In terms of diagnostic mystery, isolated recurrent corneal erosions (RCE) don’t usually offer much—their symptomology and timing are consistent to the point of being pathognomonic. Symptomatically, they present with a shout rather than a whisper. Patients report substantial—in many cases—debilitating pain that occurs acutely upon waking or in the middle of the night, as well as a dramatically watering eye and photophobia.

When paired with concomitant ocular surface disease (specifically, substantial dry eye syndrome, floppy lid syndrome or nocturnal lagophthalmos), arriving at the diagnosis can be more challenging. However, in most cases, carefully listening to patient symptoms paired with timely examination of the cornea can almost always lead to the correct diagnosis.
Despite relatively little difficulty identifying RCE and our fairly good understanding of its pathogenesis, our most frequently employed treatments for it, bland or hypertonic ointment, haven’t evolved much since RCE was first recognized 140 years ago. In this article, we’ll examine the condition, discuss its pathogenesis and compare various treatment strategies.
Where It All Begins
RCEs have their foundation in abnormalities in the junction between the patient’s corneal epithelium and Bowman’s layer, a thin acellular layer located just below the corneal epithelial basement membrane. These abnormalities may be primary in nature (caused by a dystrophy) or secondary (caused by trauma). Traumatic RCEs are the most common type, accounting for 45% to 64% of cases.1,2 Dystrophy-associated RCE, typically linked to epithelial basement membrane dystrophy (EBMD), accounts for 19% to 29% of cases.1,2 It may also be encountered in stromal dystrophies, such as lattice and granular dystrophies.
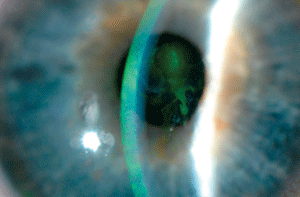
Central recurrent corneal erosion. Photo: Shaun Coombs, OD
With each type of RCE, the initial insult differs—but the anatomy involved in their formation is the same. Let’s take a deeper look.
The corneal epithelium, five to seven cells in thickness, is composed of the mature superficial layer, the evolving wing cell layer and the miotic monolayer of basal cells. Like all of the corneal epithelial layers, the basal cells are joined to adjacent cells by desmosomes. On their basal surface, they are also joined to the basement membrane, Bowman’s layer and the anterior stroma by an adhesion complex made up of hemidesmosomes and type VII collagen-anchoring fibrils.3,4
Abnormal deposition of the epithelial basement membrane—after trauma or secondary to dystrophic processes—disrupts this adhesion complex, which is thought to be causative in the genesis of RCE. When an isolated corneal abrasion occurs (leaving behind intact basement membrane), the lesion will generally heal in five to seven days with appropriate formation of the adhesion complex. However, when the basement membrane is also removed, mature adherence does not take place until six to eight weeks.2
During the interval when the causative epithelial abrasion has superficially healed, but an immature, absent or adherent complex is present, the patient is at risk for spontaneously sloughing the fragile epithelium. This sloughing nearly always takes place at night, when mild epithelial edema or ocular surface drying may weaken the epithelium’s tectonic integrity or promote adhesion to the eyelid.
Onset occurs upon waking or during REM sleep, when the shearing force generated between eyelid and corneal epithelium results in a reopening of the abrasion. EBMD-related RCEs are caused by similar, though primary, abnormalities in the adhesion complex.
RCEs may be either microform or macroform in size. Macroform RCEs present with the classic history and an epithelial defect. Microform lesions typically epithelialize between the onset of symptoms and presentation to clinic.
Treatment
Treatment of RCE can be divided into the acute and chronic phases.
- In the acute phase, the goal of therapy is defect closure. This provides subsequent symptom relief to the patient. Acute-phase RCEs, like all corneal abrasions, often respond well to patching, ointment (bland or antibiotic) or bandage soft contact lenses (BSCLs). Palliative care in the form of topical NSAIDs can effectively limit pain, but does have the potential to slow healing.
