Intravenous drug use is directly associated with a variety of localized and systemic complications. In addition, it can yield numerous ophthalmological consequences. The most devastating ocular side effects of intravenous drug use include the formation of choroidal and retinal nodules, infarction and inflammation. Typically, such associated lesions form in the posterior pole near the macula.
Eliciting a thorough patient history is crucial for appropriate testing, culturing and treatment. Prompt diagnosis and management is of utmost importance to decrease both ocular and systemic morbidity.
The following case reports describe complicated infections and potential posterior segment damage secondary to intravenous drug abuse.
Case 1

1.
Fundus photographs of the patient described in Case 1 revealed the
presence of bright retinal infiltrates, hemorrhages and dull choroidal
infarcts in both eyes as well as exudates in his right eye (O.D. left,
O.S. right).
History
A 30-year-old white male presented to our office for the first time with a chief complaint of decreased visual acuity (O.S.>O.D.). His vision had decreased suddenly about two weeks earlier but had remained stable O.U. since. He denied the presence of floaters, photopsia, diplopia, pain or discomfort.
The patient’s medical history was remarkable for post-traumatic stress disorder, bipolar affective disorder, chronic cluster headaches and depression. He did not take any medications.
The patient’s ocular history was unremarkable because he reported never undergoing a formal eye exam. His family ocular and medical histories were unremarkable.
Prior to vision loss, the patient experienced low-grade fevers, severe cluster headaches and diffuse joint pain for one month. His primary care physician (PCP) performed an extensive lab work-up, which included complete blood count (CBC), partial thromboplastin time, international normalized ratio, lupus anticoagulant, amylase, lipase, fluorescent treponemal antibody (FTA), rheumatoid factor, antinuclear antibodies, rapid plasma reagin (RPR), urinalysis and HIV screening.
All test results returned within normal limits. The patient also had a negative CT scan of the head and chest without contrast. He had not seen his PCP since receiving these results.
Upon further questioning, the patient admitted to injecting crack cocaine dissolved in vinegar intravenously. He said that he had injected the drug approximately 10 to 15 times per week for the past five years. He stated that he was aware of the risks of his behavior, but had not sought any counseling or rehabilitation.
Diagnostic Data
The patient’s uncorrected visual acuity measured 20/40 O.D. and 10/300 O.S. with direct fixation. No improvement was observed upon pinhole testing. He achieved 20/200 O.S. with eccentric fixation. His pupils were equal, round and reactive to light, without evidence of afferent defect O.U. Extraocular motility testing showed no restrictions in muscle movement. Confrontation visual fields were full to finger counting O.U.
Intraocular pressure measured 11mm Hg O.D. and 12mm Hg O.S. Anterior segment examination was unremarkable. We detected no inflammatory cells or protein flare in the anterior chamber.
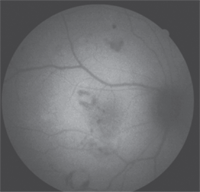
2. Fundus autofluorescence of his right eye illustrated multiple hemorrhages surrounding the white infarcts (Roth’s spots), which blocked the autofluorescence.
Gonioscopy revealed that the most posterior structure in all quadrants was the ciliary body face O.U.
No sign of microhyphema, microhypopyon, peripheral anterior synechiae or neovascularization was noted O.U. Posterior segment evaluation revealed multiple choroidal and retinal infarcts of varying duration (figure 1) as well as the presence of Roth’s spots throughout the posterior pole O.U. (figure 2).
Nerve fiber layer hemorrhages and exudates as well as retinal infiltrates were noted, indicating a septic chorioretinitis. The optic nerves appeared flat, pink and distinct, with no sign of disc edema O.U.
The left macula was affected dramatically by multiple infarcts and intraretinal edema, which correlated with the severe visual decrease in that eye.
The right eye exhibited subtle macular edema and exudates, accounting for the mild decrease in vision.
