 |
We are fortunate to have numerous interventions designed to address the ocular symptoms and local inflammatory component of chronic dry eye disease (DED), but treatment options for the neurological control of tear production have been lacking.
Tear production is an autonomic function that relies primarily on the parasympathetic nervous system, and employing efforts that support proper functioning of the parasympathetic nervous system is a new approach to DED that can result in an increase in the patient’s normal tears. Below we discuss two new treatments for neurological DED that could offer patients much-needed relief.
(Editor’s note: Dr. Driscoll developed and has a financial interest in Parasym Plus Eyes, which is discussed in this article.)
Tyrvaya
This recently FDA-approved nasal spray is a preservative-free formulation that stimulates the nicotinic acetylcholine receptors in the terminal branches of the ophthalmic trigeminal nerve within the lower nasal cavity (a localized approach to DED), according to the company. This is a natural way to stimulate tears as one-third of basal tear production is initiated by inhaling air through the nose.1 Administered twice a day, Tyrvaya is applied to the lower nasal area—the inside of where the lower crease on the nose is located. This may decrease some of the side effects such as throat irritation, coughing or sneezing.
Clinical trials showed a statistically significant increase in Schirmer scores and eye dryness symptoms compared to placebo at week four and sustained through week 12. Studies showed a positive tear production response with few side effects, and could be adjunctive to other treatments or potentially replace artificial tears with patients’ more natural tear production.
 |
|
Tyrvaya. Click image to enlarge. |
Parasym Plus Eyes
This second option approaches neurological dry eye both ocularly (locally) and systemically. Taking on DED as a systemic disorder addresses both inflammation and tear production simultaneously by supporting the autonomic nervous system.
Parasym Plus Eyes is an over-the-counter supplement that supports acetylcholine for both the nicotinic receptors of the vagus nerve and the muscarinic receptors of the lacrimal functional unit.
Stimulation of the vagus nerve helps control inflammation, and stimulation of the nerves serving the lacrimal functional unit promotes tear production. This new supplement stimulates both nicotinic and muscarinic receptors and works around genetic issues and potential nutrient deficiencies involving acetylcholine production. It does not depend upon functioning nerves to work; it depends upon the receptors themselves.
Studies showed patients notice improvement in dry eye within weeks and additional symptoms of low acetylcholine also respond (brain fog, constipation and fatigue often respond in one day).
Dry eye is a multifactorial disease and all conditions contributing to its manifestation, both local and systemic, need to be identified for proper resolution of symptoms.2 As practitioners, we understand that we need to control the inflammatory response, as well as support tear production in DED. Both functions are under control of the autonomic nervous system and can be addressed neurologically. This is a new concept in chronic DED and when recognized and treated appropriately, patients not only experience improved ocular symptoms, but increased systemic wellness as well.
Inflammation and DED
We are familiar with the inflammatory component of DED and recognize autoimmune conditions such as Sjögren’s syndrome, lupus erythematosus and rheumatoid arthritis that cause systemic inflammation, ultimately affecting the eyes. In these cases, systemic treatment, in addition to ocular treatment, is often necessary for associated DED.3 Hormone fluctuations, menopause and low androgenic hormone levels are also inflammatory, may contribute to DED and should be addressed.4,5
Beyond these well-understood conditions, however, chronic, systemic inflammation is rarely addressed, particularly by optometrists, due to the paucity of diagnostic and treatment options. Yet, chronic inflammation is not uncommon. Normal aging, for example, is an inflammatory state characterized by increased inflammatory cytokines that contribute to age-related illness (deemed “inflammaging”).6 Metabolic syndrome, endocrine disorders and even inflammatory bowel disease are systemic inflammatory conditions that can contribute to DED.7,8,9 Recognizing chronic, systemic inflammation in our patients is important because of the damage it causes, both to the eye and systemically.