- The goal of treatment in the chronic phase is to either passively allow—or therapeutically facilitate—effective formation of the anchoring complex. This is attempted either through protecting the corneal epithelium to allow time for appropriate adhesion complex formation, or through induction of scar-based adhesions from the epithelium to the anterior stroma.
The treatment of these two phases is not mutually exclusive—that is, modulation of the corneal healing response can and should be implemented while the acute episode is healing to reduce potential for future episodes. However, this idea doesn’t seem applicable to the original offending abrasion; currently, there is no research to suggest that treating traumatic corneal abrasions as an RCE will reduce the likelihood of subsequent RCE development.5
Bland or Hypertonic Ointments
Historically, the mainstay treatment for RCE has been nocturnal use of bland or hypertonic ointments. The therapeutic goal of bland ointment is to limit nocturnal friction between the corneal epithelium and the lid. Theoretically, this minimizes shearing forces and, if applied over time, allows for the epithelial adhesion complex to develop appropriately. Hypertonics are designed to limit nocturnal epithelial edema, which is thought to reduce corneal epithelial adherence.
Yet when compared directly in one study, the authors noted no difference between hypertonic ointment and bland ointment, leading them to conclude that lubrication was the sole therapeutic benefit to both approaches.4 While most studies seem to suggest a 30% to 50% recurrence rate with conservative therapy, a randomized study with 72 patients who suffered traumatic corneal injury found an actual worsening of symptoms when treated with bland ointment—although no impact on the likelihood of future RCE was seen.1,5,6
Bandage Soft Contact Lenses

Typical EBMD with gray subepithelial deposits of basement membrane. Photo: Reid Mamiya, OD
BSCLs, along with matrix metalloproteinase inhibitors and autologous serum use, occupy the middle ground between conservative and aggressive therapy. In principle, BSCLs effectively provide a buffer between the lid and the corneal epithelium. Therefore, long-term use of BSCLs could then be used to prevent RCE while the epithelial adhesion complex matures.
In the past, BSCLs were not viewed as an efficacious or even entirely safe therapy; however, since the advent of silicone hydrogel contact lenses, they seem to have taken on more of a prominent role.7 In a 2011 study, subjects who previously had failed medical-only therapy were assigned to treatment with a plano power, 8.6mm base curve Ciba Night and Day BSCL (Alcon) and prophylactic topical ofloxacin ophthalmic drops (dosed BID).8 The patients continued this treatment for three consecutive months, reporting back to clinic every two weeks for lens replacement and evaluation. Of the group, 75% had no recurrence over one year after discontinuing therapy.
Despite the small sample size of this study (12 patients), it is interesting to note that the success rate is as good or better than that reported with anterior stromal puncture (ASP) and equivalent or slightly worse than that reported with epithelial debridement with diamond burr polishing and phototherapeutic keratectomy (PTK), which are considerably more invasive and costly (especially PTK).1,9-11
MMP Inhibitors
Matrix metalloproteinases (MMPs) are a family of enzymes that play a role in the remodeling degradation of connective tissue, including epithelial basement membrane. While there are several important members of this family active in the corneal wound response, MMP-2 and MMP-9 appear to be of particular importance in RCE. Both MMP-2 and MMP-9 are produced by stromal fibroblasts and epithelial cells, respectively, and each is important in breaking down components of the epithelial adhesion complex.12
Just how deleterious are the effects of increased MMP expression on the tectonic structure of the cornea? Pseudomonas aeruginosa—a gram-negative bacterium with the potential to rapidly cause corneal perforation—secretes its own MMPs, allowing it to break down connective tissue, enabling deeper penetration.12
In regard to their role in RCE, MMP-2 and MMP-9 have been shown to increase concentration within the tear film among patients with RCE, theoretically leading to reduced stability of the epithelial basement membrane and increased potential for RCE.13 Theoretically, then, the use of tetracyclines as inhibitors of MMP activity is supported in the treatment of RCE. In a group of seven patients with RCE recalcitrant to conservative therapy, oral doxycycline was used (50mg BID for two months) with no recurrences noted.14 However, this study only evaluated RCE cases with traumatic etiologies; the sample did not include any dystrophic cases of RCE. This may have importance when selecting a treatment for RCE, as MMP activity conceptually would not be as likely to play a role in the genesis of dystrophy-associated RCE.
