40th Annual Technology ReportFollow the links below to read the other articles from our 40th annual Technology Report: Downsize Your Technology to Enhance Your Practice How Early Diagnosis Can Improve AMD Outcomes Understanding Today's OCT Technology—And Anticipating Tomorrow's |
With the US prevalence of glaucoma expected to rise to roughly 6.3 million by 2050, optometrists must be vigilant in screening patients to catch even the earliest signs of disease.1 Today’s technologies are integral to assessing patient risk and providing the data necessary to diagnose and treat long before permanent visual loss. Clinicians can use any number of tools—including corneal hysteresis (CH) testing, OCT angiography (OCT-A), 10-2 visual field testing and even electrodiagnostics—along with their routine workup to aid in glaucoma diagnosis and management. Here, we discuss how clinicians can implement these ancillary tests on a patient-by-patient basis to better diagnose and monitor glaucoma.
Corneal Hysteresis
The viscoelastic properties of the cornea and its ability to absorb energy are represented by CH, and low CH is a risk factor for glaucoma and progression.2,3 Currently, the only commercially available instrument to measure CH, based on the definition and applying bidirectional applanation tonometry, is the ocular response analyzer (ORA) from Reichert Technologies.2
Table 1. SD-OCT-A Advantages and Limitations1,2 | |
| Advantages | Limitations |
|
|
| 1. Akil H, Falavarjani KG, Sadda SR, Sadun AA. Optical coherence tomography angiography of the optic disc; an overview. J Ophthalmic Vis Res. 2017;12(1):98-105. 2. de Carlo TE, Romano A, Waheed NK, Duker JS. A review of optical coherence tomography angiography (OCTA). International J Retina and Vitreous. 2015;1:5. | |
An alternative device, the Corvis ST (Oculus), uses corneal deformation by Scheimpflug imaging or OCT, via a combination of corneal topography, bidirectional applanation and high-speed photography.4 Although this device has FDA clearance for sale in the United States, it is not as well studied as the ORA. However, the Corvis ST does provide video of the cornea deformation during testing, and it captures more than 4,000 images per second, which may reduce variables of poor fixation and reduced tear film while increasing the intraocular pressure (IOP) measurement accuracy.5 Other technologies evaluating corneal biochemical properties include electronic speckle pattern interferometry, ultrasonic elastography, high-frequency ultrasonographic analysis and Brillouin microscopy.6
For the ORA more specifically, its value denotes the pressure difference between the applanation of the cornea by an air jet and the recoil outward force by the corneal tissue, measured by an infrared laser to measure CH. In addition to CH, ORA also provides a calculated measurement of the corneal resistance factor (CRF), representing the elasticity of the cornea. Research shows CH and CRF are reduced in ocular conditions such as Fuchs’ dystrophy, as well as systemic conditions such as diabetes and lupus. Both measurements are also lowered in cases of corneal ectasia and after refractive surgery.7
The ORA is also designed to estimate IOP.8 It acquires both the corneal-compensated IOP (IOPcc) and the Goldmann IOP (IOPG). The IOPcc may be less dependent on corneal thickness compared with applanation tonometry, and research shows the IOPG is comparable with Goldmann tonometry.6,8
Studies demonstrate that CH is a repeatable value that typically shows correlation between the right and left eye and no variation between sex.9,10 In normal eyes, CH has no significant diurnal variation, but it does decrease with age similar to central corneal thickness (CCT).11,12 Studies estimate the average CH normative value ranges from 10.07mm Hg to ± 1.61mm Hg.13 Obtaining a baseline CH value prior to treatment is particularly important, as CH tends to increase as IOP decreases with ocular hypotensive therapy.1,2,14 Differences in CH based on ethnicity remain poorly defined.11,12
Concerning glaucoma and disease progression, research shows reduced CH is associated with a greater optic nerve head cup depth and size.15 Studies also demonstrate that patients with increased CH values have more optic nerve deformation when eye pressure was high, thereby allowing the ocular tissue to dissipate the pressure, providing protection against mechanical damage to the optic nerve.16
Patients with glaucoma tend to have decreased CH and CCT compared with normal subjects, and reduced CH has been correlated with progression of visual field loss.17 Studies also confirm that patients with reduced CH have a faster rate of glaucoma progression.18 When CH is not similar in both eyes of glaucoma patients, research shows a faster rate of progression in the eye with lower CH.19,20 CH may be predictive of how a patient will respond to ocular hypotensive therapy, as patients with low CH may have a larger IOP reduction compared with patients with high CH values.14 Therefore, if a patient with high CH takes an IOP-lowering medication, the IOP reduction may be less than is seen in an eye with low CH. This does not mean the drop is not efficacious, but the target IOP should be reassessed based on the cornea’s biomechanical properties.
