 Exciting new technologies for
enhancing the lives of low
vision patients include telescopic
intraocular lenses (IOLs) and
stem cells harvested from a patient's
own body for transplant in the eye.
Let's talk briefly about how they
work, and what recent research
shows about their clinical value.
Exciting new technologies for
enhancing the lives of low
vision patients include telescopic
intraocular lenses (IOLs) and
stem cells harvested from a patient's
own body for transplant in the eye.
Let's talk briefly about how they
work, and what recent research
shows about their clinical value.
Stem Cell Transplants
Pluripotent skin stem cells are being programmed as retinal pigment epithelium (RPE) stem cells for the treatment of macular degeneration.1 They are lauded as easier to grow and transplant, and are derived from the patient's own cells so they don't require an invasive procedure for harvesting. Additionally, the cells self-renew, expand in cultures and differentiate into RPE.1
In September, a Japanese woman in her 70s became the world's first patient to receive tissue derived from induced pluripotent stem cells as part of an experimental treatment to repair damage caused by age-related macular degeneration.
RPE cells are derived from human induced pluripotent stem cells. During the two-hour procedure at the Institute for Biomedical Research and Innovation in Kobe, Japan, surgeons grafted a single 1.3mm x 3.0mm sheet of RPE cells into the subretinal space of one eye. No serious hemorrhaging or complications occurred.2
 The epithelium sheet was developed
by ophthalmologist Masayo
Takahashi, MD, PhD, of the RIKEN
Center for Developmental Biology
using pluripotent stem cells, which
have the unique potential to differentiate
into almost any type of cell
found within the body. The most
prominent type of pluripotent stem
cell is the embryonic stem cell; however,
limited resources and ongoing
controversy has prevented widespread
adoption. Induced pluripotent
stem cells, on the other hand,
are artificially derived from an adult
somatic cell. In this case, the pluripotent
stem cells were derived from
the patient's own skin cells, then
converted into RPE cells and grown
into a sheet.
The epithelium sheet was developed
by ophthalmologist Masayo
Takahashi, MD, PhD, of the RIKEN
Center for Developmental Biology
using pluripotent stem cells, which
have the unique potential to differentiate
into almost any type of cell
found within the body. The most
prominent type of pluripotent stem
cell is the embryonic stem cell; however,
limited resources and ongoing
controversy has prevented widespread
adoption. Induced pluripotent
stem cells, on the other hand,
are artificially derived from an adult
somatic cell. In this case, the pluripotent
stem cells were derived from
the patient's own skin cells, then
converted into RPE cells and grown
into a sheet.
This pilot study follows an earlier preclinical safety and feasibility evaluation of human-induced pluripotent RPE cell sheets created without using artificial scaffolds.1 The research examined cell morphology, physiological behavior, gene expression, immunogenicity and tumor formation in rodent and non-human primate models.
RPE cells are formed into a sheet to be implanted into the subretinal space of a patient with AMD. Researchers believe the use of pluripotent stem cells will improve upon current treatments that are designed to halt neovascularization, but do not repair photoreceptor cell damage that may have already occurred prior to administration. The RPE graft, however, could stop further damage and may even eventually stimulate some healing of the epithelium.
Additionally, harvesting the patient's own cells from an innocuous site such as the skin reduces potential complications associated with immune rejection and avoids invasive harvesting procedures.
The current patient will be monitored for both functional integration and adverse reactions during a one-year initial intensive observation period, with subsequent follow-up observation for an additional three years. Five additional patients will be treated using the same procedure as part of the pilot study. Although early in progress, the possibilities could be life changing.
Artificial Implant Technologies
Although RPE stem cell transplantation is years away, innovative technologies available today for patients with end-stage macular diseases include the Argus II and telescopic IOLs.
