Ocular Surface HealthCheck out the other feature articles in this month's issue:- Red Eye Remedies: New and Tried-and-True - How to Answer the “Why?” of Dry Eye - Artificial Tears: What Matters and Why - A Modern Approach to Meibomian Gland Dysfunction (earn 2 CE credits) |
Lid wiper epitheliopathy (LWE) was first proposed as a contact lens (CL) friction issue in 2002. The undersurface of the upper eyelid was named the “lid wiper,” as it spread the tear film over the ocular surface with every blink, much like the windshield wiper does on an automobile.1 It is now generally accepted that any lid-to-surface friction can cause LWE.
LWE is visualized by everting the eyelid after an ophthalmic vital dye has been instilled and the area proximal to the eyelashes has been clinically inspected (Figure 1).2 An observable line at the mucocutaneous junction, called the line of Marx, is present in all eyelids, and any further staining of the tissue in the palpebral marginal conjunctiva is evidence of LWE (Figure 2).2
Because of the blinking mechanism of the eyes, LWE can be observed in both CL wearers and non-lens wearers, especially when a patient has complaints of dryness without typical clinical signs.3,4 It has also been proposed that LWE is a sign of subclinical inflammation that could exacerbate the conundrum of dry eye progression.1,4 Consistently and accurately assessing LWE is fast becoming an important component of dry eye disease (DED) identification and treatment.
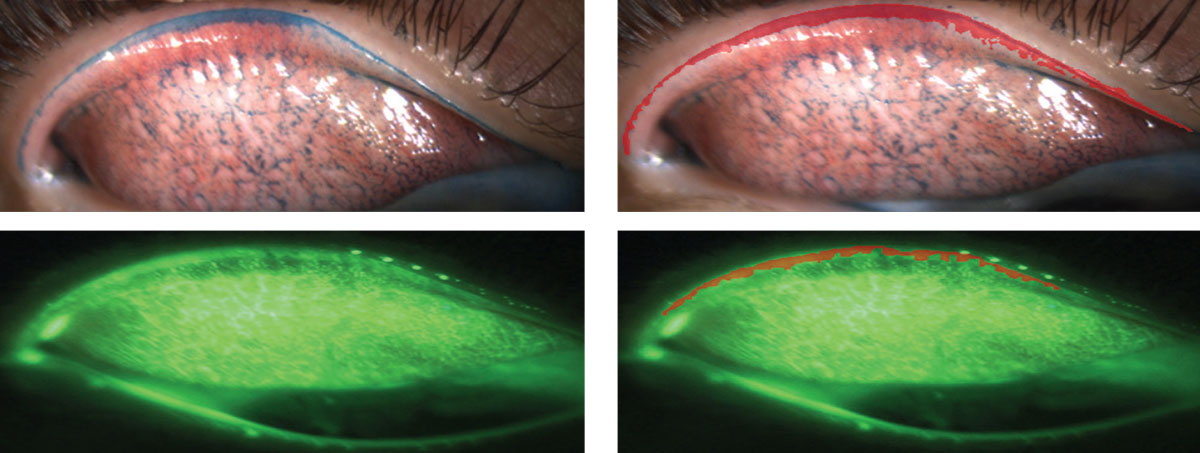 |
| Fig. 1. All images are of everted upper lids. Images on the left use lissamine green and sodium fluorescein, respectively. See the areas colored in red on the right that outline what is considered to be lid wiper epitheliopathy. Click image to enlarge. |
A Multifactorial Condition
No matter the underlying etiology, DED can result in an inadequate, unstable or otherwise defective tear film. Proper lubrication from a healthy tear film prevents the sheering stress believed to cause LWE.3,5 As such, it is not surprising that LWE is reported to be found in almost 90% of patients with known dry eye issues.4 Compromise to the epithelium of the wiping system can result in symptomatology akin to that of DED.3,6-8
A healthy tear film is vital in the management of LWE and its associated symptoms. The disruption of any of the tear film components can cause decreased lubricity, leading to increased friction between the eyelids and the ocular surface. The tear film must also be thick enough to mask any ocular surface irregularities, and all types of DED can exacerbate LWE through resultant friction-related damage.5
Meibomian gland dysfunction (MGD) and LWE are often diagnosed together.9 Atrophied or congested meibomian glands do not produce or excrete adequate lipid into the tears. This leads to altered tear viscosity, reduced tear break-up time, and a thinner tear layer.
