Anterior ischemic optic neuropathy (AION) is commonly referred to as a stroke of the optic nerve. There are two types of AION: arteritic (AAION) and non-arteritic (NAION). Vision impairment secondary to AION often includes both visual field and visual acuity loss, and the damage can range from minimal (e.g., 20/20) to severe levels (e.g., no light perception).
NAION is the most common form of AION and is more likely to occur in younger individuals than AAION. No matter the clinical presentation––whether arteritic or non-arteritic––the visual prognosis usually is poor and the visual acuity and/or visual field loss is permanent.
Here, we present a case of non-arteritic anterior ischemic optic neuropathy. Additionally, we discuss the etiology of AAION and NAION as well as routine work-up and treatment strategies for both conditions.
History
A 48-year-old white male presented to my low vision clinic on March 19, 2009 following referral by his primary care provider (PCP) for an evaluation of his “field cuts,” as well as to learn more information about “adaptive measures and resources.”
With a bit of uncertainty, the patient reported that he had been diagnosed with AION in his right eye three years ago and then in his left eye approximately one year later. Further, the patient indicated that he was diagnosed with hypertension five months earlier and was being medicated with 25mg hydrochlorothiazide (HCTZ) every morning. The patient had no other visual and/or ocular complaints and denied any flashes, floaters, redness, discharge and/or ocular/peri-orbital pain.
In addition to hypertension, his medical history was remarkable for hearing loss, tinnitus, nasal polyps and hyperlipidemia. The patient’s current medications include HCTZ, mometasone furoate, sertraline and 81mg aspirin q.d.
On February 12, 2009, his PCP performed an extensive blood work-up, which yielded normal findings (including a Westergren sedimentation rate of 9mm/h). The patient also underwent a bilateral carotid ultrasound, which indicated the presence of non-significant hemodynamic plaques. Finally, a functional vision and visual prognosis comprehension assessment was performed.
The patient indicated that he, for all intents and purposes, understood the etiology of his vision loss to be the result of “low-blood flow to the optic nerves.” His hobbies included reading and hunting, and he said that he did not experience any difficulties with mobility, near vision and/or lighting. The patient said that he wore sunglasses regularly. Lastly, he indicated that he had never worn corrective glasses or used any low vision devices.
Diagnostic Data
The patient’s entering uncorrected distance visual acuity on an Early Treatment of Diabetic Retinopathy Study (ETDRS) chart was 20/50 O.D. and 20/32 O.S. His entering uncorrected near visual acuity on a continuous text near acuity card was 0.4/1.6M O.U.
Retinoscopy revealed -0.50D +0.75D x 180 O.D. and -1.50D +0.75D x 070 O.S. Subjective trial-frame distance refraction yielded a result of +0.25D +0.25D x 015 with a visual acuity of 20/40 O.D., plano +0.75D x 085 with a visual acuity of 20/20 O.S. and a combined visual acuity of 20/25 O.U. Subjective trial-frame near refraction was +1.75D +0.25D x 015 O.D. and +1.50D +0.75D x 085 O.S. and yielded a near visual acuity of 0.4/0.5M O.U. and 20/20 O.U. on a reduced Snellen near card.
Color vision was assessed using Ishihara pseudoisochromatic plates; the patient correctly identified 10/10 plates O.D., O.S. and O.U. Extraocular muscles (EOMs) exhibited a full range of motion, with no pain and/or diplopia. Pupils were equal, round and reactive to light, with no afferent pupillary defect.
Slit-lamp biomicroscopy revealed clear lids and lashes O.U.; a fully functional and unremarkable lacrimal system O.U.; clear conjunctiva and corneas O.U.; deep and quiet anterior chambers O.U.; clear and flat irises O.U.; grade 1+ nuclear sclerotic cataracts O.U.; and a vitreal syneresis O.U.
We then instilled 1gtt of Fluress (fluorescein and benoxinate, Akorn) O.U. and measured his IOP at 15mm Hg O.D. and 16mm HG O.S. Next, we dilated the patient with 1gtt of tropicamide 1% and phenylephrine 2.5% O.U.
While waiting for dilation, we performed a Humphrey 24-2 SITA Fast visual field test. The results were reliable in the right eye, with 2/13 fixation losses, 1% false positives, 0% false negatives, an outside-normal-limits glaucoma hemifield test, a mean deviation of -20.56dB and a pattern standard deviation of 14.23dB.
