitude of modifications in crosslinking protocols, researchers are bound to achieve a successful transepithelial crosslinking result that rivals conventional epi-off crosslinking. Throughout the literature, the consensus appears to be that we need clinical trials that show effective crosslinking with longer follow-ups and larger sample sizes. Until then, I believe we should focus on increasing access to care for patients who need crosslinking. One research team out of Zurich, led by Farhad Hafazi, MD, PhD, has created a device that aims to do just that.
Crosslinking at the Slit Lamp
The C-Eye by Emagine AG, a UVA illumination device, was designed to be mounted onto the slit lamp, allowing patients to receive crosslinking while seated in an exam room.25 For epi-off crosslinking, the epithelium is removed at the slit lamp using an ethanol-based technique.25 After, the patient is relocated to a reclining chair where corneal thickness is measured while riboflavin is instilled.25 They are then moved back to the slit lamp for UVA irradiation treatment lasting for 10 minutes.25 The creator of this device says that it will “increase accessibility to treatment globally and reduce overall costs related to the procedure” by moving it from the operating room to the exam room.25 Technology like this may be the next step toward optometrists performing crosslinking. At the start of 2022, there are currently three states whose scope of practice allows ODs to perform the procedure: Virginia, Louisiana and Oklahoma.
What’s Next?
Glaukos, the creator of the first-generation iLink therapy currently FDA-approved for epi-off corneal crosslinking, is currently vying for FDA approval of an epi-on crosslinking protocol. In February 2021, Glaukos shared positive results from its pivotal Phase III trial for iLink epi-on therapy.26 The study included 279 eyes randomized in a 2:1 ratio to receive transepithelial therapy or placebo-controlled treatment.26 The study demonstrated a statistically significant improvement in Kmax at six months from baseline (-1.00D), meeting the primary efficacy endpoint.26 The company also shared that the treatment was well-tolerated, with no patients leaving the study due to adverse events.26 The next step for Glaukos includes submitting an NDA this year, and the company is aiming for FDA approval in 2023.26
Takeaways
There are many protocols that have been created to try and improve on the gold standard of crosslinking, the epi-off Dresden protocol, which is currently the only FDA-approved crosslinking method in the United States. Researchers from all over the world are looking for ways to make the procedure faster, more effective, more comfortable and even more accessible to patients. Until we find a better option, we can rest assured knowing that we do currently have a way to halt progression in our patients with progressive keratoconus and improve the quality of their lives in invaluable ways.
Dr. Arendt is an ocular disease and refractive surgery resident at Parkhurst Nuvision in San Antonio, TX. She is a member of the Refractive Surgery Alliance, American Optometric Association and Texas Optometric Association. She has no financial interests to disclose.
1. Wollensak G, Spörl E, Seiler T. Treatment of keratoconus by collagen cross linking. Ophthalmologe. 2003;100(1):44-9. 2. Ghanem V, Ghanem R, De Oliveira R. Postoperative pain after corneal collagen cross-linking. Cornea. 2013;32(1):20-4. 3. Food and Drug Administration. Photrexa. www.accessdata.fda.gov/drugsatfda_docs/label/2016/203324s000lbl.pdf. Accessed March 15, 2022. 4. Greenstein SA, Fry KL, Bhatt J, Hersh PS. Natural history of corneal haze after collagen crosslinking for keratoconus and corneal ectasia: Scheimpflug and biomicroscopic analysis. J Cataract Refract Surg. 2010;36(12):2105-14. 