11th Annual Pharmaceuticals ReportFollow the links below to read other articles from annual update on pharmaceuticals: One Size Won't Fit All: Treating Ocular Infection Sizing Up Anti-inflammatories in Dry Eye Disease Protocols and Pitfalls of Topical Steroid Use |
While systemic medications are often necessary for the patient’s long-term health, ocular side effects, as minor as dry eye and as serious as macular toxicity, can challenge the treatment process, often leading to modification or even discontinuation of the medication.
Optometrists must be prepared to manage and comanage patients who present with concurrent medication use and ocular concerns. Here, we discuss some of the commonly prescribed systemic medications with serious ocular side effects, and what to look out for.
Hydroxychloroquine
In the United States, Plaquenil (hydroxychloroquine, Sanofi-Aventis) is commonly used to treat rheumatic conditions such as rheumatoid arthritis, systemic lupus erythematosus and Sjögren’s syndrome. Plaquenil use has the potential risk of retinal toxicity.1 Overall, the incidence is low; however, risk depends on the length of therapy and cumulative dosage. After five years of use, the risk of toxicity is <1%, but increases to 2% after 10 years and nearly 20% after 20 years.1 The underlying pathophysiology of the toxicity is not well understood, although researchers do know that Plaquenil affects the metabolism of retinal cells, including the photoreceptors.2 Since the toxicity predominately affects the macula, light absorption, cone metabolism or both may play a role.3Hydroxychloroquine binds to melanin, which concentrates in the retinal pigment epithelium and prolongs the effects, even after discontinuation of the medication. Tamoxifen use and renal disease can also result in increased risk.
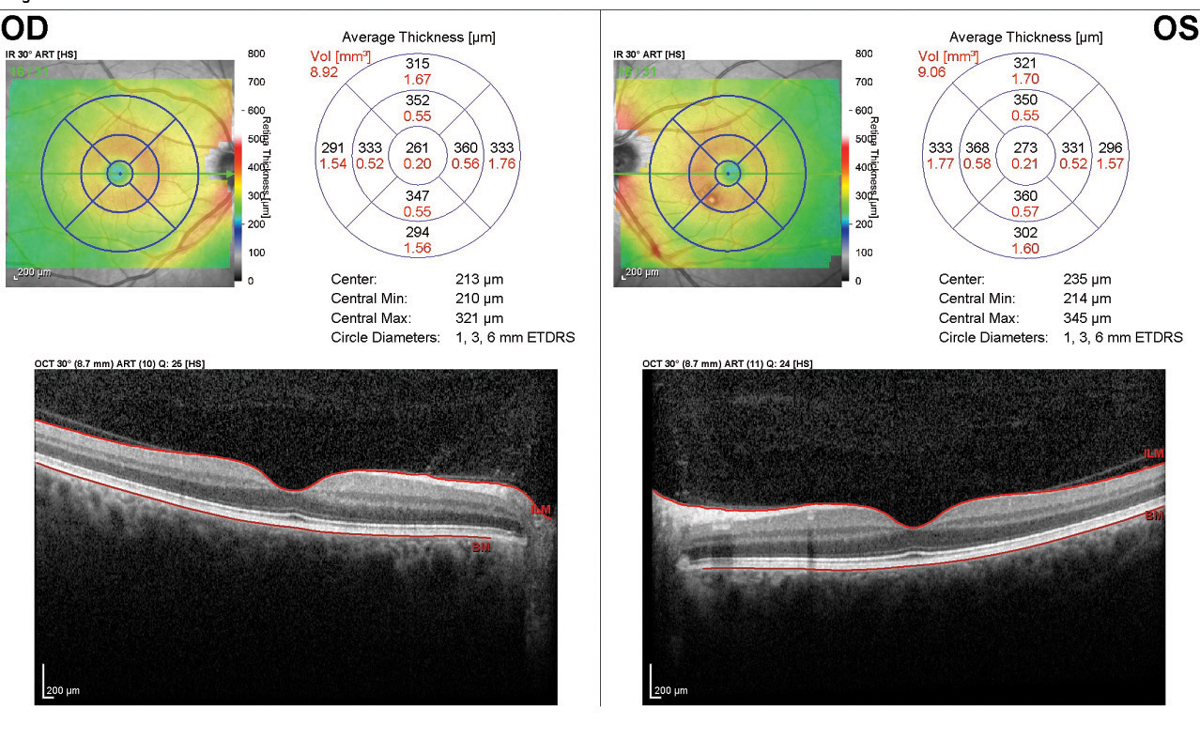 |
| This SD-OCT of a patient taking 400mg of Plaquenil daily shows no signs of maculopathy, as each eye’s inner/outer segment line is intact. Click images to enlarge. |
Optometrists must screen patients taking Plaquenil for early functional and structural changes associated with toxicity, because once a clinically evident bull’s eye maculopathy is present, the damage cannot be reversed.3 The current American Academy of Ophthalmology (AAO) recommendations advise screening with objective test for functional retinal damage—such as spectral-domain (SD) optical coherence tomography (OCT), fundus autofluorescence or multifocal electroretinogram (ERG)—in addition to a dilated fundus exam and 10-2 visual fields.1 Clinicians should use wider visual field test patterns (24-2 or 30-2) for patients of Asian descent, as research shows this patient population has early damage in a more peripheral, rather than paracentral, pattern.1
