 A 76-year-old white female visited the office in January 2008 for transfer of care. She reported that her previous eye doctor monitored her for high pressures, but that she did not require medications. She presented with no significant visual complaints.
A 76-year-old white female visited the office in January 2008 for transfer of care. She reported that her previous eye doctor monitored her for high pressures, but that she did not require medications. She presented with no significant visual complaints.
The same eye doctor followed this patient for many years and gradually noticed changes to her optic nerve. The patient was unaware of a family history of glaucoma, although her daughter was also being observed for high pressures.
She reported well-controlled gastroesophageal reflux disease, hypertension and long-standing atrial fibrillation. Current medications included Lotrel (amlodipine besylate/benazepril hydrochloride, Novartis), Protonix (pantoprazole sodium, Wyeth), Pacerone (amiodarone, Upsher-Smith Laboratories) and Coumadin (warfarin sodium, Bristol-Myers Squibb).
Diagnostic Data
The patients entering visual acuity was 20/30 O.D. and 20/25- O.S., through hyperopic, astigmatic and presbyopic correction. Pupils were round and briskly reactive to light. I noted a 1mm physiological anisocoria, which the patient reported having for many years. The anisocoria was present in photos from 2002, and did not change in either bright or dim illumination. Her extraocular motilities were full in all diagnostic positions of gaze.
Slit lamp examination of her anterior segments was unremarkable except for mild dermatochalasis O.U. Anterior chamber angles, as estimated by Van Herricks method, were grade III open O.U. Her intraocular pressure measured 21mm Hg O.D. and 19mm Hg O.S. at 10:30 a.m. by applanation. Through dilated pupils, her crystalline lenses were characterized by early nuclear and cortical cataracts, which was consistent with her best-corrected acuity.
Her optic nerves were of normal size and were characterized by a cup-to-disc ratios of 0.50 x 0.60 O.D. and 0.50 x 0.70 O.S. The inferior temporal neuroretinal rims were thinned, especially in the left eye. Her macular, vascular and peripheral retinal evaluations were unremarkable O.U. I asked her to return in one month for a full work-up. On follow-up examination, her IOP measured 20mm Hg O.D. and 22mm Hg O.S. Gonioscopy demonstrated grade II to grade III open angles, with normal trabecular pigmentation. Pachymetry readings were 509m O.D. and 499m O.S.
Standard threshold white-on-white perimetry was normal; reliability indices were fair. Heidelberg Retina Tomography-3 (HRT-3) imaging confirmed normal size optic nerves, and Moorsfield Regression Analysis indicated that the inferior temporal aspect of the neuroretinal rim was statistically aberrant O.U.
A review of her previous records revealed several significant findings. Her former eye-care provider had been following her for 25 years, and his records were meticulously kept. On average, her IOP remained in the low 20s, but had occasionally risen into the upper 20s. Visual fields have remained normal. Nevertheless, it appears that there was a documented change in her cup-to-disc ratio circa 2002, with cupping changing from 0.40 x 0.40 to 0.55 x 0.55 O.D. and 0.40 x 0.50 to 0.50 x 0.60 O.S. These results are similar to the current description of her optic nerve. Finally, this notation was made in her chart: Early optic nerve deterioration O.S. > O.D. Defer therapy at the present time.
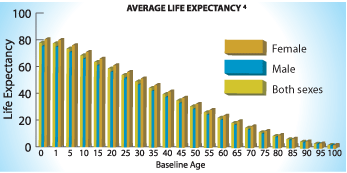
Life expectancy (years left to live) of all ethnicities in the U.S. with respect to baseline age. Consider how long your elderly patients may live before starting them on a costly, extensive treatment regimen for mild glaucoma fairly late in life.
Discussion
I reviewed my findings with the patient and, in particular, talked about her conversion from non-glaucomatous ocular hypertension to early glaucoma.
We have all seen patients in this situation, when different clinical findings suggest a shift from passive monitoring to active therapy. But, as we have seen previously, sometimes the decision to begin therapy can be clouded (See Do We Monitor or Medicate? January 2008).
The patients previous eye-care provider saw manifestations of her conversion (i.e., optic nerve changes); yet, he chose to observe rather than initiate therapy. In glaucoma management, we often have to weigh medical therapy against quality-of-life issues. In this case, my clinical judgment dictated that this patient should be conservatively, yet actively managed. I started her on one drop of Lumigan (bimatoprost, Allergan) h.s. O.U.
I based my decision to treat on several factors. First is the well-documented change, seen by the former eye-care provider, in the patients neuroretinal rim. Also, I noted a pathological aberration of her inferior temporal neuroretinal rims, which was confirmed by the HRT-3 findings of the inferior temporal regions of her right and left optic nerves. Such changes, in the presence of ocular hypertension, have been estimated to predict the development of visual field loss up to eight years in advance.1
Finally, she exhibits documented fluctuation in IOP. The exact role of IOP fluctuation has not yet been clearly established. Some authors suggest that IOP fluctuations increase the risk of development and progression, while others do not. Intuitively, though, I think that large changes in IOP play a role in glaucoma development and progression.
A secondary factor in my decision to initiate therapy is her cardiac arrhythmia and hypertension. While not considered a primary risk factor, the presence of cardiovascular abnormalities does increase a patients likelihood for glaucoma development when other risk factors exist.2
For some clinicians, the patients age may play a primary role in a decision not to treat. Since our patient is 76 years old, we must realistically consider that she could die before she suffers any visual field loss. Clearly, we cannot predict the future; however, we certainly do not want to unnecessarily subject the patient to a rigorous, expensive treatment regimen.
Considering the patients HRT-3, she could be age 84 before suffering visual field loss, which may be an argument to further defer treatment. Will this patient likely reach 84? We can take a random guessor we can make an educated guess.
The Centers for Disease Control and Prevention (CDC) and the United States Department of Health and Human Services (DHHS) collect and analyze life expectancy data.3,4 Currently, the DHHS estimates that 12.4% of the American population is over the age of 65 (37.3 million individuals), and that this will surge to 20% (71.5 million individuals) by the year 2030.4
In 2004, average life expectancy in the
In other words, the average 65-year-old American can expect to live to age 84, but a 75-year-old can expect to live to 87. Simply put, your likelihood of outpacing average life expectancy increases once you live beyond the magic milestone of age 65 and, better yet, age 75. Therefore, the older and healthier you are, the greater your chances are to outlive average life expectancy.
So, whats our patients life expectancy? Given these statistics, our 76-year-old female has a life expectancy of another 12 years. Would she suffer visual impairment from glaucoma before age 88 if left untreated? Probably. And, given that she is still very active in the community and on the golf course, I chose to intervene. Only time will tell if I made the right decision.
1. Zangwill LM, Weinreb RN, Beiser JA, et al. Baseline topographic optic disc measurements are associated with the development of primary open-angle glaucoma: the Confocal Scanning Laser Ophthalmoscopy Ancillary Study to the Ocular Hypertension Treatment Study. Arch Ophthalmol 2005 Sep;123(9):1188-97.
2. Orzalesi N, Rossetti L, Omboni S. Vascular risk factors in glaucoma: the results of a national survey. Graefes Arch Clin Exp Ophthalmol 2007 Jun;245(6):795-802.
3. The United States Department of Health and Human Services: The Centers for Disease Control and Prevention. Life Expectancy. Available at: www.cdc.gov/nchs/fastats/lifexpec.htm (Accessed April 2, 2008).
4. The United States Department of Health and Human Services: The Administration on Aging. Statistics on the Aging Population. Available at: www.aoa.gov/prof/Statistics/statistics.asp (Accessed April 2, 2008).