 |
| Parasym Plus Eyes. Click image to enlarge. |
Inflammation and the Vagus Nerve
A normal, healthy body controls the inflammatory response via the autonomic nervous system. Specifically, the vagus nerve acts as the anti-inflammatory regulator of the body.10,11
The vagus nerve releases acetylcholine, which controls the development and circulation of inflammatory cytokines and chemokines. When the release of acetylcholine is inhibited or the vagus nerve is damaged, it is no longer able to control inflammation, resulting in chronic inflammation and the continued release of damaging inflammatory mediators.12 Yet, despite its importance, testing for systemic and localized inflammatory cytokines and chemokines is quite limited in practice. It is difficult for practitioners to know if localized ocular inflammation reflects chronic, systemic inflammation because of these limitations.
As the anti-inflammatory nerve, the vagus nerve helps control inflammation throughout the body—including the eyes. This nerve also controls digestion, including stomach acid production, the pyloric valve at the base of the stomach, gastric motility, gallbladder and pancreas function, and the sphincter of Oddi, which allows release of bile and digestive enzymes into the intestines.13 When not working well, inflammation increases and digestion suffers. The vagus nerve is a parasympathetic nerve and uses acetylcholine as its neurotransmitter. If this neurotransmitter is not properly released or if it is broken down prematurely, nerve function declines.
Neurology of Basal Tear Production
Autonomic basal tear production is primarily the responsibility of the parasympathetic nervous system and involves the neurotransmitters acetylcholine and vasoactive intestinal peptide. These parasympathetic nerves use acetylcholine as their neurotransmitter and receptors are primarily muscarinic.14
The neurology of tear production depends upon functioning nerves, receptors, and the proper levels of the neurotransmitter acetylcholine that allows the nerves to communicate. If the patient’s neuroanatomy fails at any level, tear production declines.
I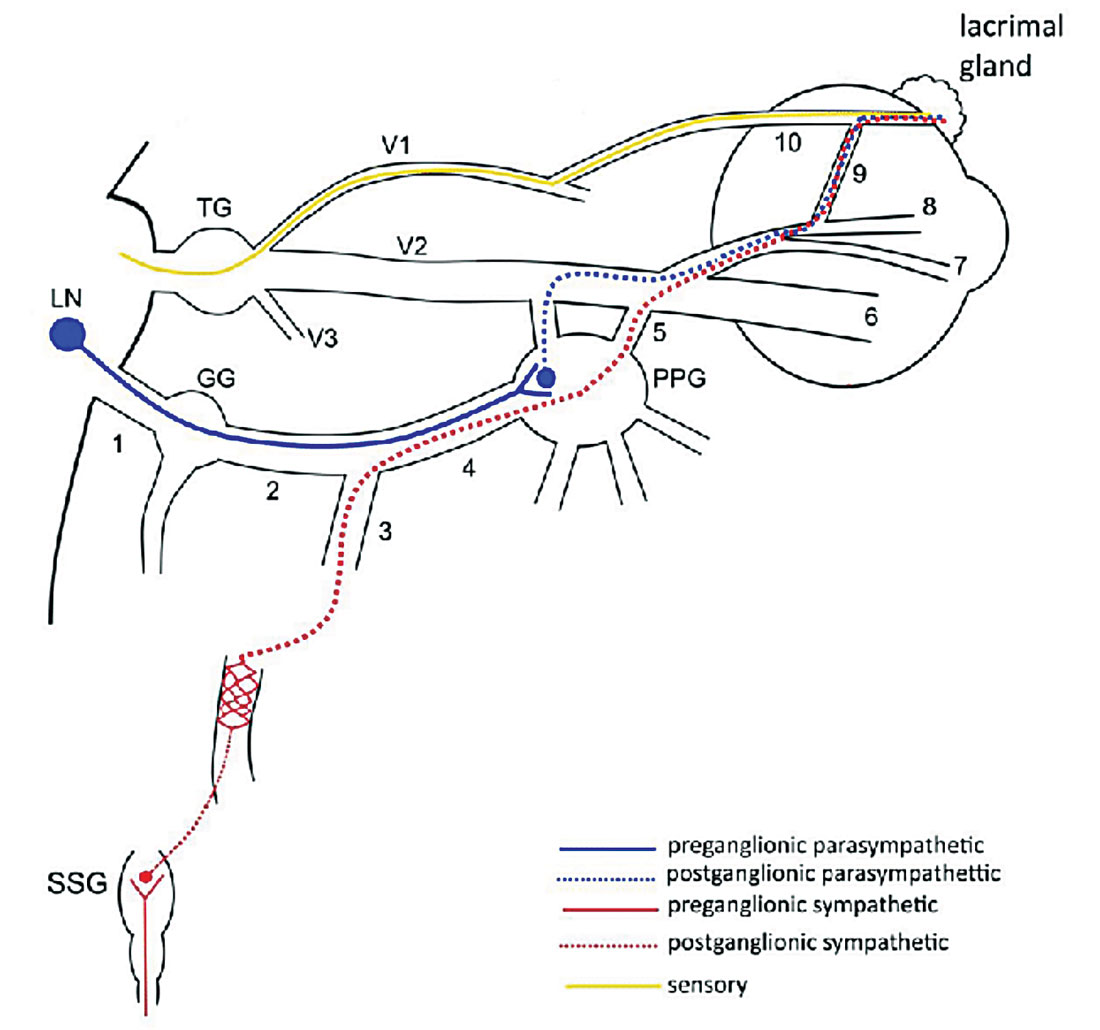 |
The neuroanatomy of tear production. Click image to enlarge. |
Acetylcholine Ties It All Together
