 |
The vascular network that nourishes the retina is a complex and essential part of vision. Obstructions may occur within these blood vessels—namely, vein or artery occlusions—that impair the normal pathway of blood throughout the retina, potentially leading to devastating visual loss.
Much like many other parts of the body, the retinal vasculature attempts to troubleshoot these occlusions and form alternative routes to restore flow. In the case of venous occlusions specifically, these mechanisms may appear as collateral vessels, which are identifiable on clinical examination and may affect the prognosis or course of venous occlusion.
Collateral FAQs
When a retinal vein occlusion (RVO) occurs, collateral channels often form to bypass the site of occlusion and offer an alternative path for blood to nourish the affected retinal area. Unlike neovascularization, these vessels are pre-existing deep within the capillary bed and only fill when necessary.
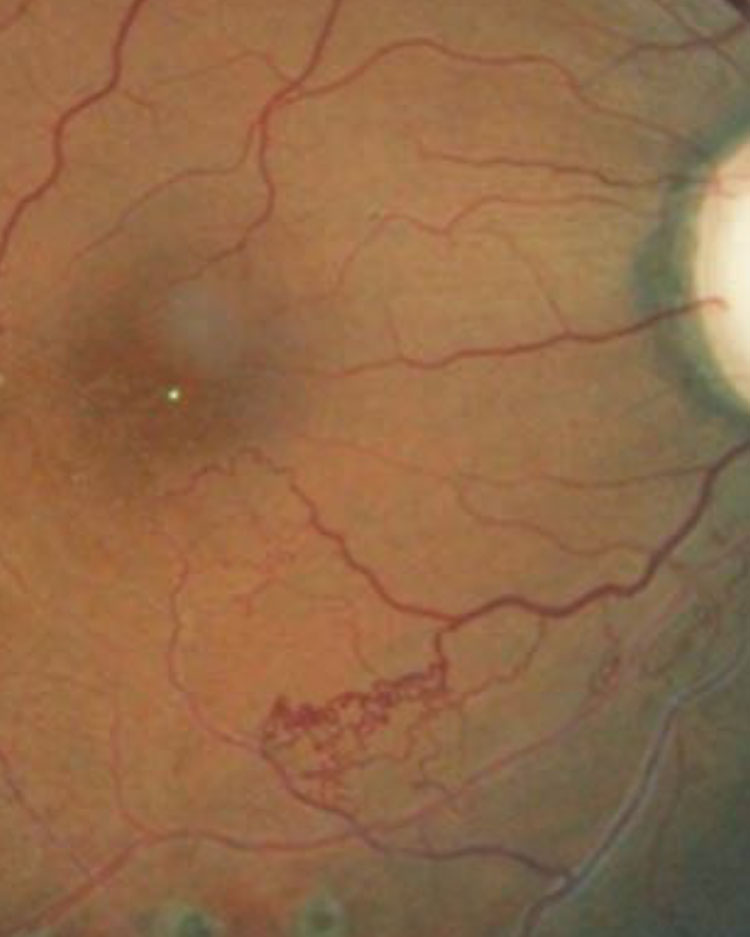 |
This patient’s retina exhibits collateral vessels bypassing an occluded vessel. Photo: Joseph W. Sowka, OD, and Alan G. Kabat, OD. Click to enlarge image. |
The filling of collateral vessels is thought to depend heavily on hemodynamic factors. Increases in hydrostatic pressure within the occluded area direct blood into areas of lower capillary pressure. This pressure gradient, resulting from hemodynamic stress, eventually leads to enlargement and filling of these already existing capillary channels in an effort to bypass the vein occlusion.1-4
Unlike other vascular responses in the retina, collaterals connect an obstructed vein to a neighboring unobstructed vein.2,3 In contrast, a vascular shunt, for example, connects dissimilar blood vessels, such as arteries, to veins.1,3
The most common cause of collateral formation is either branch or central RVOs; of these two, branch retinal vein occlusions (BRVOs) more frequently lead to collateral formation.1
Because BRVOs and central retinal vein occlusions (CRVOs) occur in different locations, collaterals that form in each case will also appear different clinically.4
In a BRVO, the obstruction occurs at sites where the retinal artery crosses a vein, or an arteriovenous crossing.5 The vein occluded at that site has surrounding, unobstructed channels that ultimately lead to the central retinal vein, where intraretinal collateral formation arises to restore blood flow in a BRVO, developing within weeks of the occlusion.1,4
On funduscopic examination, these areas appear as intraretinal tortuous blood vessels within the deep capillary bed, across the temporal raphe and other sites to bypass the occluded area. They may be difficult to distinguish from neovascularization; here, fluorescein angiography (FA) is useful. Initially following BRVO, there is minimal leakage of collateral vessels due to limited capacity and weak endothelial cells. With maturation, the vessels become larger and more stable.5
OCT-A: Go with the FlowOptical coherence tomography angiography (OCT-A) is a newer tool that allows for noninvasive visualization and analysis of retinal blood flow.1 OCT-A images are created with successive images of OCT B-scans that detect flow of motion from red blood cells within the retinal vasculature.6 The technology is faster, safer and less invasive than FA, and can also image deep capillary plexuses better than FA. Both FA and OCT-A can offer information on the location of ischemic retinal areas.7 Quantifying the area of nonperfusion allows the clinician to predict the severity and outcome of the ischemic event.6 |
Conversely, CRVO occurs at the level of the lamina cribrosa, where all branches of the central retinal vein are affected. As such, these collaterals are not intraretinal but are usually located on or around the optic disc and use the choroidal venous system for drainage.4
These vessels appear tortuous, with slow blood flow and cross the horizontal raphe, but may straighten over time or disappear when the obstructed site reopens.2,3,5 They take on the same caliber as a normal retinal vein, which is the type of vessel collaterals appear to replace.5
Warning Signs
Generally, the number or severity of collaterals seen in both types of RVO is associated with the extent of capillary nonperfusion; thus, the identification of collaterals on clinical examination is a potential marker for the degree of retinal ischemia that has taken place secondary to the occlusive event.2
It is difficult to ascertain whether collateral formation yields an improvement in visual outcome in RVOs or if they simply serve as markers for severity.