Following the examination, we ordered additional blood work, including another CBC with
platelet count and differential, troponin I, Westergren sedimentation rate, C-reactive protein (CRP) and a blood culture.
Diagnosis and Follow-Up
We tentatively diagnosed our patient with septic chorioretinitis, pending further testing. We educated the patient about the emergent nature of this condition and informed him that it likely was caused by bacterial endocarditis. We made an immediate referral to a local hospital, and indicated the need for a transesophageal echocardiogram (TEE) to confirm the diagnosis and begin prompt administration of intravenous antibiotics.
He did not report to the hospital as recommended. Upon investigation, a relative informed us that he had died from a gunshot wound to the head. Apparently, he was murdered the night of the referral. We later confirmed this report.
The lab results were received within 48 hours, revealing an elevated troponin I, sedimentation rate and CRP. The CBC remained within normal limits. The blood culture revealed growth of Staphylococcus aureus, which may have been methicillin resistant.
Although the TEE could not be obtained to confirm, we presumed the diagnosis to be bacterial endocarditis secondary to S. aureus infection, very likely due to street drug use.
Case 2
History
A 42-year-old white female presented with a chief complaint of severe blurred vision in her right eye that had persisted for a week. Her vision decreased rapidly and painlessly over the course of two to four days and had remained stable since that time. She did not complain of pain, irritation, diplopia, photopsia or floaters. Additionally, she exhibited no associated systemic signs or symptoms.
The patient’s medical history was unremarkable, but she admitted that she had not seen a PCP in at least 10 years. Her ocular history was unremarkable; however, she reported wearing glasses since age 12. Her current prescription was at least four to five years old. The patient’s family ocular and medical histories were unremarkable.
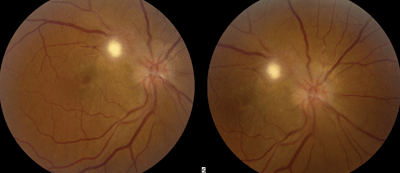
3. Color fundus photography of our patient in Case 2 revealed the position of the retinal lesion as well as the blurred disc margins. Few retinal striae can be seen located temporal to the lesion (O.D. left, O.S. right).
Upon additional questioning, the patient admitted to intravenous heroin use since age 15. She reported that, during the past year, she had reduced her drug usage to “just a few times a month.” However, she admitted to using heroin up to six times per day in the past. She acknowledged the risks of her behavior, and had attended multiple counseling sessions during the last five years. Still, she never enrolled in a formal rehabilitation program.
Diagnostic Data
Upon evaluation, the patient’s corrected visual acuity measured 20/100 O.D. and 20/20 O.S. No improvement was documented upon pinhole testing. Her pupils were equal, round and reactive to light, with no evidence of afferent defect. Extraocular motility testing showed no restrictions in muscle movement. Confrontation visual fields were full to finger counting O.U.
On color vision testing, the patient correctly identified 14/14 Ishihara plates O.U. No red desaturation was detected.
Her intraocular pressure measured 18mm Hg O.D. and 19mm Hg O.S. The anterior segment examination was unremarkable, with no evidence of inflammatory cells or protein flare in the anterior chamber. Gonioscopy revealed that the most posterior structure in all quadrants was the ciliary body face O.U. We documented no sign of microhyphema, microhypopyon, peripheral anterior synechiae or neovascularization O.U.
The posterior segment evaluation revealed a round, yellow-white pre-retinal lesion (figure 3) with surrounding telangiectasia (figure 4) and 2+ posterior vitreous cells in her right eye. Furthermore, we noted trace to 1+ cells in the anterior vitreous.
The right optic nerve had blurred margins. This presentation likely was caused by traction and vitreal inflammation rather than true disc edema, because no afferent defect was noted on pupil testing and color vision was normal.
The fundus examination of the left eye was unremarkable.