The vagus nerve uses acetylcholine as its neurotransmitter. Basal tear production stimulated by the parasympathetic nervous system also utilizes acetylcholine as its neurotransmitter. DED often involves both low tear production and inflammation and both can be affected by a single neurotransmitter—acetylcholine. Although the vagus nerve does not innervate the eye, acetylcholine is the commonality that can tie together both of these autonomic functions.
If acetylcholine is not released properly for any reason, inflammation increases and tear production decreases. Parasympathetic nerve function can decline for several reasons: damage to the nerves themselves, ingestion of medications that break down acetylcholine (e.g., anticholinergics such as Benadryl) and genetics can all affect its production. Some inflammatory cells—and the chemokines they release—can also block the release of acetylcholine. In Sjögren’s syndrome, for example, lymphocytes and chemokines begin to affect the release of acetylcholine, contributing to development and perpetuation of dry eye disease, prior to damage to the acinar cells.15
Recognizing Low Acetylcholine Levels
Acetylcholine cannot be measured through a blood test because acetylcholine breaks down very easily. Anticholinergic medications destroy acetylcholine (such as atropine and diphenhydramine) resulting in anticholinergic syndrome.
We can look at these symptoms for more subtle signs that this is affecting our patients, including increased body temperature, mydriasis (often with light sensitivity), dry eyes and mouth, flushed face and delirium, as well as orthostatic hypotension, brain fog, fatigue and a fast heart rate. Patients may be forgetful, moody, frequently constipated or easily fatigued. Dry eyes, often accompanied with dry mouth, may be assumed to be Sjögren’s syndrome despite normal testing results.
The majority of patients diagnosed with chronic fatigue syndrome/myalgic encephalomyelitis also display the majority of these symptoms, but this aspect of the illness can be overlooked without a diagnostic blood test.16 If the parasympathetic receptors are still viable in these patients, we have a new treatment option for this presentation in the form of Parasym Plus Eyes.
New Possibilities
This newfound ability to address neurological dry eye in our patients should be able to give us greater control over DED’s manifestations in our patients. These unique therapies that provide neurological support offer practitioners new avenues for effective DED management and have the potential to dramatically improve our DED patients quality of life.