Likewise, corticosteroids also can suppress MMP activity and expression. In the same study noted above, topical corticosteroids also were used during the acute stage with the goal of further suppressing MMP activity. These too were found to have a beneficial effect––although it was slightly weaker in limiting expression compared to doxycycline.14 With any use of topical corticosteroids, prospective benefit needs to be balanced with potential risk, and appropriate follow-up is required. As the anchoring complex takes two to three months on average to stabilize, therapeutic suppression of MMPs should be continued over that timeframe.2
Autologous Serum
Autologous serum topical eye drops are made out of blood drawn from the patient. The blood is centrifuged; the serum is drawn off and, in some cases, diluted. The serum solution is then packaged and used as an eye drop. The benefit of this modality is that the biochemical properties of blood serum are very similar to that of the tear film.
Its use has been explored in the treatment of a variety of ocular surface diseases and has been found to be particularly effective in accelerating the closure of persistent epithelial defects.15 In one study of eyes with RCE not amenable to standard therapy, autologous serum eye drops were used.15 Over two years of follow-up, three recurrences were noted out of 11 eyes. Autologous serum generally is accepted as safe, but serum sterility is not guaranteed. So, concomitant use with prophylactic antibiotic drops is advisable. Once again, treatment duration should reflect the timing required for full healing. Dosing should be reflective of coexisting ocular surface disease and could vary from four times per day to every hour or more.
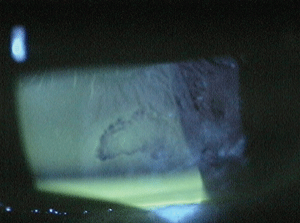
Topical sodium fluorescein clearly delineating the RCE bed.
It’s important to discuss this therapy’s inherent limitations with patients—it can be costly and cumbersome. It takes several steps to obtain: finding a compounding pharmacy to prepare the sample, a lab to draw the sample and sending it to the compounding pharmacy. The patient must undergo a series of blood tests to ensure the sample is HIV negative and free of hepatitis prior to processing of the blood. Autologous serum constitutes experimental therapy, a point that should be covered with patients and reflected in the follow-up.
Superficial Keratectomy
For RCE that is unresponsive to moderate treatment, consider more aggressive approaches, such as delamination of the corneal epithelium and basement membrane with or without polishing of Bowman’s layer, ASP and PTK.
The premise behind superficial keratectomy is that if irregularities in the epithelium and anchoring complex are removed and allowed to grow back in a controlled environment, the structures may normalize as they develop.
The effectiveness of superficial keratectomy alone, without polishing of Bowman’s layer, seems to be in line with less aggressive approaches.
When combined with diamond burr polishing of Bowman’s layer, the effect is enhanced.16 A 2009 study reported a 6% recurrence rate in 25 eyes, compared to 18% in a patient-blinded group that received debridement alone.16 Other groups have confirmed similar findings.16
ASP and PTK
So far, the strategies we discussed have focused on protecting the epithelium as it heals to allow for appropriate formation of the anchoring complex. The goal with ASP and PTK is slightly different—here, we want to create new anchoring junctions.
- ASP. In anterior stromal puncture, a 23- to 25-gauge needle is inserted shallowly into the cornea, penetrating the anterior stroma in a grid-like pattern throughout the bed of erosion. The goal is to create small pinpoint scars into the anterior stroma. The rationale for ASP is that RCE occurs much less frequently when traumatic abrasions penetrate the anterior stroma, compared to those that are simply epithelial in nature.11
Different reports have found that the effectiveness of the procedure varies from 60% to 80%.1,9,11 ASP should be reserved for RCE cases that have their beds out of the visual axis, as there is some potential for reduction in best spectacle-corrected visual acuity if performed in or near the visual axis secondary to scarring.