CH may be more strongly associated with glaucoma risk and progression than CCT, which has been standard of care in glaucoma evaluations since the Ocular Hypertensive Treatment Study.18,21 The exact mechanism of action between CH and glaucoma pathogenesis is not completely understood, but research indicates it may play a viable role in glaucoma management.12
OCT-Angiography
Ocular blood flow (OBF), oxidative stress, reduced axoplasmic flow and genetics have all been implicated in the pathogenesis of glaucoma.22 Research shows that OBF tends to be lower in glaucoma patients, specifically normal-tension glaucoma, compared with healthy controls.23 The result of low OBF is reduced perfusion pressure, which leads to reduced oxygen supply and release of hypoxic mediators. This mechanism may be secondary to autodysregulation within the vascular system.23 Here, OCT-A technology can help to uncover an OBF impairment to the optic nerve and peripapillary retina. Research demonstrates that reduced OBF is a potential risk factor for glaucoma development and progression, as blood flow to the optic nerve, derived from the central retinal artery and posterior ciliary arteries, can be reduced in glaucoma patients (Table 1).24-27
However, OBF was difficult to quantify, as the peripapillary capillary network is poorly characterized with fluorescein angiography.28 OCT-A now provides a view into the microcirculation of the optic nerve and peripapillary retina, confirming an association between glaucoma and the severity of OBF impairment.22 This may provide a means of detecting the disease in its earliest, “pre-perimetric,” stage.
While a dense microvascular capillary network exists around the optic nerve head in normal eyes, glaucomatous eyes have delicate capillaries that can be easily attenuated and disrupted, leading to capillary dropout.29 Disc flow index and vessel density values represent quantifiable measures of perfusion around the optic nerve and are derived from algorithms using OCT-A. These values—although not currently a component of commercially available OCT-A software in the United States—could potentially help establish a normative standard data set to differentiate between normal and glaucomatous eyes, as well as stratify damage in glaucoma.25
Research shows a direct correlation between optic nerve perfusion and structural and functional damage in glaucoma.30 Specifically, reduced flow index and vessel density measurements correlating to reduced peripapillary and optic nerve perfusion are associated with increased visual field mean deviation and OCT retinal nerve fiber layer (RNFL) and ganglion cell-inner plexiform loss.25,31
Using OCT-A, investigators may better understand whether OBF causes glaucoma or if it is a structural manifestation of the process. Researchers know microvasculature dropout occurs in the peripapillary retina in the location of RNFL defects in glaucomatous eyes.32,33 The volume of capillary loss and optic nerve damage in glaucomatous eyes is proportional, suggesting the loss of peripapillary capillary structure is the result of the proceeding RNFL atrophy and glaucomatous neuropathy.34
Visual Field Testing
Research shows the central 16 degrees of the retina contains approximately 50% of the total ganglion cell distribution but only represents approximately 7% of the total retina area.35 The advances in OCT technology have made it easier to see this more concentrated area of ganglion cells on a pseudocellular level with ganglion cell analysis (GCA). This is especially useful when evaluating glaucoma suspects with normal RNFL and no defects on a 30-2 or 24-2 (white on white) visual field. Recent studies indicate that early glaucomatous damage can occur within the central 10 degrees of the visual field with little to no change noted at the neuroretinal rim.36 When thinning at the rim is present, it tends to be located at the inferior temporal neuroretinal rim, which projects to the inferior temporal region of the retinal ganglion cells (RGCs) of the macula—known as the macular vulnerability zone (MVZ).37 These RGCs are at a higher risk of damage due to their projection to the inferior region of the optic disc, which is more susceptible to damage. Typical visual field protocols fail to thoroughly and accurately detect subtle abnormalities in this region. Due to the projection of the axons from the RGCs within the MVZ, damage will consequently give the patient a superior paracentral scotoma—visible on a 10-2 visual field—classified as arcuate, comma-shaped or nasal step in appearance. The superior ganglion cell axons project to the temporal region of the optic disc, which is less susceptible to early glaucomatous damage, making an inferior paracentral visual field defect less likely.37
OCT GCA and 10-2 visual field testing does come with limitations. Because retinal anatomy varies from person to person and within ethnicities, limited normative databases can be an obstacle to proper diagnosis.38
In addition, segmentation errors due to concurrent macular pathology, specifically in the inner retinal layers, can confound the ganglion cell data. However, using 10-2 visual field testing in patients with a suspicious optic nerve appearance with unremarkable 24-2 or 30-2 visual field exams, in conjunction with OCT GCA, may help to rule out early macular damage due to early glaucomatous disease in a subset of glaucoma suspects.