Severe MGD can cause tear saponification with inspissated, clogged and capped gland orifices. Altered tear composition oftentimes leads to a hyperosmolar condition, which can further damage the ocular surface and the lid wiper region.10 Hyperosmolar tears are also implicated in goblet cell damage or destruction, and, a lack of secreted mucins could lead to problems with lubricity.5,11 Any of these factors, and more likely a combination of these factors, can lead to dryness, discomfort, epithelial changes, and concurrent LWE.
Contributing Factors
LWE can be present on both the upper and lower eyelids, though each is likely due to differing etiologies. Upper LWE has been reported to be associated with greater patient discomfort and symptomatology; whereas, the lower lid LWE has not.9,10,12,13 Multiple theories have been proposed to explain this disparity. It is possibly due to differences in lid anatomy. For example, there are more meibomian glands present in the upper lid than the lower lid, which can lead to a higher prevalence of MGD in the upper lid.9 One study postulated that upper LWE is caused by decreased tear volume and/or viscosity, while lower LWE is attributed to altered tear osmolarity and increased contact time of the tears on the lower lid surface.11
A major contributing factor to LWE is the anatomy of the blink itself, as the upper lid travels across a greater surface area during a typical blink.5 Greater contact time between the upper lid and the dry or irregular ocular surface could result in more epithelial damage and increased patient discomfort. The repeated movement of the lower lid along the nasal bulbar conjunctiva may be related to higher lid-parallel conjunctival fold (LIPCOF) scores, which correlate with increased LWE due to shared related mechanisms of friction.14-16
LIPCOFs are small visible folds in the inferotemporal and inferonasal quadrants of the bulbar conjunctiva, parallel to the lower lid margin.17 Their presence may relate to increased the shear forces between the bulbar conjunctiva and palpebral conjunctiva of the eyelid during blinking.18 Shear forces due to friction may be the root cause of LWE, particularly in cases of DED and those with CL discomfort.
Lid anatomy changes as a factor of age and with differing demographics. The prevalence of LWE in the upper and lower lids decreases with increasing age, due to changes in lid laxity that alter the shearing forces of blinks.16 There is also a higher prevalence of LWE in Asians, which may be explained by lid anatomy and meibum composition differences between ethnicities.10 As with DED, there are multiple contributing factors that are not distinctly separate.
Proposed Mechanisms
The studies that demonstrated increases in LWE in CL patients proposed several possible, and likely interrelated, mechanisms contributing to damage of the stratified squamous epithelial cells that make up the lid wiper. Based on current theories, a combination of mechanical shearing forces during the blink and decreased tear film stability can result in LWE in CL wearers.1,14 It has been widely demonstrated and accepted that CL wear will alter the normal biochemical structure of the tear film and affect the rate of evaporation.19-24