Likewise, the results were reliable in the left eye, with 0/13 fixation losses, 0% false positives, 0% false negatives, an outside-normal-limit glaucoma hemifield test, a mean deviation of -12.68dB and a pattern standard deviation of 15.24dB. In the right eye, the field was interpreted as a dense, inferior, altitudinal defect that extended nasally above the horizontal midline—which was perceived as a sign of NAION (Figure 1). In the left eye, we also noted a dense, inferior, altitudinal defect (Figure 2). Accordingly, we determined that the patient wasn’t legally blind.
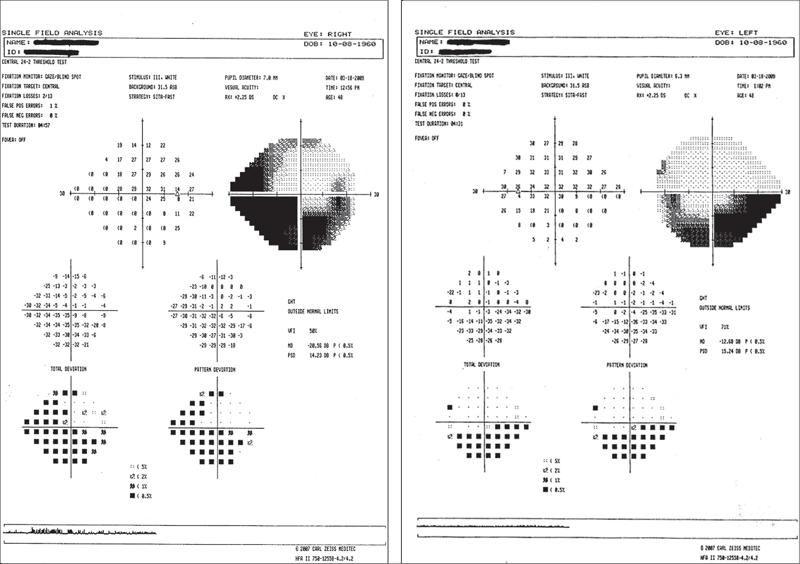
1, 2. Here are the visual fields test results of our patient (O.D. left, O.S. right). The right eye exhibited a dense, inferior, altitudinal defect that extended nasally above the horizontal midline, which was perceived as a sign of NAION. We also noted a dense, inferior, altitudinal defect in the left eye. The results were highly reliable in both eyes.
Dilated binocular indirect ophthalmoscopy and indirect biomicroscopy revealed clear media O.U.; cup-to-disc ratios of 0.1 x 0.1 O.U.; distinct margins O.U.; 2+ superior segmental pallor O.U.; attenuated vasculature O.U.; homogenous background O.U.; clear and flat maculae O.U.; and peripheral retinas that were negative for holes, tear, breaks and/or detachments 360° O.U.
Following fundoscopy, we ruled out superior segmental hypoplasia after noting the normal limit size of both optic nerves (1.7mm to 1.8mm). We also ruled out disc-at-risk/crowed discs due to the bilateral presentation and no evidence of occlusive events (e.g., diffuse retinal hemorrhages in all four quadrants, dilated and tortuous vasculature, cotton-wool spots and/or cherry-red spot of the macula that would indicate central retinal vein occlusion, afferent pupillary defect and markedly reduced visual acuities that would indicate central retinal artery occlusion).
After ruling out the aforementioned conditions, and given the patient’s history of hypertension, we diagnosed him with NAION. We then referred him to my colleague in visual rehabilitation, specifically to address the inferior field cuts’ impact on his mobility.
Additionally, we notified his PCP of the exam findings and recommended yearly comprehensive eye examinations. We encouraged the PCP to consider referring the patient to a cardiologist to rule out any cardiac turbulence that could have produced the small emboli that caused the ischemia.
Finally, we issued an updated, reading-only spectacle prescription, as well as a pair of yellow 54% NoIR fit-over goggles for full-time wear.
Discussion
Anterior ischemic optic neuropathy typically manifests suddenly and unilaterally, but often progresses to the contralateral eye over time. It is the result of ischemia of the posterior ciliary circulation just posterior to the lamina cribrosa. The posterior ciliary artery (PCA) travels along the optic nerve from the ophthalmic artery to the choroid, supplying the optic nerve with tiny penetrating branches.