Electrodiagnostics
Even in early glaucoma, research suggests one third to half of RGCs are dead or dysfunctional before defects are present on visual fields.39,40 Electrodiagnostics may one day allow clinicians to detect this early dysfunction before the presence of defects, providing a predictive measure of conversion in glaucoma suspects. Several innovations are useful in research and may eventually be in clinical practice:
Pattern electroretinogram (pERG). Evidence suggests pERG can assist in recognizing early glaucomatous damage by assessing RGC layer dysfunction. A study comparing RNFL OCT with pERG revealed decreased pERG amplitudes correlated with reduced RNFL, indicating dysfunctional RGCs.40 Researchers also speculate a lag time of 10 years exists before 10% of the RNFL is lost in early glaucoma.41 In another study, glaucoma suspects had biannual pERGs over six years, approximately half of whom went on to receive IOP-lowering medication.42 The pERG amplitudes of the untreated patients progressively declined, while the treated patients demonstrated stability or declined at a slower rate, suggesting lowering IOP may decrease the rate of RGC death or possibly reverse the RGC dysfunction in early disease.42
In patients with ocular hypertension, pERG testing can help predict stability or progression to glaucomatous optic neuropathy at least one year prior to visual field findings.43 While OCT GCA shows thinning due to RGC death, pERG may be able to detect RGCs with reduced function at an early, reversible stage of the disease.
Multifocal electroretinogram (mfERG). Although mfERG shows promise for diagnosing early glaucoma, positive studies have not translated into clinical diagnostic use in human populations.44,45 Investigators still struggle to quantify and extrapolate data to pinpoint RGC dysfunction in human populations.12
Full-field electroretinogram (ffERG). The photopic negative response (PhNR) of the ffERG may also correlate with RGC function. A study comparing RGC thickness with PhNR response shows a correlation within the central macula, the location of highly concentrated RGCs.46 Another study also shows correlation with decreased PhNR amplitudes in ocular hypertensive patients.47 These findings are promising for the early diagnostic stages of glaucoma, but more evidence is needed to apply this data in a clinical setting.
Although electrodiagnostic testing is not routinely performed in the primary eye care setting for glaucoma, it may one day serve as an adjunct tool in glaucoma management.
New diagnostic and imaging tools already aid in the early detection and treatment of glaucoma, as well as provide an avenue to better understand the pathogenesis of the disease. With more technology at our disposal, we must tailor our testing to each patient presentation by evaluating both modifiable and unmodifiable risk factors, patient demographics and long-term prognosis. Advancements help us better detect glaucomatous neuropathy at its earliest cellular manifestations, and we must consider the risk of functional visual loss over the patient’s lifespan and weigh this against any negative effects of therapy on daily living.