When comparing symptomatic and asymptomatic soft CL wearers, the mucin, MUC5AC, significantly decreased in symptomatic patients, and the study concluded CL wearers with dry eye symptoms had decreased mucin concentrations at the ocular surface.14 Increased tear film evaporation resulting from incomplete blinks accelerates the precipitation of deposits on the lens surface.25 An incomplete blink approximately doubles the interblink interval and tear evaporation time for the inferior corneal surface or CL, and deposits are more easily precipitated on a CL anterior surface when the pre-lens tear layer thins or evaporates.25
The frictional forces that are the suspected underlying mechanism of LWE are, therefore, the greatest during a complete blink following an incomplete blink, as the fluid layer between the lens and lid wiper is the thinnest and provides less lubrication than between full blinks.25 After the tear film has evaporated in this area, a concentration of tear constituent remains and increases the tear osmolarity. However, a study exploring tear osmolarity, comfort and LWE was unable to demonstrate a relationship among the three.26 The authors proposed that this is possibly due to lack of diagnostic equipment currently capable of testing the tear osmolarity on the surface of the lens.25
The Tear Film and Ocular Surface (TFOS) International Workshop on Contact Lens Discomfort updated the definition of CL discomfort and proposed environmental factors that play a role.27 In assessing the impact of low humidity on CL wearers, researchers demonstrated that LWE grades increased significantly in the upper and lower lids and that artificial tears did not improve the LWE when used two hours after the patient left the humidity chamber.28
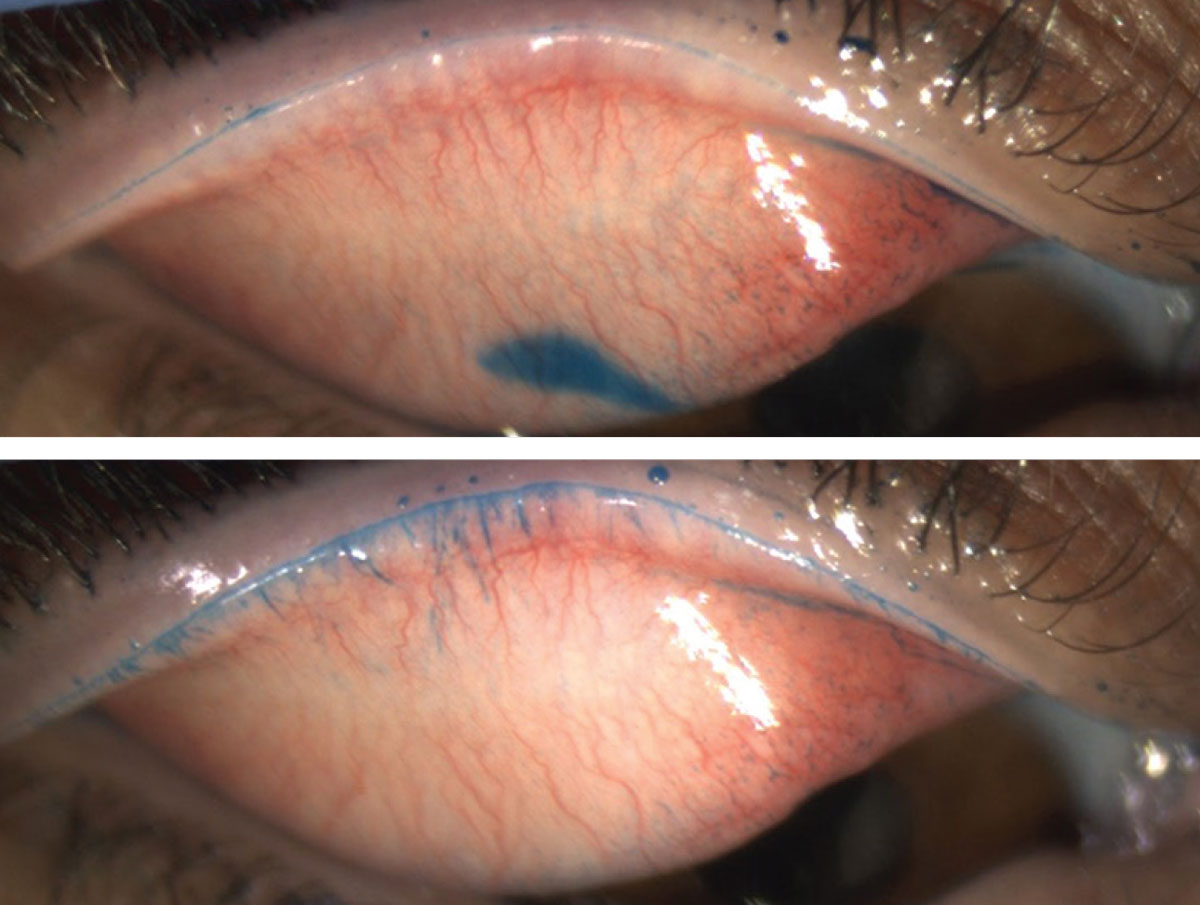 |
| Fig. 2. The image on top only reveals the line of Marx, whereas the image underneath shows additional proximal LWE staining. Click image to enlarge. |
Avoid Falling Short
Understanding the anatomy is crucial to properly identifying LWE. There are several possible reasons why LWE was not previously a focal point of conversation in research and clinical care.3 Eyelids are infrequently everted during routine anterior segment evaluations. When eyelids are everted, the lid margins are generally not inspected; instead, the attention is typically directed to the tarsus. Vital dye staining to show compromise of the tissue prior to lid eversion is uncommon in practice. Not only is vital dye staining important for visualization of LWE but timing also matters.3
Clinical inspection can influence findings when using vital dyes. One study found that a non-specific time point in making observations with sodium fluorescein (NaFl) can yield misleading results due to a reduction in the clinical presentation of LWE.29 Similarly, observation without sufficient dye instillations may not yield the full extent of LWE.