High-resolution OCT can detect localized thinning in the parafoveal region, and loss of the inner/outer segment line, known as the ellipsoid zone, on SD-OCT is considered one of the earliest signs of potential toxicity. Fundus autofluorescence abnormalities have been reported prior to visual field loss and early photoreceptor damage will show an increase in autofluorescence due to outer segment debris accumulation.2 Multifocal ERG produces local responses across the posterior pole and will show paracentral ERG depression in early Plaquenil toxicity.
The medication dosage is also an important factor in monitoring for toxicity. Plaquenil is typically prescribed as 200mg or 400mg daily. Clinicians should use real weight rather than ideal weight when calculating dosage because ideal weight can cause an overdose in thin individuals.1 The target dosage is usually less than or equal to 5mg/kg of real weight. Because the cumulative dose poses the highest risk of retinal toxicity, the AAO’s recommendations call for a baseline examination at the initiation of therapy and then annual screening after five years of treatment for low risk patients.1-3 More frequent screening is recommended for high-risk patients, including those on a higher dose and those with renal or liver disease.1
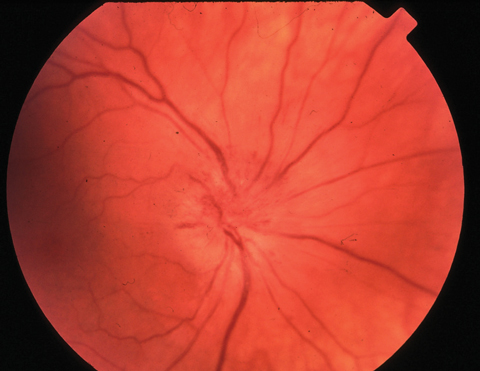 |
| NAION, seen here, has been linked to the use of PDE 5 inhibitors. |
Topiramate
This sulfa-derived monosaccharide is an effective anti-seizure medication that enhances the action of gamma-aminobutyric acid, decreases the action of glutamate and blocks sodium channels. It is also a common treatment option for migraine and any number of other conditions such as bipolar, post-traumatic stress and obsessive-compulsive disorders.4
ODs must carefully monitor patients taking topiramate, as it can cause ciliary body effusion, which relaxes the zonules and allows lens thickening, resulting in a myopic shift of as much as -9.00D.5,6 It can also cause anterior rotation of the ciliary processes, possibly inducing appositional acute angle-closure glaucoma.7,8 Topiramate-induced angle-closure typically occurs within the first two weeks of starting the medication or within hours of doubling the dose.6
The incidence of myopia and acute angle-closure with topiramate use is a well-recognized adverse reaction, but the true incidence is generally thought to be low. A 2004 study cited only 86 cases of topiramate-associated acute angle closure and 17 cases of acute bilateral myopia in the literature at the time.9 Since 2004, fewer than 50 additional case reports have been published.10
B-scan ultrasound, ultrasound biomicroscopy and anterior segment OCT can reveal the anatomic changes that create this reaction.