A surrounding pial plexus derived from these small branches supplies the posterior part of the optic nerve. The optic nerve head receives its arterial blood supply from an anastomotic arterial circle (the circle of Zinn-Haller) that is formed by anastomoses between side branches from the nearby pial arterial network and choroidal vessels.1
AAION and GCA
Giant cell arteritis (GCA) almost exclusively causes AAION. GCA is a systemic vasculitis which preferentially involves medium- to large-sized arteries.2 (However, other conditions, such as periarteritis nodosa, should be considered as a causative etiology as well.3) GCA has a special predilection to involve the PCA, thus resulting in thrombotic occlusion.2
GCA most commonly affects individuals over the age of 55 with a higher incidence in white females.1,4-6 Symptoms of GCA may include general fatigue, weight loss, fever, temple pain, neck pain, pain on chewing, scalp tenderness when combing their hair, headache, anemia, joint pain and tender temporal artery.4-6 However, it has been noted that approximately 21.2% of patients with visual loss due to GCA have occult GCA.2 This means that, contrary to universal beliefs, one in five patients with GCA is at risk for vision loss without showing any of the classic GCA symptoms.2
Ocular signs and symptoms of GCA may include sudden, unilateral, painless decreased visual acuity and color vision (dyschromatopsia); positive afferent pupillary defect; altitudinal visual field defects (usually inferior); swollen optic disc (chalky-white pallor or atrophy after six to eight weeks); cotton-wool spots; and in the event of anterior chamber ischemia, ocular hypotony and/or marked exudation in the anterior chamber (most often misdiagnosed as anterior uveitis).2,6
Visual Rehabilitation for AION Patients with Field Loss
Management of visual field loss includes orientation and mobility
training as well as prisms (both yoked and un-yoked), mirrors and visual
field awareness systems. The purpose of orientation and mobility
training is to improve scanning techniques while teaching and
reinforcing sound and safe navigational techniques and skills. Prisms,
mirrors and visual awareness systems function to expand the patient’s
visual field. Mirrors reflect the field that the patient does not see
and prisms shift/superimpose this field into another field that he or
she can still see. Furthermore, visual awareness systems utilize prisms.
For example, the Gottlieb Visual Field Awareness system orients a small
circular prism that is situated outside the visual axis of the
spectacle lens that allows the patient to scan into the prism to gain
awareness of the field they are missing.
Approximately 30% of individuals who experience permanent vision loss secondary to AAION will first experience episodes of transcient visual loss (e.g., amaurosis fugax).1 Another 5% to 10% of AAION patients will experience transient diplopia secondary to ischemia of the optic nerve head, extraocular muscles or cranial nerves.1 Furthermore, peripapillary, retinal and/or choroidal ischemia in the presence of AAION is highly suggestive of GCA.7
• Work-up. The diagnostic work-up for GCA includes erythrocyte sedimentation rate (ESR), complete blood count (CBC), fasting blood glucose, C-reactive protein, venereal disease research laboratory test (VDRL), fluorescent treponemal antibody absorption test (FTA-ABS), antinuclear antibody test (ANA), fibrinogen, platelets and temporal artery biopsy.5,6 Classically, a diagnosis of GCA has hinged upon these five criteria advocated by the American College of Rheumatologists:2
1. Fifty years of age or older at onset.
2. New onset of localized headache.
3. Temporal artery pulse.
4. Elevated ESR.
5. Positive temporal artery biopsy.
Keep in mind that a patient is classified as having GCA if at least three out of the five criteria are met. However, it must be noted that this diagnostic criteria is highly controversial, because there is a significant risk for vision loss––especially with regards to occult GCA (few or no accompanying systemic symptoms). As a result, there is a movement amongst clinicians to signify temporal artery biopsy as the gold standard diagnostic test for GCA.