Optimal LWE Identification
An evidence-based approach with both NaFl and lissamine green (LG) dyes can reveal and identify LWE in practice.30 The proper quantity of dye and the ideal timing for clinical observation can fully uncover this condition. For both LG and NaFl, a repeat instillation of dye (two drops of 10µL LG and 2µL NaFl) is necessary to reveal the full extent of LWE staining.30 Optimal viewing time is critical with using NaFl, as clinicians need to wait five minutes if using one drop, or three to five minutes when using two drops.30
The latest TFOS DEWS II report recommends waiting three to six minutes after repeat NaFl instillation with strips when observing LWE (Figure 3).31 Similarly, research showed that when using two drops of LG, optimal observation can occur between one and five minutes. When using a strip wetted with one drop of saline observation should occur after between one and four minutes.31 A simple and
easy-to-remember clinical protocol would be to use two drops of either dye and observe the eyelid after three minutes (Table 1). Improper observation timing and inadequate dye quantity can easily mislead a clinician into prematurely concluding that LWE is not present.
Impregnated paper strips are more commonly used in clinical practice than liquid vital dyes. As such, it is nearly impossible to know the exact dye volume delivered with paper strip instillation. This is further rationale to require two instillations to ensure adequate dye delivery to the ocular tissues.
Table 1. Step-by-step LWE Identification Procedure | |
| 1. | Instill one drop of vital dye (LG or NaFl) |
| 2. | Wait one minute |
| 3. | Instill second drop of same vital dye |
| 4. | Wait three minutes |
| 5. | Evert lid and evaluate the lid margin for LWE |
Treatment and Management
To address the multifactorial mechanisms of increased LWE in CL wearers, eye care practitioners must employ a variety of weapons from their DED arsenal. The preventive treatments for LWE should augment hydrodynamic lubrication and reduce or eliminate excess trauma. Preventive measures should also counteract possible associated inflammation—assist in shear stress reduction and prevent of “dry” contact between the eyelid wiper and the CL surface.32
Recently published data on treating DED subjects with concurrent LWE compared a lipid-based nano-emulsion drop (Systane Complete, Alcon) and a non-lipid-based aqueous drop (Systane Ultra, Alcon). The study found that both treatments significantly reduced LWE.33
Lipid emulsion artificial tears and meibomian gland management can substantiate the diminished tear cushion (especially the lipid layer) between the lid and CL, steroids can reduce para-inflammation from microtrauma of the shearing forces during blinks, punctal plugs can improve the tear volume available to the ocular surface and ointment at night after lens removal can be helpful.30
Other considerations include discontinuing lens wear or changing lens materials or wear schedule.30,32,34,35 One report demonstrated an improvement in upper LWE with the use of 0.1% hyaluronic acid (HA) drops and discontinuation of lens wear.15 Others found that adding HA to standard carboxymethylcellulose (CMC) artificial tear drops improved clinical performance without additional adverse effects and should be considered in these cases.36
LWE and Contact Lenses
Some have proposed that LWE could be the missing link between CL discomfort and the lack of clinical signs.1 One study demonstrated an increase in LWE in symptomatic soft CL wearers (80%) compared with asymptomatic lens wearers (13%) that was most likely a product of the characteristics of the tear film between the lens surface and the lid wiper.1
Though LWE may occur in the absence of CL wear, many studies have shown an increase in LWE in CL wearers, including gas permeable lenses (GPs) and both hydrogel and silicone-hydrogel soft lenses.3,15,34,37 One study in particular documented and classified six slightly different patterns of LWE in silicone-hydrogel CL wearers.34 Just like corneal staining patterns can differ by patient and etiology, the same is true for LWE. Clinical examination of any staining proximal to the line of Marx on the lid margin is key, especially considering it may not yield one particular and exact pattern of staining.