Below are some relevant case examples:
Case 1A 47-year-old male presented with best-corrected visual acuity of 20/20 OD and OS. Slit lamp examination revealed Krukenberg spindles on the corneal endothelium and midperipheral iris transillumination defects OU. His IOPs were 16mm Hg OD and 21mm Hg OS by GAT. Using 4-mirror gonioscopy, ciliary body band was seen 360 degrees, with 2+ pigment and a 35-degree angle approach with no anterior synechiae, recession or neovascularization of the angle OU. Upon dilation, we found his cup-to-disc ratios were 0.55 x 0.55 OD and 0.75 x 0.75 OS with pink and distinct margins OU. We ordered ancillary testing, including SD-OCT (Figures 1 and 2), SD-OCT-A (Figures 3 and 4) and visual field 24-2 (Figure 5), which depicted structural but not functional findings of pigmentary glaucoma OS. In this case of pre-perimetric, unilateral pigmentary glaucoma, peripapillary capillary density is affected in conjunction with the RNFL and ganglion cell structural findings prior to functional field loss. | |
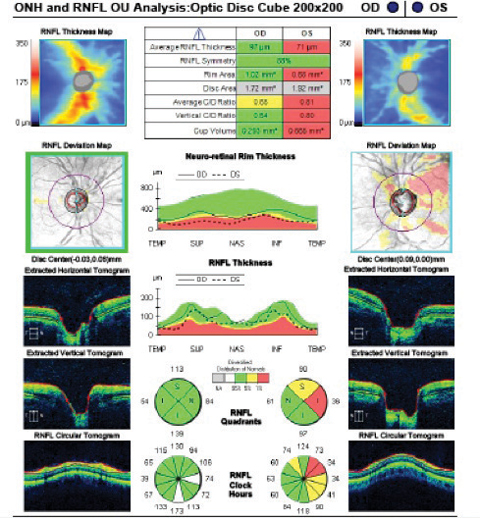 | 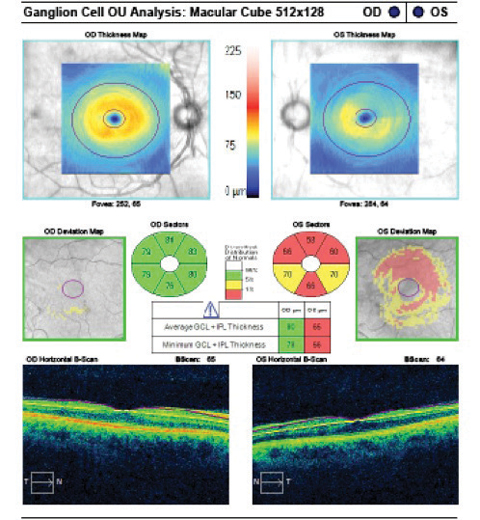 |
| Fig. 1. The SD-OCT optic disc cube scan shows significant asymmetry in average thickness OD to OS. Superior temporal RNFL thinning is seen on the deviation map compared with normative data OS. The quadrant and clock hour plots show associated thinning OS. Click image to enlarge. | Fig. 2. The GCA shows significant asymmetry in both the average and minimum thickness. Depressions in the RGC-inner plexiform layer thickness are illustrated in all sectors OS, greatest superiorly, correlating with the RNFL findings OS. Click image to enlarge. |
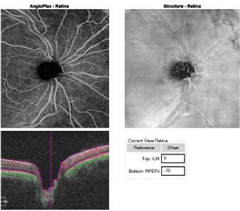 | 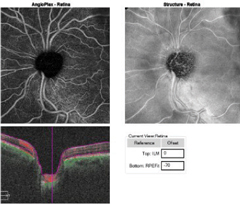 |
| Fig. 3a. This OCT-A angioplex en face scan of the patient’s OD (left) with segmentation of the retina shows a dense microvascular network surrounding the right optic nerve. Click image to enlarge. | Fig. 3b. The OCT-A angioplex en face scan OS (right) with segmentation of the retina shows the significant dropout in the peripapillary capillary network greatest superior temporal to the optic nerve. Click image to enlarge. |
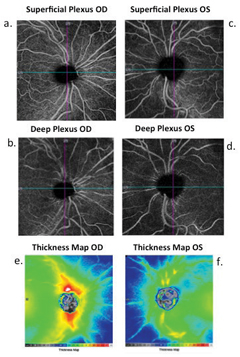 | Fig. 4. The superficial and deep capillary plexus segmentation of the OD and OS show significant asymmetry in the peripapillary capillary densities OD>OS (a and b, c and d, respectively). This asymmetry is most evident in the superficial capillary plexus OS. Images (e and f) depict significant thickness asymmetry OD>OS. Click image to enlarge. |
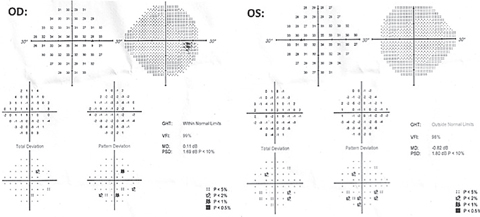 | Fig. 5. These 24-2 visual field results show scattered point depressions OD and OS with good reliability. No glaucomatous cluster defects were noted OU. Click image to enlarge. |
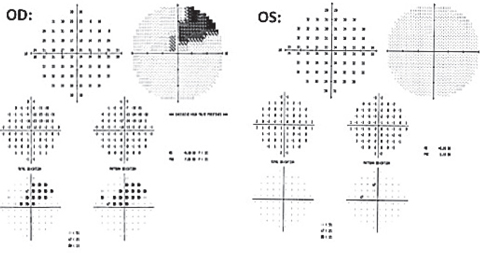