• Treatment. Treatment of AAION requires immediate and aggressive therapeutic intervention to prevent permanent visual loss. Visual compromise caused by AAION tends to be more severe than that caused by NAION. More specifically, initial vision loss can progress to hand motion or no light perception in up to 54% of AAION patients.7 And, when left untreated, vision loss may become bilateral within days to weeks in at least 50% of cases.7
Systemic corticosteroids should be initiated upon diagnosis of GCA. If the patient experiences accompanying vision loss, steroids may need to be administered intravenously.8 Typically, patients with GCA on systemic steroid therapy will experience a rapid and dramatic relief of headache and malaise within 24 hours; however, just 4% to 15% of patients with arteritic AION experience an improvement in vision loss.1 But, if vision improvement does occur, the patient will usually experience improved visual acuity with persistent and lasting visual field defects.9
NAION
NAION is the most common form of AION and is usually seen in younger patients. However, several researchers have suggested that NAION can affect a patient at any age, with about 10% of incidences occurring in individuals less than 45 years of age.4,7 Additionally, while NAION affects both males and females equally, up to 95% of all cases occur in whites.4,7
From an etiological and pathogenic perspective, NAION presents in one of two forms:
––Transient non-perfusion or hypoperfusion of the optic nerve head.
––Embolic lesion of the arteries/arterioles that feed the optic nerve head.
Transient non-perfusion or hypoperfusion is the most common cause of NAION. The majority of NAION cases are due to transient reductions in blood pressure during sleep.2 This explains why approximately 75% of patients with NAION report a sudden loss of vision upon awakening.4 Less common causes of hypoperfusion include transient reductions in blood pressure secondary to shock and/or a sharp increase in IOP. Embolic causes of NAION are less likely, but when compared to NAION as a result of hypotensive events, the extent of optic nerve head damage is much more extensive and severe.2
Common diseases and/or conditions that could potentially lead to NAION include hypertension, diabetes mellitus, hyperlipidemia, rheumatoid arthritis, herpes zoster, anemia, sickle cell trait, syphilis, Behçet’s disease, antiphospholipid antibody syndrome, carotid occlusive disease, polyarteritis nodosa and nocturnal hypotension.2,4-6,10
• Work-up. Typically, NAION is diagnosed on ocular examination. Examination usually reveals the presence of sudden and painless vision loss, optic neuropathy, a relative afferent pupillary defect, visual field loss, optic disc edema and possibly peripapillary hemorrhages.1 Further, progressive worsening of vision over a few days or weeks is not uncommon.1
It must be noted that a small or absent cup is not truly a risk factor or predisposing consideration for NAION, but merely a secondary, contributory feature.2 The sequence of events for NAION begins as subclinical ischemia of the optic nerve; then axoplasmic flow stasis in the optic nerve fibers; then axonal swelling; then asymptomatic optic disc edema; and finally, compression of the intervening capillaries by swollen axons in a crowded disc. Therefore, a crowed disc is a secondary contributing factor because its impact occurs only after the process of NAION has started.2
Additionally, when disc edema is present, it may be diffuse or segmental and will involve only the superior or inferior portion of the optic disc. Findings of inferior or superior segmental edema may correspond to a division of the circle of Zinn-Haller into distinct upper and lower halves.7 One to two months after the onset of visual loss, disc edema often resolves and optic disc pallor manifests.1
Regarding long-term visual prognosis, researchers in the Ischemic Optic Neuropathy Decompression Trial found that up to 43% of patients who presented with visual acuities worse than 20/64 spontaneously regained three lines at six-month follow-up, with 31% sustaining that benefit at 24-month follow-up.3 Therefore, in acute AION, initial visual loss may resolve spontaneously but never to the acuity level prior to the event. Therefore, despite the prospect of spontaneous improvement, the initial visual acuity loss will remain permanent to some extent. Given that spontaneous visual field improvement is rare, permanent acuity loss is very common when the patient experiences associated visual field defects.2
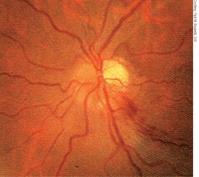
Significant optic disc edema, as seen here, is a hallmark symptom of NAION.
Because our patient’s visual symptoms began three years ago in his right eye and two years ago in his left eye, the visual loss likely is permanent. Therefore, we did not initiate treatment, but rather strongly urged that his primary care provider address and rule out all possible systemic etiologies.
The process of diagnosing NAION is very similar to that of AAION. This is due to the necessity of ruling out AAION, not due to the necessity of confirming the diagnosis of NAION. In fact, NAION is widely a diagnosis of exclusion when GCA is effectively ruled out as the cause of a presenting AION. Nevertheless, when considering a diagnosis of NAION, the universal symptom is a visual field defect.2 Therefore, perimetry is one of the most critical diagnostic tests. Common perimetry findings in NAION patients are an absolute inferior nasal sector defect followed by a central scotoma, relative inferior altitudinal defect and absolute inferior altitudinal defect.2
Upon further analysis of his visual field results, our patient demonstrated what could be described as an absolute nasal sector defect and relative inferior altitudinal defect O.D. and a relative inferior altitudinal defect O.S.