5. Tzamalis A, Romano V, Cheeseman R, et al. Bandage contact lens and topical steroids are risk factors for the development of microbial keratitis after epithelium-off CXL. BMJ Open Ophthalmol. 2019;4(1):e000231. 6. Elsevier’s Dictionary of Psychological Theories. 7. Nicula C, Rednik A, Nicula A, et al. Comparative results between “epi-off” accelerated and “epi-off” standard corneal collagen crosslinking-UVA in progressive keratoconus – 7 years of follow-up. Ther Clin Risk Manag. 2021;17:975-88. 8. Miraftab M, Hashemi H, Abdollahi M, et al. The efficacy of standard versus accelerated epi-off corneal cross-linking protocols: a systematic review and sub-group analysis. Int Ophthalmol. 2019;39(11):2675-83. 9. Richoz O, Hammer A, Tabibian D, et al. The biomechanical effect of corneal collagen cross-linking (CXL) with riboflavin and UV-A is oxygen dependent. Transl Vis Sci Technol. 2013;2(7):6. 10. Kamaev P, Friedman M, Sherr E, Muller D. Photochemical kinetics of corneal cross-linking with riboflavin. Invest Ophthalmol Vis Sci. 2012;53(4):2360-7. 11. Jiang L, Jiang W, Qiu S. Conventional vs. pulsed-light accelerated corneal collagen cross-linking for the treatment of progressive keratoconus: 12-month results from a prospective study. Exp Ther Med. 2017;14(5):4238-44. 12. Seiler T, Hafezi F. Corneal cross-linking-induced stromal demarcation line. Cornea. 2006;25(9):1057-9. 13. Spadea L, Di Genova L, Tonti E. Corneal stromal demarcation line after 4 protocols of corneal crosslinking in keratoconus determined with anterior segment optical coherence tomography. J Cataract Refract Surg. 2018;44(5):596-602. 14. Jacob S, Kumar D, Agarwal A, et al. Contact lens-assisted collagen cross-linking (CACXL): a new technique for cross-linking thin corneas. J Refract Surg. 2014;30(6):366-72. 15. Chen X, Stojanovic A, Eidet J, Utheim T. Corneal collagen cross-linking (CXL) in thin corneas. Eye Vis (London). 2015;2(1):15. 16. Kling S, Hafezi F. An algorithm to predict the biomechanical stiffening effect in corneal cross-linking. J Refract Surg. 2017;33(2):128-36. 17. Hafezi F, Kling S, Gilardoni F, et al. Individualized corneal cross-linking with riboflavin and UV-A in ultrathin corneas: the sub400 protocol. Am J Ophthalmol. 2021;224:133-42. 18. Stulting RD, Trattler WB, Woolfson JM, et al. Corneal crosslinking without epithelial removal. J Cataract Refract Surg. 2018;44(11):1363-70. 19. Baiocchi S, Mazzotta C, Cerretani D, et al. Corneal crosslinking: Riboflavin concentration in corneal stroma exposed with and without epithelium. J Cataract Refract Surg. 2009;35(5):893-9. 20. Wollensak G, Iomdina E. Biomechanical and histological changes after corneal crosslinking with and without epithelial debridement. J Cataract Refract Surg. 2009;35(3):540-6. 21. Chang S, Chi R, Wu C, Su M. Benzalkonium chloride and gentamicin cause a leak in corneal epithelial cell membrane. Exp Eye Res. 2000;71(1):3-10. 22. Majumdar S, Hippalgaonkar K, Repka M. Effect of chitosan, benzalkonium chloride and ethylenediaminetetraacetic acid on permeation of acyclovir across isolated rabbit cornea. Int J Pharm. 2008;348(1):175-8. 23. Caporossi A, Mazzotta C, Paradiso AL, et al. Transepithelial corneal collagen crosslinking for progressive keratoconus: 24-month clinical results. J Cataract Refract Surg. 2013;39(8):1157-63. 24. Jouve L, Borderie V, Sandali O, et al. Conventional and iontophoresis corneal cross-linking for keratoconus: efficacy and assessment by optical coherence tomography and confocal microscopy. Cornea. 2017;36(2):153-62. 25. Hafezi F, Richoz O, Torres-Netto E, et al. Corneal cross-linking at the slit lamp. J Refract Surg. 2021;37(2):78-82. 26. Glaukos announces positive phase 3 trial results for iLink epi-on investigational therapy. Eyewire. February 25, 2021. eyewire.news/articles/ glaukos-announces-positive-phase-3-trial-results-for-ilink-epi-on-investigational-therapy/. Accessed March 15, 2022. |