Case 2We saw a 64-year-old male with suspicion of glaucoma secondary to asymmetrical optic nerve cupping and mild ocular hypertension. We followed him with OCT of the RNFL and 24-2 visual field exams over a period of five years without treatment secondary to symmetrical RNFL exams and clean visual field exams OU. Visual acuity was 20/20 OU with normal anterior and posterior segment findings. IOP ranged from 22mm Hg to 24mm Hg in both eyes. Over the course of one year, an RNFL slit defect formed inferiorly at the inferotemporal neuroretinal rim OD, noted on fundus photos (Figure 1) and OCT RNFL deviation map imaging (Figure 2). A 24-2 visual field was performed to ascertain if there was a correlation with this new defect (Figure 3). While repeat 24-2 testing showed no glaucomatous cluster defects (Figure 4), 10-2 visual fields (Figure 5) and OCT imaging (Figures 6 and 7) showed structural changes that warranted attention. Secondary to the appearance of the OD optic nerve, as well as IOPs in the 20s OU, we started the patient on a prostaglandin analog in each eye at night, which lowered his pressures to 15mm Hg OU. The patient has since had selective laser trabeculoplasty with pressures in the low teens at an acceptable target range value OU. | |
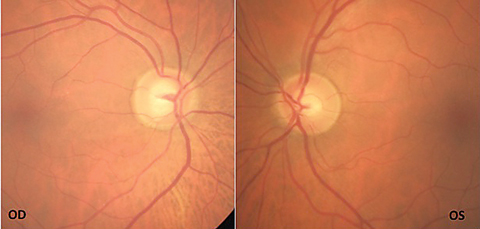 | Fig. 1. The patient’s fundus photos show inferior temporal rim thinning with vessel bayoneting within the optic nerve OD. Click image to enlarge. |
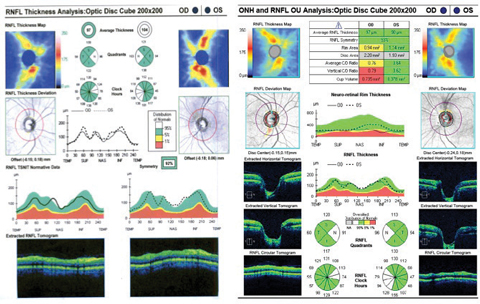 | Fig. 2. Although there is a slit defect inferiorly, the RNFL analysis still interpreted this area as normal compared with the normative data. The OCT RNFL also depicts inferior temporal RNFL thinning on the deviation map and clock hour map OD compared with the normative data. Click image to enlarge. |
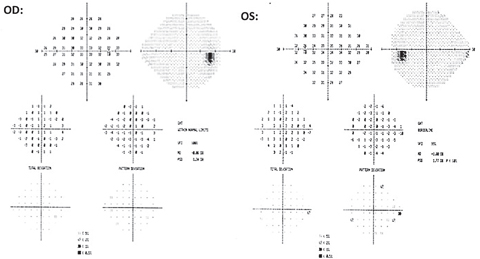 | Fig. 3. The 24-2 testing revealed no glaucomatous clusters OD and no correlation to the new RNFL defect OD. An inferior nasal cluster defect was noted OS and did not correlate to structural OCT findings. This cluster was found to be non-repeatable over successive fields. Click image to enlarge. |
 | Fig. 4. Repeated 24-2 testing shows point depressions and no glaucomatous cluster defects OU. Click image to enlarge. |
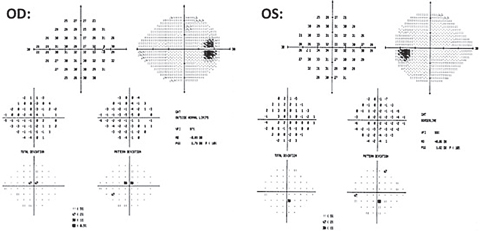
 | Fig. 5. A 10-2 central visual field was ordered to further investigate the 24-2 structural findings OD. It depicts a dense cluster superior/superior nasal to fixation correlating to the inferior temporal ganglion cell/inner plexiform thinning noted on GCA OD. Click image to enlarge. |
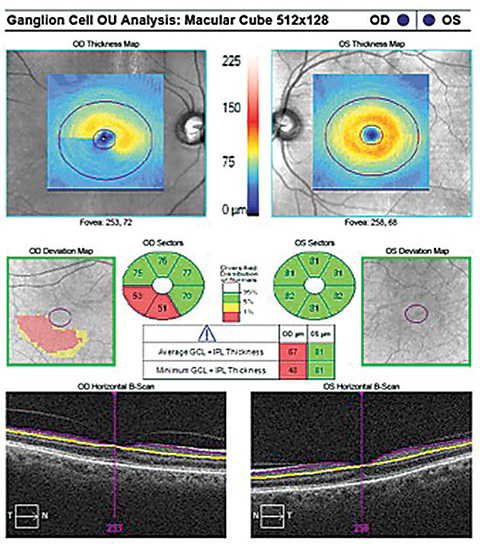 | Fig. 6. The GCA illustrates inferior temporal sector RGC-IPL thinning OD correlating to the clock hour thinning of the OCT RNFL OD. Click image to enlarge. |