Another study used imaging to study increased density of the microvascular network of the lid wiper after six hours of CL wear and demonstrated a significant correlation with CL discomfort.38
However, according to a meta-analysis, the number of studies failing to show an association between LWE and CL discomfort were roughly equal to those that did find a correlation.28
Due to the recent discovery of the lid wiper and the potential clinical impacts, further studies must be done to determine the true association between LWE and CL. Several have recommended a standardized protocol for LWE identification to better compare LWE severity across studies.28,30,35,39,40
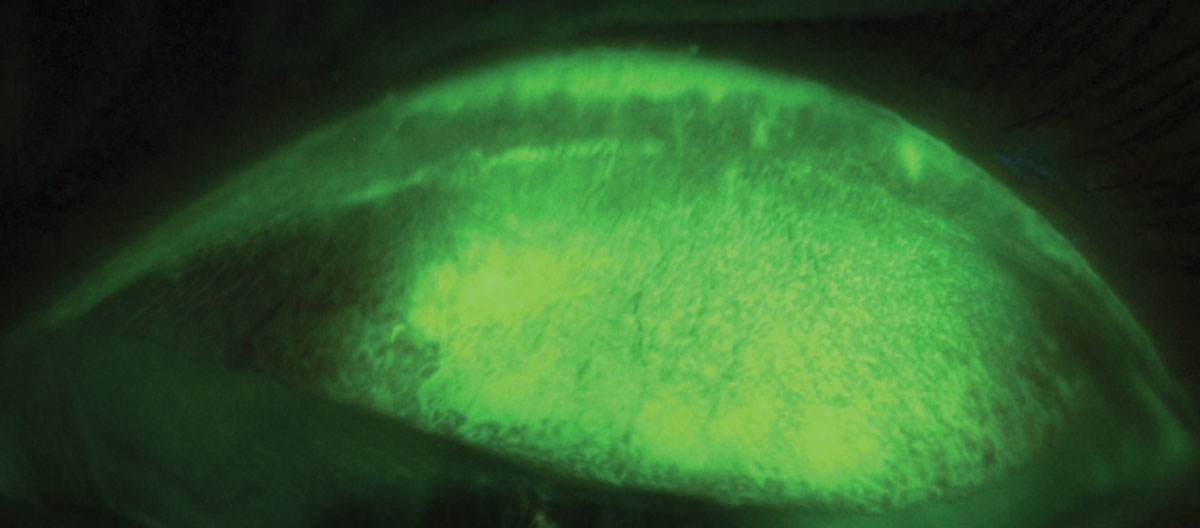 |
| Fig. 3. LWE is apparent after staining with NaFl. Click image to enlarge. |
Predicting Successful CL Wear
Novel clinical tests designed to predict success in neophyte CL wearers demonstrated a close relationship between LWE and hyperemia and between LWE and LIPCOF scores.14,41 Another study also attempted to predict success of new silicone hydrogel wearers, and the researchers noted a significant increase in LWE over six months.37 Interestingly, the latter study found that LWE was associated with tear volume but not tear meniscus height, suggesting that tear volume of the surface plays a role in decreasing friction along the lower lid margin.
Until we learn more about the role that it plays in CL discomfort, make sure to consider LWE during your clinical examinations moving forward. Any interventions that can keep CL wearers from becoming symptomatic are advantageous for a successful practice.
Microtrauma due to blinking over a non-lubricious ocular surface can exacerbate the DED and confound CL wearers. Pay close attention to the anatomy around the lid margins and the lid wiper. In order to correctly examine these tissues, carefully deliver adequate dye volume through a repeat administration to avoid premature observation and false diagnosis of the lid wiper post-dye instillation. Proper adherence to the optimal methodology to identify LWE may lead to more successful outcomes.
Dr. Lievens is a professor and chief of internal clinics of The Eye Center at the Southern College of Optometry in Memphis.
Dr. Roberts and Dr. Rayborn practice at The Eye Center.
Dr. Norgett is a senior lecturer at Anglia Ruskin University in Cambridge, England.
Dr. Briggs is a senior statistical consultant at the Mark Wainwright Analytical Centre in UNSW Sydney.