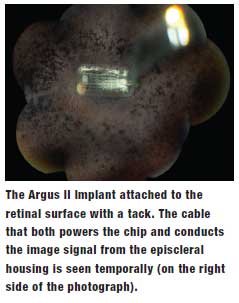
• Argus II (Second Sight Medical Products). Essentially a circuit board that allows for 32,000-pixel imaging, this device is inserted onto the macula. By stimulating a wireless system and spectacles as a transducer, it sends a current to the occipital lobe, allowing patients to see images, shadows and shapes. It is indicated in patients 25 years or older with late-stage retinitis pigmentosa progression if the patient sees only bare light or has no light perception, according to the manufacturer. Eligible patients must have no previous history of useful form vision. If the patient is phakic, the surgeon removes their natural lens during the implantation procedure.3
Recent small studies continue to show significant improvement in vision after implantation. In a study of 27 patients from 2011, 93% improved their ability to repeatedly touch a small square projected on a touch-screen monitor.4 In another study, researchers evaluated 12-month outcomes in six patients with no better than light perception. All patients visualized a bright light on the ceiling and a dark line on the floor after the surgery and improved in Goldman visual field tests. One patient had postoperative elevated intraocular pressure and another experienced a detached choroid following surgery.5 None of the patients had wound dehiscence, endophthalmitis or retinal detachments.
 The FDA approved the Argus II in February 2013; currently, 13 centers across the US perform the surgery.
The FDA approved the Argus II in February 2013; currently, 13 centers across the US perform the surgery.
• Telescopic IOLs. These IOLs can magnify retinal images onto viable peripheral retinal locations in patients with low vision. There are three types of telescopic implants:
– IOL VIP Revolution (Veni Vidi). Billed as "the IOL for visually impaired people," this device magnifies images at the macula. Two lenses are inserted into the capsular bag, with a plastic ring providing support.6 Available in the UK and Ireland, the Revolution is not approved yet for use in the US.
– IMT (VisionCare). The Implantable Miniature Telescope "enlarges images in front of the eye approximately 2.2 or 2.7 times their normal size," according to the company. Two lenses are inserted in the capsular bag through a large corneal incision and are contained within a small tube mounted on a circular disc. Inform patients that they may lose peripheral vision, as the lens is implanted in only one eye, whichever is worse.6 The IMT is approved for use in the US.
– iolAMD (London Eye Hospital Pharma). Also known as the Hubble implant, the iolAMD is made up of two small lenses that are injected into the eye through a microincision. The lenses are implanted in whichever eye is worse. It takes one to two months for the lenses to improve a patient's vision.7 It's not yet available here, but the US is in talks with the UK manufacturer to obtain clinical data on the device.
Make sure patients know their vision will not be corrected like in cataract surgery, and they will need glasses for both near and distance vision. They'll still need low vision aids to assist them, as well.6 The future is bright, with potential for new technology to make the lives of patients with low vision easier.
Drs. Karpecki and Shechtman have no direct financial interest in any of the products mentioned.
1. Kamao H, Mandai M, Okamoto S., et al Characterization of human induced pluripotent stem cell-derived retinal pigment epithelium cell sheets aiming for clinical application. Stem Cell Reports. 2014 Jan 23;2(2):205-18.
2. RIKEN Center for Developmental Biology. First RPE cell sheet graft transplant. Available at: www.riken.jp/en/pr/topics/ 2014/20140912_1/. Accessed October 6, 2014.
3. University of Michigan Kellogg Eye Center. Argus II Retinal Prosthesis System: Retinal implant for individuals with late-stage retinitis pigmentosum. Available at: www.kellogg.umich.edu/ argusii/. Accessed November 21, 2014.
4. Ahuja A, Dorn JD, Caspi A et al. Blind subjects implanted with the Argus II retinal prosthesis are able to improve performance in a spatial-motor task. Br J Ophthalmol. 2011;95(4):539-43.
5. Rizzo S, Belting C, Cinelli L. Am J Ophthalmol. The Argus II Retinal Prosthesis: 12-month outcomes from a single-study center.2014 Jun;157(6):1282-90. Published online ahead of print February 19, 2014.
6. Luff AJ. Understanding telescopic lens implant surgery. Available at: www.sapphire-eyecare.co.uk/iol/understanding.htm. Accessed November 21, 2014.
7. iolAMD website. iolAMD Frequently Asked Questions. Available at: http://iolamd.com/faq-patients.php. Accessed November 21, 2014.