 | Fig. 7. Serial OCT RNFL progression analysis shows progressive RNFL thinning inferior temporally OD over successive exams compared with baseline one and two. No RNFL thinning is noted OS over six exams compared with normative data. Click image to enlarge. |
Drs. Mazzarella and Cole are staff optometrists at the W.G. “Bill” Hefner VAMC in Salisbury, NC.
| 1. The National Eye Institute. Glaucoma, Open-angle. https://nei.nih.gov/eyedata/glaucoma. Accessed May 17, 2017. 2. Deol M, Taylor DA, Radcliffe NM. Corneal hysteresis and its relevance to glaucoma. Curr Opin Ophthalmol. 2015;26(2):96-102. 3. Medeiros FA, Meira-Freitas D, Lisboa R, et al. Corneal hysteresis as a risk factor for glaucoma progression: a prospective longitudinal study. Ophthalmology. 2013;120(8):1533-40. 4. Hon Y, Lam AK. Corneal deformation measurement using Scheimpflug noncontact tonometry. Optom Vis Sci. 2013;90:e1–8. 5. Oculus. The New Oculus Corvis STL received FDA clearance in the US. www.oculus.de/us/national-press/information/the-new-oculus-corvis-stl-received-fda-clearance-in-the-us. Accessed June 29, 2017. 6. Pinero D, Alcon N. Corneal biomechanics: a review. Clin Exper Optom. 2015;98(2):107-16. 7. Luce DA. Determining in vivo biochemical properties of the cornea with an ocular response analyzer. J Cataract Refract Surg. 2005;31(1):156-62. 8. Ehrlich JR, Haseltine S, Shimmyo M, Radcliffe NM. Evaluation of agreement between intraocular pressure measurements using Goldmann applanation tonometry and Goldmann correlated intraocular pressure by Reichert’s ocular response analyser. Eye (London). 2010;24(10):1555-60. 9. Mandalos A, Anastasopoulos E, Makris L, et al. Inter-examiner reproducibility of Ocular Response Analyzer using the waveform score quality index in healthy subjects. J Glaucoma. 2013;22(2):152-5. 10. David VP, Stead RE, Vernon SA. Repeatability of ocular response analyzer metrics: a gender-based study. Optom Vis Sci. 2013;90(7):691-9. 11. Haseltine SJ, Pae J, Ehrlich JR, et al. Variation in corneal hysteresis and central corneal thickness among black, hispanic and white subjects. Acta Ophthalmologica. 2012;90(8):626-31. 12. Leite MT, Alencar LM, Gore C, et al. Comparison of corneal biomechanical properties between healthy blacks and whites using the Ocular Response Analyzer. Am J Ophthalmol. 2010;150(2):163-168.e161. 13. Sedaghat MR, Mohammad Zadeh V, Fadakar K, et al. Normative values and contralateral comparison of anterior chamber parameters measured by Pentacam and its correlation with corneal biomechanical factors. Saudi J Ophthalmol. 2017;31(1):7-10. 14. Agarwal DR, Ehrlich JR, Shimmyo M, Radcliffe NM. The relationship between corneal hysteresis and the magnitude of intraocular pressure reduction with topical prostaglandin therapy. British J Ophthalmol. 2012;96(2):254-7. 