Dr. Allen is a professor of optometry and visual science at Anglia Ruskin University.
Dr. Vianya-Estopa is a senior lecturer of optometry at Anglia Ruskin University.
1. Korb DR, Greiner J V, Herman JP, et al. Lid-wiper epitheliopathy and dry-eye symptoms in contact lens wearers. CLAO J. 2002;28(4):211-6. 2. Efron N, Brennan NA, Morgan PB, Wilson T. Lid wiper epitheliopathy. Prog Retin Eye Res. 2016;53:140-74. 3. Korb DR, Herman JP, Greiner JV, et al. Lid wiper epitheliopathy and dry eye symptoms. Eye Contact Lens. 2005;31(1):2-8. 4. Korb DR, Herman JP, Blackie CA, et al. Prevalence of lid wiper epitheliopathy in subjects with dry eye signs and symptoms. Cornea. 2010;29(4):377-83. 5. Cher I. A new look at lubrication of the ocular surface: fluid mechanics behind the blinking eyelids. Ocul Surf. 2008;6(2):79-86. 6. Knop E, Korb DR, Blackie CA, Knop N. The lid margin is an underestimated structure for preservation of ocular surface health and development of dry eye disease. Dev Ophthalmol. 2010;45:108-22. 7. Varikooty J, Srinivasan S, Jones L. Atypical manifestation of upper lid margin staining in silicone hydrogel lens wearers with symptoms of dry eye. Contact Lens Anterior Eye. 2008;31(1):44-6. 8. Pult H, Purslow C, Murphy PJ. The relationship between clinical signs and dry eye symptoms. Eye. 2011;25(4):502-10. 9. Korb DR, Blackie CA. Meibomian Gland Diagnostic Expressibility: Correlation With Dry Eye Symptoms and Gland Location. Cornea. 2008;27(10):1142-7. 10. Li W, Yeh TN, Leung T, et al. The relationship of lid wiper epitheliopathy to ocular surface signs and symptoms. Investig Ophthalmol Vis Sci. 2018;59(5):1878-87. 11. Golebiowski B, Chim K, So J, Jalbert I. Lid margins: Sensitivity, staining, meibomian gland dysfunction, and symptoms. Optom Vis Sci. 2012;89(10):1443-9. 12. Siddireddy JS, Vijay AK, Tan J, Willcox M. The eyelids and tear film in contact lens discomfort. Contact Lens Anterior Eye. 2018;41(2):144-53. 13. Navascues-Cornago M, Morgan PB, Maldonado-Codina C. Lid margin sensitivity and staining in contact lens wear versus no lens wear. Cornea. 2015;34(7):808-16. 14. Berry M, Pult H, Purslow C, Murphy PJ. Mucins and Ocular Signs in Symptomatic and Asymptomatic Contact Lens Wear. Optom Vis Sci. 2008;85(10):E930-8. 15. Pult H, Purslow C, Berry M, Murphy PJ. Clinical tests for successful contact lens wear: relationship and predictive potential. Optom Vis Sci. 2008;85(10):E924-9. 16. Shiraishi A, Yamaguchi M, Ohashi Y. Prevalence of upper- and lower-lid-wiper epitheliopathy in contact lens wearers and non-wearers. Eye Contact Lens. 2014;40(4):220-4. 17. Bandlitz S, Purslow C, Murphy PJ, Pult H. Lid-parallel conjunctival fold (LIPCOF) morphology imaged by optical coherence tomography and its relationship to LIPCOF grade. Cont Lens Anterior Eye. 2019;42(3):299-303. 18. Pult H, Tosatti SG, Spencer ND, et al. Spontaneous blinking from a tribological viewpoint. Ocul Surf. 2015;13(3):236-49. 19. Craig JP, Willcox MDP, Argüeso P, et al. The TFOS International Workshop on Contact Lens Discomfort: report of the contact lens interactions with the tear film subcommittee. Invest Opthalmol Vis Sci. 2013;54(11):TFOS123. 20. Refojo M. Tear Evaporation Considerations and Contact Lens Wear. Considerations in Contact Lens Use under Adverse Conditions. Washington, DC: National Academies Press. 1991. 