Most ocular issues associated with topiramate resolve after discontinuing the medication. Oral hypotensives can help control intraocular pressure, and cycloplegics can help retract the ciliary processes.
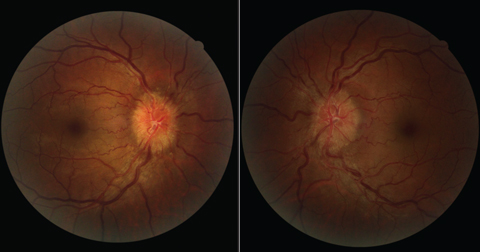 |
| Six weeks after starting isotretinoin for the treatment of acne, this patient with increased intracranial hypertension presented with bilateral optic disc edema. Click images to enlarge. |
Phosphodiesterase Inhibitors
The active ingredient in erectile dysfunction drugs, phosphodiesterase 5 (PDE 5) inhibitors block the predominant metabolizer of cyclic guanosine monophosphate that stimulates the smooth muscle relaxation necessary for erection.
The most common ocular complaint with PDE 5 inhibitors is a bluish tinge or haze to vision along with increased light sensitivity. Other ocular side effects include decreased color vision, blurred or transitory decreased vision with central haze, changes in light perception, transient ERG changes, conjunctival hyperemia, ocular pain, mydriasis (may be an emotional effect) and retinal vascular occlusions and subconjunctival hemorrhages, which may be secondary to exertion.
Ischemic optic neuropathy (ION) is the most concerning potential side effect. Although infrequent (3% of cases) in those taking low doses (25mg to 50mg), incidence can reach 11% in those taking 100mg and 50% at 200mg.11 Symptoms begin 15 to 30 minutes after taking the medication and peak at 60 minutes. Research suggests ION has a temporal association with PDE 5 inhibitors, but this remains controversial.12 For one, many patients taking these medications also have risk factors for ION, and the vascular risk factors for nonarteritic anterior ION (NAION) often overlap with vascular risk factors for erectile dysfunction.
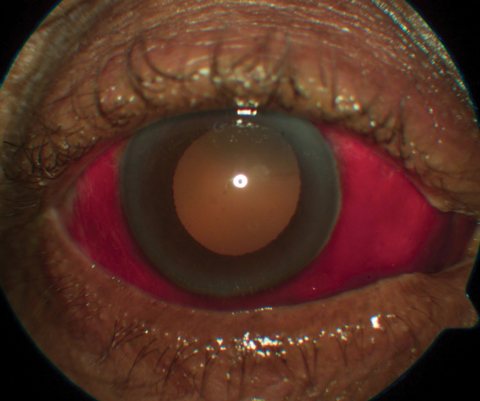 |
| This patient being anti-coagulated with warfarin presented with subconjunctival hemorrhage. Click image to enlarge. |
Fingolimod
Gilenya (fingolimod, Novartis), the first oral treatment for relapsing-remitting multiple sclerosis (MS), is effective in reducing the number of relapses and improving overall disability progression over long-term follow up.13 Fingolimod is an immunomodulating agent that binds to sphingosine-1-phosphate (S1P) receptors on the lymphocytes and prevents lymphocyte release from the lymph nodes, reducing lymphocyte migration into the central nervous system.14 This spares the system from attack by these myelin-reactive lymphocytes.