- PTK. PTK surfaced as a treatment for RCE when it was realized that recurrence of RCE in eyes undergoing photorefractive keratectomy was reduced.4 The exact mechanism is uncertain; however, it has been shown that type VII collagen fibers and hemidesmosomes (both major components of the epithelial-anchoring complex) increased in the laser-treated corneas of monkeys.4
In the treatment of RCE, PTK involves applying the excimer laser to the bed of the RCE, either transepithelially or after epithelial debridement. The success rate is variable but generally high (ranging from 74% to 100%); however, the cost associated with the device and the procedure keep it from being used as a frontline therapy.11,17,18 It should be reserved for cases in which more affordable, less invasive therapies have failed.
Rather than using a cookie-cutter treatment paradigm, I have found it reasonable to approach RCE in a scientific manner in my practice. In otherwise healthy eyes, I’ve had good success with a silicone hydrogel BSCL worn for 10 to 12 weeks and replaced every other week. I always pair this with a prophylactic antibiotic eye drop that has minimal epithelial toxicity, such as ciprofloxacin, while the epithelial defect persists. In the first month, I require the patient to return to the clinic so I can change the BSCL—I use jeweler’s forceps to assist in atraumatic removal.
The benefits of
amniotic membrane tissue in the management of inflammatory and
non-healing corneal wounds have been well documented during the last 20
years. Amniotic membrane works in these cases by reducing inflammatory
mediators, reducing vascularization, providing an artificial basement
membrane for re-epithelialization, reducing the scar response, providing
antimicrobial effects and promoting appropriate innervation.19,20
The
indications for amniotic membrane include: Stevens-Johnson syndrome,
acid or alkali injury, pain relief for bullous keratopathy,
re-epithelialization of neurotrophic ulcers and as a surgical adjunct in
pterygium, glaucoma and limbal-grafting procedures.20 Given its
mechanism of action in enhancing epithelialization and reducing
inflammation (i.e., MMP activity), amniotic membrane could have a place
in the treatment of RCE. While
there are sporadic case reports of successful RCE treatment with
ProKera, there are no well-designed studies on the modality. In
addition, the life of the amniotic membrane is roughly two weeks on the
eye—which does not fit with the six- to eight-week pathogenic timeframe
of RCE. Coupled with the costs and regulation associated with biologic
tissue, amniotic membrane grafting likely should be relatively far down
the list of possible therapies for RCE.
Do Amniotic Membrane Grafts Mesh Well with RCE Treatment?
Recently, amniotic membrane grafting has
become an increasingly popular topic in the optometric community,
possibly due to the ProKera ring (Bio-Tissue) becoming more widely
available. This device is an amniotic membrane sheet supported on a 16mm
plastic ring. It can be applied simply as a large-diameter contact
lens, though the ring itself is much thicker than a standard contact
lens.
However, if the patient has significant lid margin disease or has failed with the BSCL approach in the past, I use oral doxycycline and a soft steroid-antibiotic combination drop, such as Zylet (loteprednol 0.5% and tobramycin 0.3%, Bausch + Lomb). This provides some antibacterial coverage, especially in the acute stage when an epithelial defect is present.
The two strategies of BSCL and MMP suppression with doxycycline could be easily combined as well. But, further suppression using a topical corticosteroid should be avoided––especially prior to closure of the epithelial defect.
I reserve surgical referral for recalcitrant cases of RCE. It could be expected that patients with RCE caused by dystrophic processes will be less amenable to conservative therapy. In these cases, I refer for PTK or epithelial debridement with diamond burr polishing sooner in the disease process.
While the large number of therapeutic options we have at our disposal can seem overwhelming at first, we generally can achieve good success rates in minimizing the recurrence of this painful condition if we remember to treat each patient as an individual, while keeping the different risks and benefits of each therapy in mind.
Dr. Bronner is a staff optometrist at the Pacific Cataract and Laser Institute in Kennewick, Wash.
1. Reidy JJ, Paulus MP, Gona S. Recurrent erosions of the cornea: epidemiology and treatment. Cornea. 2000 Nov;19(6):767-71.
