
A 38-year-old white female presented as a new patient seeking a second opinion about her recently diagnosed glaucoma. Her last doctor prescribed Travatan (travoprost, Alcon) one drop O.D. h.s. Her systemic meds included Alesse (levonorgestrel/ ethinyl estradiol, Wyeth) and Singulair (montelukast, Merck). She reported no allergies to medications.
She explained that she was initially worked up for glaucoma because, about a year before, her intraocular pressure measured in the mid-50s O.D. and in the teens O.S. She was initially treated with Cosopt (dorzolamide/timolol, Merck) one drop O.D. b.i.d.
Approximately eight months ago, her IOP in the left eye increased to 32mm Hg, and she was told to begin therapy with Cosopt in this eye as well. However, therapy was discontinued in her left eye approximately one month later, and she was switched to the Travatan q.h.s. in the right eye only. She reported no family history of glaucoma or any antecedent trauma.
Diagnostic Data
Entering visual acuity was 20/20 O.U. through low myopic astigmatic correction. Refraction was non-contributory. Pupils were equal, round and reactive to light and accommodation with no afferent pupillary defect. Color vision was also normal. Applanation tensions were 15mm Hg O.D. and 14mm Hg O.S. at 3:45 p.m.
Angles were grade IV by von Herrick estimation, and gonioscopy confirmed that the angles were open to the ciliary body with minimal trabecular pigmentation O.U. Pachymetry readings were 548m O.D. and 565m O.S. Initial threshold fields were normal O.U.
Cup-to-disc ratios were 0.55 x 0.65 O.D. and 0.30 x 0.35 O.S. The neuroretinal rim in the right eye did not respect the ISNT (inferior-superior-nasal-temporal) rule, as the inferior rim was quite thinned from 6 to 8 oclock. The neuroretinal rim in the left eye was plush and well perfused, and it respected the ISNT rule. Her macular, vascular, peripheral retinal and vitreal evaluations were all normal.
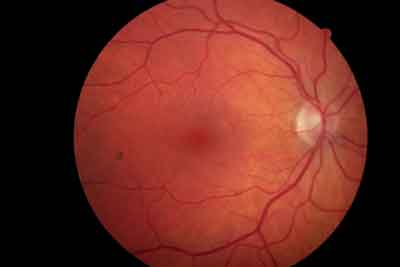 |
| The neuroretinal rim was thinned, from 6 to 8 oclock, in the patients right eye. |
Because this was my first visit with the patient, I explained to her the nature of glaucoma at length and discussed her particular findings. I told her that I believed she had glaucoma in her right eye and was probably developing it in the left. I told her to continue using Travatan q.h.s. O.D. only and to come back in two months for optic nerve analysis and a repeat pressure evaluation.
Discussion
This case brings up several patient management points we must consider, especially when seeing a patient for a confirmatory consultation. First, consider that glaucoma can be very difficult to accurately diagnose, especially in the early stages. This is especially true when clinical findings are slightly outside what we would consider normal. A confounding factor: At the completion of the first visit, we know very little about the overall stability (or instability) of the patients glaucomatous situation.
Overall stability of the glaucoma patient is critical in determining whether your treatment plan is successful. If the optic nerves remain entirely stable, visual fields show no progression, and IOPs are at target levels and remain so, then your treatment plan is adequate in staving off further vision loss and/or optic nerve damage.
In this case, I knew the patients previous eye-care provider, and I consider this doctor to be a well-seasoned, precise clinician. Because this doctor saw the patient over several visits, I decided not to change her therapy unless (or until) I determined that she was not clinically stable. This, of course, can take several months.
Determining stability of visual fields is relatively easy and objective. Current technology allows us to monitor fields over time, and various imaging systems can give us objective measurements of the optic nerve to determine stability.
IOP, however, may or may not appear stable over several visits, but the initial reference point is critical for determining clinical stability. In a particular patient, for example, IOP may never deviate by more than 3mm Hg from, say, 18mm Hg. But that doesnt necessarily mean that the patient is clinically stable. In this example, how do you know that 18mm Hg is an adequate reference point to ultimately determine stability?
In practice, we call this reference point our target pressure. Have we met our target pressure? Are we consistently at our target pressure? These questions are easy to answer.
But setting the target pressure is the hard part. Ideally, the target pressure is the pressure at which no further visual field or optic nerve damage occurs. Actually, target pressure should be the highest pressure at which no further damage occurs, so that we do not over-medicate or over-treat our patients. Setting and achieving an excessively low target pressure certainly assures a very low probability of progression, though perhaps at the cost of excessive therapy needed to stabilize the patient.
So how do you set a target pressure? In my opinion, there are three primary and two secondary factors, although opinions vary. The three primary determinants are extent of optic nerve damage, extent of visual field loss and central corneal thickness (CCT). Generally, the greater the nerve damage and field loss and the thinner the CCT, the lower the target IOP.
The secondary determinants are pretreatment IOP and the patients age. Age plays a role, especially in younger patients who have glaucoma (as in the present case). Younger patients usually require more aggressive therapy and lower target IOPs to stabilize their condition over decades of therapy.
Of course, target pressures will always be somewhat on a sliding scale, as there is no one set way to establish a target pressure. And, target pressures must be reassessed periodically and adjusted.
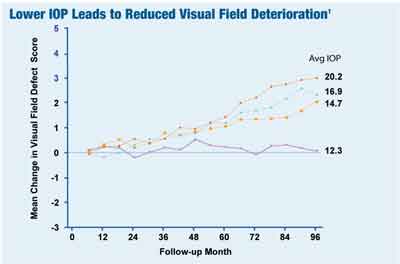 |
| In the Advanced Glaucoma Intervention Study, a low target pressure (12mm Hg) was enough to stave off further damage in glaucoma patients with advanced disease. |
The Advanced Glaucoma Intervention Study (AGIS) has also given us valuable information for determining the target pressures for most glaucoma patients.1 If you look at target pressure as a barometer of preventing devastating vision loss, then the data from AGIS can shed some light on actual clinical values for optic nerve stabilization. If you think about preventing optic nerve damage in the worst possible set of circumstances (patients in the AGIS study had advanced neuroretinal rim loss and advanced visual field loss), then you can see that the consistent IOP average of 12mm Hg in this study was adequate to stave off further damage in this patient population with advanced disease.1 (Take note that in the AGIS study, the value of 12mm Hg was an absolute IOP value and not modified by a CCT correction factor.)
Accordingly, patients who have moderate amounts of nerve and field damage may be adequately managed with target pressures in the mid-teens, while patients who have minimal damage and field loss may be adequately managed with target pressures in the upper teens or even low 20s. Of course, there will be individual variations in setting the target IOP, but AGIS at least gives us a starting point.
While we can determine stability of visual fields and optic nerve characteristics by objective measurements, we subjectively set the benchmark by which we measure intraocular pressure. Setting this target IOP early and reassessing it regularly assures us that our patients IOPs are on the mark.
1. The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol 2000 Oct;130(4):429-40.
Vol. No: 143:10Issue:
10/15/2006