Dr. Karpecki is medical director for Keplr Vision and the Dry Eye Institutes of Kentucky and Indiana. He is the Chief Clinical Editor for Review of Optometry and chairman of the affiliated New Technologies & Treatments conferences. A fixture in optometric clinical education, he provides consulting services to a wide array of ophthalmic clients. Dr. Karpecki’s full disclosure list can be found here.
Dr. Driscoll’s work encompasses postural orthostatic tachycardia syndrome (POTS), chronic dry eye, vagus nerve disorders and Ehlers-Danlos syndrome. She is the founder and president of Genetic Disease Investigators and the clinical director of POTS Care. An inventor with three patents to date, she is also the founder and president of TJ Nutrition, makers of Parasym Plus Eyes.
| 1. Gupta, A, Heigle T, Pflugfelder SC. Nasolacrimal stimulation of aqueous tear production. Cornea. 1997;16(6):645-8. 2. Calonge M, Enríquez-de-Salamanca A, Diebold Y, et al. Dry eye disease as an inflammatory disorder. Ocul Immunol Inflamm. 2010;18(4):244-53. 3. Henrich CF, Ramulu PY, Akpek EK. Association of dry eye and inflammatory systemic diseases in a tertiary care-based sample. Cornea. 2014;33(8):819-25. 4. Nguyen CQ, Peck AB. Unraveling the pathophysiology of Sjogren syndrome-associated dry eye disease. Ocul Surf. 2009;7(1):11-27. 5. Truong S, Cole N, Stapleton F, Golebiowski B. Sex hormones and the dry eye. Clin Exp Optom. 2014;97(4):324-36. 6. De Martinis M, Franceschi C, Monti D, Ginaldi L. Inflamm-ageing and lifelong antigenic load as major determinants of ageing rate and longevity. FEBS Lett. 2005;579(10):2035-9. 7. Tang YL, Cheng YL, Ren YP, et al. Metabolic syndrome risk factors and dry eye syndrome: a Meta-analysis. Int J Ophthalmol. 2016;9(7):1038-45. 8. Chopra R, Chander A, Jacob JJ. The eye as a window to rare endocrine disorders. Indian J Endocrinol Metab. 2012;16(3):331-8. 9. Mrugacz M, Sredzińska-Kita D, Cyrta-Jarocka E, Bakunowicz-Lazarczyk A. Dry eye syndrome and cataract as ocular manifestations of Crohn’s disease. Klin Oczna. 2005;107(7-9):509-10. 10. Pavlov VA, Tracey KJ. Controlling inflammation: the cholinergic anti-inflammatory pathway. Biochem Soc Trans. 2006;34(6):1037-40. 11. Pavlov VA, Wang H, Czura CJ, et al.. The cholinergic anti-inflammatory pathway: a missing link in neuroimmunomodulation. Mol Med. 2003;9(5-8):125-34. 12. Czura CJ, Tracey KJ. Autonomic neural regulation of immunity. J Intern Med. 2005;257(2):156-66. 13. Rogers RC, McTigue DM, Hermann GE. Vagal control of digestion: modulation by central neural and peripheral endocrine factors. Neurosci Biobehav Rev. 1996;20(1):57-66. 14. Dartt DA. Neural regulation of lacrimal gland secretory processes: relevance in dry eye diseases. Prog Retin Eye Res. 2009;28(3):155-77. 15. Zoukhri D, Kublin CL. Impaired neurotransmitter release from lacrimal and salivary gland nerves of a murine model of Sjögren’s syndrome. Invest Ophthalmol Vis Sci. 2001;42(5):925-32. 16. Driscoll D. Correcting the missing piece in chronic fatigue syndrome – part 1: discovery. www.vagusnervesupport.com/wp-content/uploads/2017/11/Chronic-Disease-Digest-Text-Correcting-the-Missing-Piece-in-Chronic-Fatigue-Syndrome.pdf. January 2016. Accessed December 1, 2021. |

