Contact Lenses
Icuity H2O
Hydrogel Vision Corp. has expanded its product line with a new lens and new modality—Icuity H2O one-week replacement. This soft contact lens offers patients a more frequent replacement schedule without the cost of a daily disposable lens, the company says.
Made with hioxiflcon A, a non-ionic ultra hydrating material that retains 99% of its water content throughout the wearing time, Icuity H2O is designed to keep lens comfort, movement and fit stable throughout the day. This material keeps lenses clear and free of unhealthy protein deposits and features a thin and smoothly contoured edge for improved fit and comfort, the manufacturer says.
Icuity H2O is available in a median (8.6) base curve with powers of +6.00 to -10.00 and a steep (8.3) base curve with powers of -0.25 to -10.00.
You can order free diagnostic sets of Icuity H2O directly through Hydrogel Vision or through any of its authorized distributors.
Visit
www.hydrogelvision.com.

Optical Display
Sol Sun Center
The new Sol Sun Center by Eye Designs allows you to create a designated selling area for sunwear within your optical. This stylish, modern display unit showcases sunwear, cases and point of purchase materials.
Patients understand exactly where to browse and shop for sunwear, which allows them to make decisions quicker and leads to a more efficient selling time for the optical staff and the patient, the company says.
Featuring backlit illumination with LED lighting, the three columns of frosted adjustable shelves, plus the panel of Wave frame holders, display a total of 92 frames. The Sol Sun Center can be made to match any existing Eye Designs furniture collection or it can be incorporated into any optical interior.
Visit
www.eyedesigns.com.
Dry Eye Measurement
Lipiview Ocular Surface Interferometer
By granting clearance to Lipiview’s second generation software, the FDA also approved additional features and broadened the indication for use of the TearScience’s Lipiview ocular surface interferometer. TearScience can now claim Lipiview measures the absolute thickness of the tear film lipid layer in nanometers.
By identifying lipid layer deficiency (the most common cause of dry eye), physicians can use Lipi-
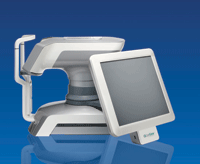
view as part of a full dry eye assessment to determine which type of dry eye a patient has, the company says.
The upgraded Lipiview software also assesses a patient’s blinking process during examination, which enables physicians to identify patients who are partial blinkers—a condition that may limit lipid production and impact the ocular surface.
Lipiview v2.0 software will be available to all new customers, and current customers will be upgraded by the second quarter of 2013.
Visit
www.tearscience.com.
NSAID Ophthalmic Drop
Ilevro Suspension
Alcon launched Ilevro (nepafenac 0.3% ophthalmic suspension), a new once-daily treatment option for pain and inflammation associated with cataract surgery. In two double-masked, randomized clinical trials, this nonsteroidal, anti-inflammatory prodrug demonstrated superior clinical efficacy compared to its vehicle, Alcon says.
In the studies, patients treated with Ilevro were less likely to have ocular pain and measurable signs of inflammation (cells and flare) at the end of treatment than those treated with its vehicle.
Visit
www.alcon.com for full prescribing info.