As sphingolipids are the third most numerous lipid in the retina, fingolimod-associated macular edema (FAME) is a possible side effect, reported in two pivotal studies that investigated the drug’s safety and efficacy.13,15 S1P receptors in retinal blood vessel endothelial cells are responsible for maintaining cell adhesion complexes, so when fingolimod downregulates the S1P receptors, it also downregulates adhesion complexes and increases retinal vascular permeability, resulting in macular edema.16 In one study, 0.5% of patients receiving 0.5mg of daily fingolimod and 1.0% of those receiving 1.25mg of daily fingolimod developed macular edema.15
Most patients who develop FAME do so within four months of starting treatment, and it resolves after the drug is discontinued.16 Clinicians should screen patients taking fingolimod with a complete eye examination and macular OCT at baseline and again in three to four months. Clinicians should exercise caution when treating diabetic patients due to the preexisting blood-retinal barrier compromise.
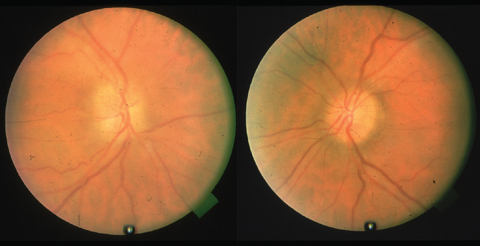 |
| This patient’s toxic optic neuropathy is due to amiodarone use. Click images to enlarge. |
Isotretinoin
An analog of vitamin A, this is used to treat severe, recalcitrant nodular acne, psoriasis and disorders of keratinization. It alters skin lipid composition, reduces sebaceous gland size and decreases sebum production, in addition to having anti-inflammatory properties.17
Isotretinoin-induced meibomian gland dysfunction leads to eye dryness, ocular surface irritation, decreased contact lens tolerance, photophobia, blepharoconjunctivitis, decreased vision and even an increase in hordeolum and chalazion frequency.18 Studies link isotretinoin to the development of secondary increased intracranial hypertension due to increased resistance to cerebrospinal fluid outflow.
Patients will often present with headaches (typically postural), blurred vision, subjective visual obscurations and tinnitus. Increased intracranial pressure can cause bilateral optic nerve head edema, which can last for weeks after discontinuing the medication and can be vision-threatening if left undetected. While a serious complication, increased intracranial pressure is rare.
 |
| This patient presented two days after bisphosphonate infusion with severe ocular inflammation. Click images to enlarge. |
Tamoxifen
This oral anti-estrogen medication is used to treat advanced metastatic breast cancer and as an adjunctive endocrine therapy following resection in early disease. Endocrine therapy works to combat steroid hormone assisted tumor growth in hormonally dependent breast cancer cells. Tamoxifen competes with estradiol for receptors in breast, uterine and vaginal tissues as well as in tumors containing high concentrations of estrogen receptors.19
Ocular toxicity from tamoxifen presents as a crystalline retinopathy with yellow-white refractile bodies confined to the plexiform and nerve fiber layers, typically found in the macular and paramacular areas. Sometimes, it can be associated with macular edema and a whorl-like superficial keratopathy.20 Ocular toxicity is typically associated with high doses in excess of 120mg daily; it is not common with the standard daily dose of 20mg.21,22
SD-OCT imaging helps to detect subtle maculopathy associated with tamoxifen use. Low dose usage, for example, can produce pseudo-cystic cavities in the macula, which do not leak on fluorescein angiography and do not increase retinal thickness.22
Research suggests Müller cell dysfunction contributes to tamoxifen ocular toxicity because the medication inhibits the glutamate-aspartate transporter present in Müller cells.23 Excess glutamate, in turn, may cause Müller cell dysfunction and neuronal apoptosis, creating cavitary spaces.24 One study found that full-thickness macular holes were five times more likely in patients taking tamoxifen, possibly caused by the foveal cavities.25 All patients on tamoxifen should have a baseline and yearly dilated eye examination with SD-OCT of the macula.