• Treatment. Treatments for NAION are minimal and unsubstantiated at best.2,4-6 These include optic nerve sheath decompressions, subtenon injections of vasodilators, intravitreal bevacizumab injections, intravenous or topical intraocular pressure-lowering agents, vasopressors, stellate ganglion block, levodopa, carbidopa, diphenylhydantoin, anticoagulants, oral corticosteroids and, more recently, intravitreal triamcinolone acetonide.1,2,10
Triamcinolone acetonide injections have been shown to decrease the duration of disc edema while increasing the prospect of a recovery in visual acuity, but not in visual field loss.10 Despite this success, the use of triamcinolone acetonide is not without controversy because, with such a high spontaneous recovery rate in non-treated patients, it is thought that the benefit of such an injection might be exaggerated.2 Further, intravitreal triamicinolone injection in an eye with NAION can be harmful because of the potential for associated IOP increase.2
The primary treatment modality for NAION consists of 80mg to 325mg of aspirin q.d.5,6 This treatment modality is controversial, however, because although aspirin has well documented effects on thromboembolic conditions, NAION is more often a hypotensive condition.2 Furthermore, a few studies have shown that aspirin neither benefits eyes with NAION nor prevents the development of NAION in an unaffected eyes.2
Consequently, due to a lack of viable and effective treatments for clinicians who manage these patients, the primary goal is to rule out an AAION, detect and preemptively control vascular risk factors in the hope of thwarting disease progression and/or occurrence in the contralateral eye (e.g., avoid steps to reduce the likelihood of nocturnal hypotension via consultation with primary care provider to eliminate q.h.s. anti-hypertensive medications).4,10
Because the incidence of AION increases with age, vision rehabilitation clinics should be prepared to treat an increasing number of baby boomers. During the next decade, eye care professionals should be prepared to appropriately handle these patients because a sudden, painless and unilateral loss of vision is often the presenting symptom.
And perhaps, within a few more years, increasingly effective treatments for AAION and NAION will be developed to help reduce the incidence of permanent vision loss.
Dr. Pruitt is the staff optometrist at the Minneapolis Veteran Affairs Medical Center in Minnesota. He is responsible for the outpatient polytrauma/traumatic brain injury and low vision care clinics.
1. Luneau K, Newman N, Biousse V. Ischemic optic neuropathies. Neurologist. 2008 Nov;14(6):341-54.
2. Hayreh S. Ischemic optic neuropathy. Prog Retin Eye Res. 2009 Jan;28(1):34-62.
3. Newman NJ, Scherer R, Langenberg P, et al. The fellow eye in NAION: report from the ischemic optic neuropathy decompression trial follow-up study. Am J Ophthalmol. 2002 Sep;134(3):317-28.
4. Windsor L, Windsor R. Understanding stroke of the optic nerve: Anterior ischemic optic neuropathy. Vision Enhancement. 2002 Spring;7(2):24.
5. Kunimoto DY, Kanitkar KD, Makar MS (eds.). The Wills Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease. Philadelphia: Lippincott Williams & Wilkins; 2004.
6. Kaiser PK, Friedman NJ, Pinead R. The Massachusetts Eye and Ear Infirmary Illustrated Manual of Ophthalmology. Philadelphia: Saunders; 2004.
7. Arnold AC. Ischemic Optic Neuropathy. In: Miller NR, Newman NJ, Biousse V, et al. (eds.). Clinical Neuro-Ophthalmology. 6th ed. Vol. 1. Philadelphia: Williams & Wilkins; 2005:349-84.
8. Chan CC, Paine M, O’Day J. Steroid management in giant cell arteritis. Br J Ophthalmol. 2001 Sep;85(9):1061-4.
9. Foroozan R, Deramo VA, Buono LM, et al. Recovery of visual function in patients with biopsy-proven giant cell arteritis. Ophthalmology. 2003 Mar;110(3):539-42.
10. Sohn BJ, Chun BY, Kwon JY. The effect of an intravitreal triamcinolone acetonide injection for acute nonarteritic anterior ischemic optic neuropathy. Korean J Ophthalmol. 2009 Mar;23(1):59-61.