Diagnosis and Follow-Up
Immediately, we referred her to the hospital for blood work and intravenous antimicrobial therapy. The patient received an MRI of the brain and orbits, both with and without contrast. Additionally, she underwent FTA, RPR, HIV screening, Lyme titer, angiotensin-converting enzyme, Toxocara screening, purified protein derivative, chest X-ray, CBC testing and a blood culture.
Further, a vitreal culture was taken the following day. A mold infection of unknown species was identified in the vitreal cultures. All other blood testing was negative. The patient was started on intravenous amphotericin B, because of its broad-spectrum coverage. The mold did not reproduce in the mycology lab, which was necessary for species identification.
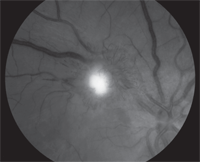
4. Magnified red-free image of the retinal lesion outlined the surrounding
telangectatic vessels in her right eye.
Because amphotericin B has poor vitreal penetration when prescribed orally, the patient also was started on 5µg/0.1mL intravitreal injection of the drug. She received two in-patient intravitreal injections during the five-week hospitalization period. Following discharge, the patient was transitioned from intravenous to oral amphotericin B by her infectious disease specialist. Recently, she reported a moderate improvement in vision with mild distortion. Approximately five and a half weeks after the initial diagnosis, her visual acuity measured 20/50 O.D. and 20/20 O.S.
Vitreous cells were not observed at follow-up. The pre-retinal nodule had formed into a fibrotic scar, causing retinal traction and striae (figure 5). Blood cultures were performed again both two and four weeks after hospital admittance; all cultures returned negative. Eight weeks after initial culture, we identified the mold as a Malbranchea species.
During the next year, we will closely monitor the patient for advancing retinal traction and a potential detachment O.D. We educated her extensively on the risks associated with intravenous drug abuse, and she promised to begin an official rehabilitation program as soon as possible.
Discussion
Recreational injection of street drugs directly is associated with a variety of local and systemic complications. It is also linked to the transmission of infectious diseases through needle sharing and sexual activity. The most serious ocular complications have been reported from the use of crack or crack-cocaine, methylenedioxymethamphetamine or diamorphine (heroin) injections.1
Ophthalmological complications include corneal ulcers, reduced corneal sensitivity, microtalc retinopathy, retinal or choroidal infarcts, central retinal artery or vein occlusion, endophthalmitis, nystagmus, and cerebrovascular accidents that cause neuro-ophthalmic manifestations.2
Retinal or choroidal infarct, inflammation or infiltrates cause some of the most devastating visual sequelae, because they typically are located in the posterior pole.1 These signs are indicative of general septic chorioretinopathy. The underlying cause is bacterial or fungal (or, less commonly, parasitic). Inflammatory causes of chorioretinopathy must also be ruled out.
Eliciting a history of illicit intravenous drug abuse is imperative when septic chorioretinopathy is suspected. This helps to facilitate prompt testing for the most commonly associated pathogens and also could help guide the most appropriate treatment regimen.
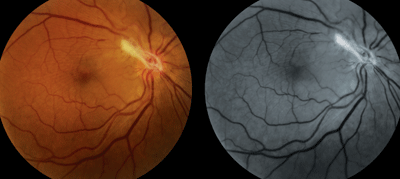
5. Color and red-free fundus photography of the patient’s right eye five weeks after the initial diagnosis and commencement of treatment. Fibrotic scarring now is present in the area of the nodule, with moderate traction and retinal striae throughout the macula and superior vascular arcade.