2. Hykin PG, Foss AE, Pavesio C, Dart JK. The natural history and management of recurrent corneal erosion: a prospective randomised trial. Eye (Lond). 1994;8(Pt 1):35-40.
3. Nishida T. Cornea. In: Krachmer JH, Mannis MJ, Holland EJ (eds.). Cornea: Fundamentals, diagnosis and management. 2nd ed. Philadelphia: Elsevier Mosby; 2005:3-26.
4. Watkins A, Macaluso DC, Feldman ST. Pathogenesis of sterile corneal erosions and ulcerations. In: Krachmer JH, Mannis MJ, Holland EJ (eds.). Cornea: fundamentals, diagnosis and management. 2nd ed. Philadelphia: Elsevier Mosby; 2005:151-64.
5. Eke T, Morrison DA, Austin DJ. Recurrent symptoms following traumatic corneal abrasion: prevalence, severity, and the effect of a simple regimen of prophylaxis. Eye (Lond). 1999 Jun;13(Pt 3a):345-7.
6. Reeves SW, Kang PC, Zlogar DF, et al. Recurrent corneal erosion syndrome: a study of 364 episodes. Ophthalmic Surg Lasers Imaging. 2010 Mar 9:1-2.
7. Williams R, Buckley RJ. Pathogenesis and treatment of recurrent erosion. Br J Ophthalmol. 1985 Jun;69(6):435-7.
8. Fraunfelder FW, Cabezas M. Treatment of recurrent corneal erosion by extended-wear bandage contact lens. Cornea. 2011 Feb;30(2):164-6.
9. O’Brart DP, Muir MG, Marshall J. Phototherapeutic keratectomy for recurrent corneal erosions. Eye (Lond). 1994;8(Pt 4):378-83.
10. McLean EN, MacRae SM, Rich LF. Recurrent erosion. Treatment by anterior stromal puncture. Ophthalmology. 1986 Jun;93(6):784-8.
11. Das S, Seitz B. Recurrent corneal erosion syndrome. Surv Ophthalmol. 2008 Jan-Feb;53(1):3-15.
12. Tuli S, Goldstein M, Schultz GS. Modulation of corneal wound healing. In: Krachmer JH, Mannis MJ, Holland EJ (eds.). Cornea: Fundamentals, diagnosis and management. 2nd ed. Philadelphia: Elsevier Mosby; 2005:133-50.
13. Ramamurthi S, Rahman MQ, Dutton GN, Ramaesh K. Pathogenesis, clinical features and management of recurrent corneal erosions. Eye (Lond). 2006 Jun;20(6):635-44.
14. Dursun D, Kim MC, Solomon A, Pflugfelder SC. Treatment of recalcitrant recurrent corneal erosions with inhibitors of matrix metalloproteinase-9, doxycycline and corticosteroids. Am J Ophthalmol. 2001 Jul;132(1):8-13.
15. del Castillo JM, de la Casa JM, Sardina RC, et al. Treatment of recurrent corneal erosions using autologous serum. Cornea. 2002 Nov;21(8):781-3.
16. Wong VW, Chi SC, Lam DS. Diamond burr polishing for recurrent corneal erosions: results from a prospective randomized controlled trial. Cornea. 2009 Feb;28(2):152-6.
17. Ewald M, Hammersmith KM. Review of diagnosis and management of recurrent erosion syndrome. Curr Opin Ophthalmol. 2009 Jul;20(4):287-91.
18. Stasi K, Chuck RS Update on phototherapeutic keratectomy. Curr Opin Ophthalmol. 2009 Jul;20(4):272-5.
19. Tseng SC. Amniotic membrane transplant for persistent corneal epithelial defect. Br J Ophthalmol. 2001 Dec;85(12):1400-1.
20. Dua HS, Gomes JA, King AJ, Maharajan VS. The amniotic membrane in ophthalmology. Surv Ophthalmol. 2004 Jan-Feb;49(1) 51-77.