Ethambutol
This medication is most commonly used as part of the lengthy quadruple antibiotic regimen for the treatment of tubercuolosis (TB) and in the multi-drug therapy for mycobacterium avid complex lung disease.26 It is a metal chelator, which prevents cell wall synthesis in mycobacteria by inhibiting arabinosyl transferase.27
Ethambutol is known to cause optic neuropathy characterized by bilateral central vision loss, color vision loss, central or cecocentral visual field scotomas and eventual optic atrophy. The exact mechanism is unknown, but researchers theorize it is due to either decreased mitochondrial copper, which is necessary for oxidative phosphorylation, or the chelation of zinc, which inhibits lysosomal activation.28,29
Toxicity is dose related with an incidence of 18% with more than 35mg/kg/day, 5% to 6% with doses of 25mg/kg/day, 3% with 20mg/kg/day and less than 1% at less than or equal to 15 mg/kg/day.30
Between 30% and 64% of patients will show some visual recovery if the optic neuropathy is detected early and the medication is discontined.27 Patients should have a baseline exam with vision screening, color vision, Amsler grid and visual field testing, in addition to a dilated fundus exam and nerve fiber layer OCT. They should be re-examined monthly while on the ethambutol therapy.
Tamsulosin
Flomax (tamsulosin, Boehringer Ingelheim Pharmaceuticals) is a selective α1A-adrenergic receptor antagonist used to treat benign prostate hyperplasia by relaxing smooth muscles in the neck of the bladder and in the prostatic urethra. These receptors are also found on the iris dilator muscle, leading to ocular effects of the medication. Selective α1A antagonists are known to cause intraoperative floppy iris syndrome (IFIS) during cataract extraction and a likelihood of iris prolapse through the surgical wound.31 The exact mechanism of IFIS is unknown, although one study suggests chronic blockade of the α1A-adrenergic receptors can decrease smooth muscle tone and cause diffuse atrophy of the iris dilator.32 The researchers found histologic evidence of decreased iris dilator muscle thickness in cadaver eyes with a history of tamsulosin use compared with cadaver control eyes.32 Surgeons and comanaging ODs must be aware that IFIS may still occur several years after discontinuing the drug, and stopping tamsulosin preoperatively is of uncertain benefit.
Atrial Fibrillation Agents
Several vascular medications come with significant ocular effects ODs must be on the lookout for:
Amiodarone is an effective anti-arrhythmic medication for ventricular tachycardia and fibrillation and atrial fibrillation. It blocks calcium, potassium and sodium channels in cardiac tissue, which prolongs the cardiac action potential and refractory period. It is also a weak beta blocker and vasodilator.
Ocular side effects of amiodarone include corneal verticillata or whorl-like keratopathy, which occur in 70% to 100% of patients. This keratopathy is related to the dose and duration of treatment, generally appears after at least one month of therapy and will resolve upon discontinuation of the medication. Amiodarone can also cause anterior lenticular subcapsular opacities in 50% to 60% of patients. Both the corneal and lenticular deposits are typically asymptomatic.33
Amiodarone has also been linked to optic neuropathy, with a clinical presentation similar to NAION. Amiodarone-induced optic neuropathy should be a diagnosis of exclusion, considering most patients taking amiodarone have vascular risk factors already increasing their NAION risk. One report found an incidence of amiodarone-induced optic neuropathy at roughly 2% compared with a general incidence of 0.3%.34 It has an insidious onset, milder degree of vision loss, longer duration of disc edema and is more commonly bilateral than NAION.35 Amiodarone has a long half-life, averaging 58 days, but up to 142 days, and accumulates in the lysosomes of multiple tissues, including the optic nerve, so optic disc swelling can persist for months after discontinuing the medication.36,37
Digoxin inhibits the cell membrane Na+/K+-ATPase, creating an increase in intracellular calcium and sodium and extracellular potassium. This leads to stronger myocardial contraction and excitability and a decrease in the conduction and depolarization velocity in the atrioventricular node, which resets the rhythm of the heart rate. Digoxin is a second-line treatment for atrial fibrillation and in severe symptomatic systolic heart failure.38
Approximately 95% of patients in the toxic range will report ocular symptoms, the most common being xanthopsia, or yellowing vision.39 Cyanopsia (blue vision) and chloropsia (green vision) have also been reported.40 Other ocular symptoms include flashing, scintillating or moving lights or spots, blurred, hazy, misty or snowy vision and difficulty reading. Clinical ocular finding include decreased visual acuity, central scotoma, color vision defects and delayed b-wave time and decreased b-wave amplitude on ERG. While still under debate, most presume ocular symptoms are related to Na+/K+ ATPase inhibition.38 Symptomatic patients should be referred back to their cardiologist for monitoring.