Bacterial pathogens, specifically those of the Staphylococcus genus, are the most common cause of infection in intravenous drug users.2 Without question, eye care clinicians should be most concerned about the potential for methicillin-resistant Staphylococcus aureus. Other common pathogens associated with this behavior include streptococci, gram-negative bacilli, enterococci, Fusarium, Aspergillus and Candida.2
The development of Roth’s spots is one of the most serious presentations in intravenous drug users who are suspected of bacterial chorioretinitis or endocarditis. Roth’s spots are white-centered hemorrhages that classically are indicative of bacteremia and bacterial endocarditis; however, they also are seen in diseases such as leukemia, pernicious anemia, sickle cell disease and connective tissue disorder.3
In bacterial endocarditis, Roth’s spots are formed as a result of thrombocytopenia and a low-grade, disseminated, intravascular coagulopathy.4 The clinically viewed, white-centered hemorrhages are most likely caused by anoxia, which causes a sudden increase in venous pressure. This causes capillary rupture and extrusion of whole blood. Platelet release causes the coagulation cascade to initiate, eventually causing a platelet-fibrin thrombus surrounded by heme.5
Because of this specific pathology, Roth’s spots are now part of the standard used to determine a diagnosis of bacterial endocarditis.6
According to the standard Duke criteria, infective endocarditis definitely is present under three conditions:6,7
- Two major clinical criteria are present.
- One major and three minor clinical criteria are present.
- Five minor clinical criteria are present.
Major clinical criteria include persistently positive blood cultures for organisms that typically cause bacterial endocarditis, vegetations or abscesses present in heart valves (as seen on echocardiogram), evidence of new echocardial damage, or culture evidence of infection with Coxiella burnetii.
Minor critical criteria include fever, the presence of a predisposing valvular condition or intravenous drug abuse, vascular phenomenon (includes emboli to organs or brain and hemorrhages in the mucous membranes around the eyes), immunologic phenomenon (includes Roth’s spots and Osler’s nodules), and positive blood cultures that do not meet the strict definitions of the major criteria.7
Compared to other classification systems, the Duke Criteria exemplifies the highest validity. Multiple studies have shown its predictive value to be approximately 80%, and it rarely rejects any infective endocarditis that is ultimately pathologically confirmed.6-8 The patient in Case 1, although unable to undergo further testing, had a probable diagnosis of bacterial endocarditis according to these criteria. A definitive diagnosis would have required additional testing, including a TEE.
Early treatment of bacterial endocarditis is crucial to maintaining low morbidity and mortality rates. Treatment consists of prolonged parenteral therapy of bacteriocidal agents.
Serial blood cultures are necessary to document sterilization. As previous noted, the most common pathogen associated with bacterial endocarditis in intravenous drug users is Staphylococcus aureus, which accounts for more than half of these infections.9
The most common treatment regimen for S. aureus infective endocarditis is intravenous nafcillin and an aminoglycoside for two weeks.10 Several studies also have evaluated a regimen of oral antimicrobials, because many intravenous drug users refuse hospitalization. However, these agents have not been as successful. Standard of care remains a two-week hospitalization period with serial blood cultures.11
Fungal infections also may cause choroidal or retinal nodules, infarcts and inflammation in intravenous drug abusers. However, Roth’s spots are not typically caused by fungal or parasitic infections.
In intravenous drug users, the most common cause of fungal chorioretinopathy is Candida.12 Chorioretinopathy caused by Candida presents as a round, white, fluffy lesion with a mild to moderate vitritis. This presentation must be differentiated from toxoplasmosis, which is similar in appearance; however, the active lesion often is located directly adjacent to a chorioretinal scar. Other differentials include tuberculosis, syphilis, Lyme disease, sarcoidosis and Toxocara.3
Endogenous fungal endophthalmitis typically is caused by a chorioretinitis that subsequently progresses into the vitreous. Transient fungemia may seed in the highly vascularized choroid.13 Once in the choroid, the yeast proliferates, invokes an inflammatory response and can subsequently rupture into the vitreous cavity. Once in the vitreous cavity, the infection is considered a true fungal endophthalmitis.14
Unless the vitreous is involved, treatment should consist of oral antifungals. A vitritis secondary to a fungal infection is best treated with early vitrectomy and intravitreal amphotericin B.13,15 Be sure to consult an infectious disease specialist once a diagnosis of fungal chorioretinitis or endophthalmitis is made.