In BRVO, additional clinical features are associated with collateral formation, including a greater length of time of macular edema. This is potentially due to the longer duration of the ischemic event, generally, and the subsequent opportunity to develop collaterals in that timeframe.
Patients with collaterals have a smaller area of retinal hemorrhages that can be attributed to chronicity and resorption of hemes. Research also shows a correlation between younger patients with BRVO and the formation of collaterals, likely due to their ability to more readily undergo vascular remodeling.4
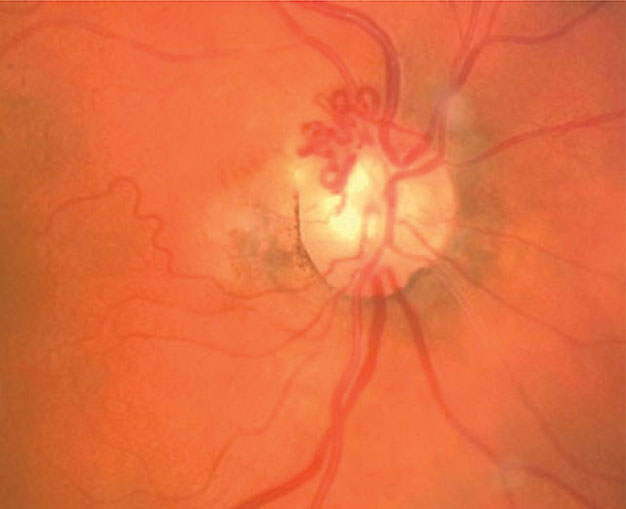 |
These collaterals formed over the optic disc in response to a CRVO. Click to enlarge image. |
Road to Recovery
The timing of laser therapy in the treatment of BRVO complications such as macular edema or neovascularization is critical for prognosis. Based on the Branch Retinal Vein Occlusion Study, laser treatment is considered three to six months following the BRVO. The typical time for collaterals to form and mature is approximately six to 24 months. Laser treatment in areas of nonperfusion prior to this can impair collateral vessel formation. However, using a laser to destroy collaterals that have formed can increase neovascular leakage.5
While collateral vessel formation is a natural mechanism to restore blood flow and reduce ischemic damage, research shows patients with CRVO and collaterals actually have poorer visual outcomes. One study found 52% of patients had acuity of 20/70 or worse, compared with 35% of CRVO eyes without collaterals.2,4 Patients with collaterals also had macular edema for twice as long and a lower chance of visual improvement compared with those who didn’t have collaterals.4
These factors indicate that CRVO with collateral formation is more of a marker for severity of ischemia. With the reduction in capillary density in severe cases, collaterals are more likely to arise, or patients who develop collaterals may have fewer available tributaries.2
In any vein occlusion case, identifying collaterals through clinical examination can be an important tool in determining the severity of the ischemic event and provide a clue as to the patient’s ultimate visual prognosis.
| Collaterals vs. Neovascularization | |||
| Origin | Appearance and Location | FA Testing |
Collaterals | Pre-existing vessels that fill to bypass an occlusion. | Tortuous and larger caliber vessels around the peripapillary plexus. | Minimal leakage in very early stages; no leakage in mature vessels. |
Neovascularization | New vessels formed by angiogenic cytokines and endothelial proliferation in response to ischemia.1 | Fine meshwork of vessels at the level of the vitreous.7 | Permeable to fluorescein. |
1. Freund KB, Sarraf D, Leong BCS, et al. Association of optical coherence tomography angiography of collaterals in retinal vein occlusion with major venous outflow through the deep vascular complex. JAMA Ophthalmol. 2018;136(11):1252-70. 2. Lee HE, Wang Y, Fayed AE, et al. Exploring the relationship between collaterals and vessel density in retinal vein occlusions using optical coherence tomography angiography. PLoS One. 2019;14(7):1-13. 3. Henkind P, Wise GN. Retinal neovascularization, collaterals, and vascular shunts. Br J Ophthalmol. 1974;58:413-22. 4. Weinberg DV, Wahle AE, Ip MS, et al. Score Study Report 12: development of venous collaterals in the Score Study. Retina. 2013;33(2):287-95. 5. Im CY, Lee SY, Kwon OW. Collateral vessels in branch retinal vein occlusion. Korean J Ophthalmol. 2002;16:82-87. 6. Heiferman MJ, Griebenow EJ, Gill MK, et al. Morphological implications of vascular structures not visualized on optical coherence tomography angiography in retinal vein occlusion. Ophthalmic Surg Lasers Imaging Retina. 2018;49(6):392-6. 7. Tsai G, Banaee T, Conti FF, et al. Optical coherence tomography angiography in eyes with retinal vein occlusion. J Ophthalmic Vis Res. 2018;13:315-32. |