Anticoagulants are used to help prevent clotting in a wide variety of systemic disorders. The ocular and systemic side effects occur with varying frequencies depending on the specific medication. Platelet inhibitors such as clopidogrel, dipyridamole and aspirin are effective but with a lower incidence of side effects.
Warfarin is a potent oral anticoagulant that poses a high risk of bleeding disorders. It inhibits vitamin K-dependent gamma-carboxylation of coagulation factors II, VII, IX and X. Warfarin can be affected by other medications, as well as a patient’s dietary intake of vitamin K. Because of its narrow therapeutic window, warfarin requires frequent monitoring via laboratory testing.41
Direct-acting oral anticoagulants (DOACs) are used to treat patients with venous thromboembolism and nonvalvular atrial fibrillation.42 Research shows DOACs are as effective as warfarin in preventing stroke due to atrial fibrillation while also having a lower overall bleeding risk, especially intracranial bleeding. In addition, they are less affected by diet and medication interactions.43
Atrial fibrillation agents all carry the risk of increased bleeding. The most worrisome is the potential for intracranial hemorrhage. Ocular side effects of anticoagulants include subconjunctival hemorrhage, retinal hemorrhage, orbital hemorrhage causing mass effect and exacerbation of bleeding from other ocular conditions such as proliferative diabetic retinopathy and exudative macular degeneration.
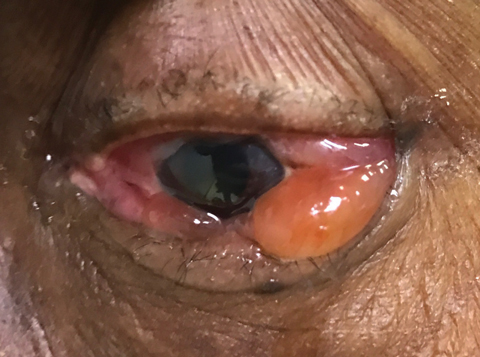
Bisphosphonates
These are widely used in the treatment of osteoporosis, Paget’s disease, metastatic bone disease, multiple myeloma and hypercalcemia of malignancy. Bisphosphonates are rapidly cleared from circulation and are absorbed into bone mineral surfaces, where they slow osteoclasts to allow osteoblasts to rebuild bone.44
The most common ocular side effect of bisphosphonates is a self-limiting conjunctivitis.45 Uveitis and scleritis, two serious possible ocular side effects, might not resolve unless the medication is discontinued.45 One study found scleritis and uveitis were rare in bisphosphonate users and that 43% of the patients who developed either condition had a systemic condition already associated with uveitis or scleritis.46 Researchers can only speculate that bisphosphonates use might be a precipitating factor for ocular inflammation in patients already prone to these conditions.44 Patients who develop serious ocular inflammation need oral and topical anti-inflammatories and often require discontinuation of biphosphonates for resolution.
Ocular side effects from systemic medications can be serious and sight threatening. It is crucial for ODs to catalogue each patient’s systemic medications, discuss the possible side effects and monitor appropriately. Often, additional testing is warranted to screen for effects and detect early toxicity.
Dr. Hunter is an assistant professor of ophthalmology at Loyola University Medical Center in Maywood, IL. She is the current national chair of the American Academy of Optometry’s admittance committee.
Dr. Marciniak works at the Jesse Brown VAMC, teaches at Illinois College of Optometry and works in private practice. She is a member of the Advanced Competency in Medical Optometry committee.
1. Armor MF, Kellner U, Lai TY, et al. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology. 2016;123(6):1386-94. |