At minimum, the duration of treatment for fungal endophthalmitis is five weeks. Ultimately, however, the treatment schedule is dictated by improvement documented on ophthalmological examinations.16
Unfortunately, the species of the mold in Case 2 could not be determined during the treatment period. Therefore, we used broad-spectrum anti-fungals and achieved moderate success.
A diagnosis of septic chorioretinitis warrants further investigation into a patient’s social history; so if you see it, you must inquire about recreational drug use. This is especially true if the patient is in a high-risk population for illicit drug abuse.
Prompt diagnosis and management is crucial to lowering the risk for ocular and systemic morbidity and mortality associated with these findings. Comanage with an infectious disease specialist early in the treatment course for the best possible outcome.
We must also offer and encourage patient education and rehabilitation services immediately following any hospitalization requirements.
Intravenous drug abuse is associated with a variety of local and systemic complications. Numerous ophthalmological consequences can be observed with or without patient symptoms. Observation of suspicious signs warrants a thorough history, including recreational drug use. Eliciting a connection between intravenous drug abuse and clinical signs is essential to quickly and accurately diagnose and manage both the ocular and systemic complications.
Dr. Legge is in private practice at Wyomissing Optometric Center in Pennsylvania. She graduated from Salus University in 2012 with a concentration in advanced retinal studies.
1. Firth A. Ocular sequelae from the illicit use of class A drugs. Br Ir Orthopt J. 2004 Jan;1:10-8.
2. Cherubin C, Sapira J. The medical complications of drug addiction and the medical assessment of the intravenous drug user: 25 years later. Ann Intern Med. 1993 Nov 15;119(10):1017-28.
3. Wills Eye Hospital. The Wills Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease, 5th ed. Philadelphia: Lippincott; 2008.
4. Ling J, James B. White centered retinal hemorrhages (Roth spots). Postgrad Med J. 1998 Oct;74(876):581-2.
5. Urbano F. Peripheral signs of endocarditis. Hosp Phys. 2000 May;5:41-6.
6. Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med. 1994 Mar;96(3):200-9.
7. Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000 Apr;30(4):633-8.
8. Hoen B, Selton-Suty C. Evaluation of the Duke criteria versus the Beth Israel criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 1995 Oct;21(4):905-9.
9. Mathew J, Addai T, Anand A, et al. Clinical features, site of involvement, bacteriologic findings, and outcome of infective endocarditis in intravenous drug users. Arch Intern Med. 1995 Aug 7-21;155(15):1641-8.
10. Chambers HF, Miller RT, Newman MD. Right-sided Staphylococcus aureus endocarditis in intravenous drug abusers: two-week combination therapy. Ann Intern Med. 1988 Oct 15;109(8):619-24.
11. Heldman AW, Hartert TV, Ray SC, et al. Oral antibiotic treatment of right-sided staphylococcal endocarditis in injection drug users: prospective randomized comparison with parenteral therapy. Am J Med. 1996 Jul;101(1):68-76.
12. Klotz SA, Penn CC, Negvesky GJ, Butrus SI. Fungal and parasitic infections of the eye. Clin Microbiol Rev. 2000 Oct;13(4):662-85.
13. Aguilar G, Blumenkrantz M, Egbert P, McCulley J. Candida endophthalmitis after intravenous drug abuse. Arch Ophthalmol. 1979 Jan;97(1):96-100.
14. Omuta J, Uchida K, Yamaguchi H, Shibuya K. Histopathological study on experimental endophthalmitis induced by bloodstream infection with Candida albicans. Jpn J Infect Dis. 2007 Feb;60(1):33-9.
15. Essman TF. Treatment outcomes in a 10-year study of endogenous fungal endophthalmitis. Ophthalmic Surg Lasers. 1997 Mar;28(3):185-94.
16. Popovich K, Malani PN, Kauffman CA, et al. Compliance with Infectious Diseases Society of America guidelines for ophthalmologic evaluation of patients with candidemia. Infect Dis Clin Pract. 2007;15:254.