15. Prata TS, Lima VC, Guedes LM, et al. Association between corneal biomechanical properties and optic nerve head morphology in newly diagnosed glaucoma patients. Clin Exp Ophthalmol. 2012;40(7):682-8. 16. Wells AP, Garway-Heath DF, Poostchi A, et al. Corneal hysteresis but not corneal thickness correlates with optic nerve surface compliance in glaucoma patients. Invest Ophthalmol Vis Sci. 2008;49(8):3262-8. 17. Congdon NG, Broman AT, Bandeen-Roche K, et al. Central corneal thickness and corneal hysteresis associated with glaucoma damage. Am J Ophthalmol. 2006;141(5):868-75. 18. Hazem H, Mahmoud L, Ahmed AZ. Corneal biomechanics in asymmetrical normal-tension glaucoma. Clin Ophthalmol. 2016;10:503-10. 19. Anand A, De Moraes CG, Teng CC, et al. Corneal hysteresis and visual field asymmetry in open angle glaucoma. Invest Ophthalmol Vis Sci. 2010;51(12):6514-8. 20. De Moraes CV, Hill V, Tello C, et al. Lower corneal hysteresis is associated with more rapid glaucomatous visual field progression. J Glaucoma. 2012;21(4):209-13. 21. Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study. Arch Ophthalmol. 2002;120(6):701-13. 22. Nakazawa T. Ocular blood flow and influencing factors for glaucoma. Asia-Pacific J Ophthalmol. 2016;5(1):38-44. 23. Flammer J, Konieczka K, Flammer AJ. The role of ocular blood flow in the pathogenesis of glaucomatous damage. US Ophthalmic Review. 2011;4(2):84-7. 24. Hayreh SS. Blood supply of the optic nerve head and its role in optic atrophy, glaucoma, and oedema of the optic disc. British J Ophthalmol. 1969;53(11):721-48. 25. Wang X, Jiang C, Ko T, et al. Correlation between optic disc perfusion and glaucomatous severity in patients with open-angle glaucoma: an optical coherence tomography angiography study. Graefe’s Arch Clin Exp Ophthalmol. 2015;253(9):1557-64. 26. Tobe LA, Harris A, Hussain RM, et al. The role of retrobulbar and retinal circulation on optic nerve head and retinal nerve fibre layer structure in patients with open-angle glaucoma over an 18-month period. British J Ophthalmol. 2015;99(5):609-12. 27. Satilmis M. Rate of progression of glaucoma correlates to retrobulbar circulation and intraocular pressure. Am J Ophthalmol. 2003;135(5):664-9. 28. Spaide RF, Klancnik JM, Cooney MJ. Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol. 2015;133(1):45-50. 29. Liu L, Jia Y, Takusagawa HL, et al. Optical coherence tomography angiography of the peripapillary retina in glaucoma. JAMA Ophthalmol. 2015;133(9):1045-52. 30. Leveque PM, Zeboulon P, Brasnu E, et al. Optic disc vascularization in glaucoma: value of spectral-domain optical coherence tomography angiography. J Ophthalmol. 2016;2016:6956717. 31. Lommatzsch C, Koch JM, Claussnitzer H, Heinz C. [OCT angiography of the glaucoma optic nerve]. Klinische Monatsblatter fur Augenheilkunde. 2017. 