21. Korb DR. Tear film—contact lens interactions. In: Sullivan DA (ed). Lacrimal Gland, Tear Film, and Dry Eye Syndromes. Advances in Experimental Medicine and Biology. Vol 350. Boston, MA. Springer. 1994:403-10. 22. Guillon M, Guillon JP. Hydrogel lens wettability during overnight wear. Ophthalmic Physiol Opt. 1989;9(4):355-9. 23. Maldonado-Codina C, Efron N. Dynamic wettability of pHEMA-based hydrogel contact lenses. Ophthalmic Physiol Opt. 2006;26(4):408-18 24. Lin M, Copley KA, Radke CJ. Assessment of pre-lens tear-film stability by slit-lamp examination, placido ring reflection, IBUT and advancing contacting angles: do they correlate? Invest Ophthalmol Vis Sci. 2006;47(13):95. 25. McMonnies CW. Incomplete blinking: exposure keratopathy, lid wiper epitheliopathy, dry eye, refractive surgery and dry contact lenses. Cont Lens Anterior Eye. 2007;30(1):37-51. 26. Stahl U, Jalbert I. Exploring the links between contact lens comfort, osmolarity and lid wiper staining. Cont Lens Anterior Eye. 2018;41(1):110-6. 27. Nichols KK, Redfern RL, Jacob JT, et al. The TFOS International Workshop on Contact Lens Discomfort: report of the definition and classification subcommittee. Invest Ophthalmol Vis Sci. 2013;54(11). 28. Jones L, Varikooty J, Keir N, et al. The evaluation of lid wiper epitheliopathy in contact lens wearers in a controlled low humidity environmental exposure chamber. Invest Ophthalmol Vis Sci. 1977;54(15):5475. 29. Markoulli M, Isa NA, Papas EB. Temporal characteristics of sodium fluorescein in the tear meniscus. Optom Vis Sci. 2017;94(2):166-73. 30. Lievens CW, Norgett Y, Briggs N, et al. Optimal methodology for lid wiper epitheliopathy identification. Cont Lens Anterior Eye. May 14, 2020. [Epub ahead of print]. 31. Wolffsohn JS, Arita R, Chalmers R, et al. TFOS DEWS II Diagnostic Methodology report. Ocul Surf. 2017;15(3):539-74. 32. Lin MC, Yeh TN. Mechanical complications induced by silicone hydrogel contact lenses. Eye Contact Lens Sci Clin Pract. 2013;39(1):114-23. 33. Craig JP, Muntz A, Wang MTM, et al. Six-month therapeutic profiles of lipid and non-lipid-based artificial tear supplements in managing dry eye. Poster presented at American Academy of Optometry Meeting 2020. 34. Varikooty J, Srinivasan S, Subbaraman L, et al. Variations in observable lid wiper epitheliopathy (LWE) staining patterns in wearers of silicone hydrogel lenses. Cont Lens Anterior Eye. 2015;38:471-76. 35. Schulze M-M, Srinivasan S, Hickson-Curran SB, et al. Lid wiper epitheliopathy in soft contact lens wearers. Optom Vis Sci. 2016;93(8):943-54. 36. Nichols JJ, Lievens CW, Bloomenstein MR, et al. Dual-polymer drops, contact lens comfort, and lid wiper epitheliopathy. Optom Vis Sci. 2016;93(8):979-86. 37. Best N, Drury L, Wolffsohn JS. Predicting success with silicone-hydrogel contact lenses in new wearers. Cont Lens Anterior Eye. 2013;36(5):232-7. 38. Deng Z, Wang J, Jiang H, et al. Lid wiper microvascular responses as an indicator of contact lens discomfort. Am J Ophthalmol. 2016;170:197-205. 39. Varikooty J, Lay B, Jones L. Optimization of assessment and grading for lid wiper epitheliopathy. Optom Vis Sci. 2012:88. 40. Muntz A, Subbaraman LN, Craig JP, Jones L. Cytomorphological assessment of the lid margin in relation to symptoms, contact lens wear and lid wiper epitheliopathy. Ocul Surf. 2020;18(2):214-20. 41. Pult H, Murphy PJ, Purslow C. A novel method to predict the dry eye symptoms in new contact lens wearers. Optom Vis Sci. 2009;86(9):E1042-50. |