32. Lee EJ, Kim S, Hwang S, et al. Microvascular compromise develops following nerve fiber layer damage in normal-tension glaucoma without choroidal vasculature involvement. J Glaucoma. 2017;26(3):216-22. 33. Lee EJ, Lee KM, Lee SH, Kim TW. OCT angiography of the peripapillary retina in primary open-angle glaucoma. Invest Ophthalmol Vis Sci. 2016;57(14):6265-70. 34. Quigley HA, Hohman RM, Addicks EM, Green WR. Blood vessels of the glaucomatous optic disc in experimental primate and human eyes. Invest Ophthalmol Vis Sci. 1984;25(8):918-31. 35. Hood DC, Raza AS, de Moraes CG, et al. Glaucomatous damage of the macula. Progress in Retinal and Eye Research. 2013;32:1-21. 36. Traynis I, De Moraes CG, Raza AS, et al. Prevalence and nature of early glaucomatous defects in the central 10 degrees of the visual field. JAMA Ophthalmol. 2014;132(3):291-7. 37. Hood DC, Raza AS, de Moraes CG, et al. Initial arcuate defects within the central 10 degrees in glaucoma. Invest Ophthalmol Vis Sci. 2011;52(2):940-6. 38. Varma R, Tielsch JM, Quigley HA, et al. Race-, age-, gender-, and refractive error-related difference in the normal optic disc. Arch Ophthalmol. 1994 Aug;112:1068-76. 39. Kerrigan-Baumrind LA, Quigley HA, Pease ME, et al. Number of ganglion cells in glaucoma eyes compared with threshold visual field tests in the same persons. Invest Ophthalmol Vis Sci. 2000;41(3):741-8. 40. Curcio CA, Allen KA. Topography of ganglion cells in human retina. J Comparative Neurol. 1990;300(1):5-25. 41. Banitt MR, Ventura LM, Feuer WJ, et al. Progressive loss of retinal ganglion cell function precedes structural loss by several years in glaucoma suspects. Invest Ophthalmol Vis Sci. 2013;54(3):2346-52. 42. Ventura LM, Feuer WJ, Porciatti V. Progressive loss of retinal ganglion cell function is hindered with IOP-lowering treatment in early glaucoma. Invest Ophthalmol Vis Sci. 2012;53(2):659-63. 43. Bach M, Unsoeld AS, Philippin H, et al. Pattern ERG as an early glaucoma indicator in ocular hypertension: a long-term, prospective study. Invest Ophthalmol Vis Sci. 2006;47(11):4881-7. 44. Wilsey L, Gowrisankaran S, Cull G, et al. Comparing three different modes of electroretinography in experimental glaucoma: diagnostic performance and correlation to structure. Doc Ophthalmol. 2017;134(2):111-28. 45. Palmowski AM, Allgayer R, Heinemann-Vemaleken B. The multifocal ERG in open angle glaucoma-a comparison of high and low contrast recordings in high- and low-tension open angle glaucoma. Doc Ophthalmol. 2000;101(1):35-49. 46. Wilsey LJ, Fortune B. Electroretinography in glaucoma diagnosis. Curr Opin Ophthalmol. 2016;27(2):118-24. 47. Cvenkel B, Sustar M, Perovsek D. Ganglion cell loss in early glaucoma, as assessed by photopic negative response, pattern electroretinalgram and spectral-domain optical coherence tomography. Doc Ophthalmol. 2017 May;31:1-12. |

