Because dry eye disease (DED), like its associated pathologies, most often involves an inflammatory process, logic may dictate that successful treatment requires nothing more than eliminating the inflammation. However, treatment is rarely so simple. Clinicians no longer concern ourselves solely with treating the inflammation—today, we aim to treat its underlying cause.
Dry eye and its components are not always easy to diagnose. As we learned from the DEWS report, DED is a multifaceted disease of the tears and ocular surface that results in symptoms such as dryness, itching, stinging, burning, blurry vision, a gritty or sandy sensation, excessive tearing, photophobia and, last but not least, visual disturbance.1-3
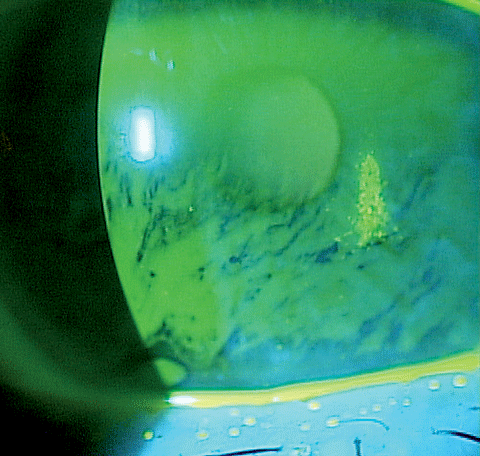 |
| Corneal staining in this dry eye patient shows epithelial barrier disruption. A variety of new tools to combat the factors associated with dry eye pathogenesis have hit the market in the last few years. Click image to enlarge. |
Once a diagnosis is made, subsequent treatment should include an evaluation for systemic or environmental factors, especially if the presentation is not consistent with the patient’s age or overall health.
This article provides an overview of the latest methods and devices available for optometrists to treat dry eye, specifically those involving evaporative dry eye.
The Rising Tide
Dry eye typically stems from one of two etiologies: aqueous deficiency or meibomian gland dysfunction (MGD).3 The curveball in diagnosing is that both can be present at once.3 Years ago, when dry eye was considered simply a nuisance, aqueous-deficient dry eye (ADDE) treatment included artificial tears and punctal occlusion, while MGD care was limited to a low-dose, long-term, oral doxycycline or tetracycline and warm compresses. Then, Restasis (cyclosporine, Allergan) came along and upended the conventional wisdom. Suddenly, the eye care community had a demonstrably efficacious way to increase tear production. Since then, the discipline has shown a growing interest in tackling what was often dismissed as a nuisance complaint.
And it’s a nuisance not just for MGD patients—who experience a lasting impact on their quality of life—but for clinicians as well, who are seeing a dramatic rise in DED prevalence.3-6 A 2012 Gallup study of dry eye sufferers projected growth of 10.2% over the next 10 years.6
Research shows that DED is present in approximately half of patients who wear contact lenses.7 In patients with diabetes, it is reportedly as high as 54%.8 In those with glaucoma or ocular hypertension, it is as high as 59%.9 A recent prospective study of patients scheduled for DED using the International Task Force scale—where two is considered moderate level of DED—indicated that 80.9% of patients scheduled for cataract surgery scored a two or higher.10 Yet, only 22.1% of these patients were previously diagnosed with DED.10
Something in the AirA 24-year-old white male presented in the morning with complaints of red, irritated eyes after working on a computer all day. However, upon examination, his eyes looked remarkably healthy. Another optometrist previously diagnosed him with computer eyestrain and prescribed computer glasses, which offered only some relief. The patient was rescheduled for an end-of-day appointment, during which he presented with highly irritated eyes with a 3+ conjunctival edema and 1++ injection OU. He was prescribed Lotemax (loteprednol etabonate, Bausch + Lomb) OU QID for two weeks. Upon follow up, the patient reported improvement of his symptoms; however, it was clear something in his workplace was causing the problem. He denied awareness of any window or vent creating airflow. He was asked to investigate any possible airflow in his work environment and taper the Lotemax over the next two weeks. At follow up, he reported that he stopped using the Lotemax the day after his last visit and had not had any problems since. He had found an air vent that was indirectly creating airflow on his face at work, and had adjusted the vent accordingly, which resolved the issue. The lesson: despite our ever-increasing arsenal of sophisticated therapies for dry eye, sometimes paying careful attention to the conditions under which our patients work, live and play will yield simple adjustments that can make lasting improvements. |
Add to these statistics the realization that the dry eye prevalence increases with age, and our aging population continues to grow, and it’s reasonable to be on the lookout for this disease process in all patients older than 65 years.11
The bottom line: you’re quite likely to encounter dry eye at an increasing rate, and with so many treatment methods available, you can be responsible for restoring quality of life to patients whether they developed dry eye after surgery, years of contact lens use or simply due to age.
Tear Film Anatomy
To properly treat MGD, clinicians should first have a firm grasp of the anatomy of the tear film and the origins of each layer—critical elements that help you determine the most appropriate treatment to resolve the underlying disease process that resulted in DED.
To briefly review, the tear film is made up of three layers: mucin, aqueous and lipid.12
The mucin layer, which is about 0.02µm to 0.05µm thick, is the innermost and thinnest layer. It maintains tear film stability and is a product of the conjunctival goblet cells.12 Epithelial cells produce glycocalyx to help bind the mucin layer to the epithelial surface.12
The aqueous layer, about 0.7µm thick, is the thickest of the three. This middle layer is formed from the secretions of the lacrimal and accessory glands. It contains electrolytes, proteins, antibodies, oxygen, carbon dioxide, minerals and glucose.12
The outer layer, which is comprised of lipids, is approximately 0.1µm thick and contains esters and glycerol as well as fatty acids. Its function is to prevent the evaporation of aqueous. This layer is a product of the meibomian glands and the glands of Zeiss.12
Investigators have shown that MGD is present in as many as 86 out of 100 cases of dry eye.5 The exact etiology of MGD is unknown. It is clear, however, that two pathologies seem to be key: decrease in the lipid layer due to keratinization of the terminal duct anatomy and an accumulation of cellular and lipid material within the duct lamina.13
Much of the research—and product development—has focused on enhancing the meibomian gland function by reversing or eliminating these two pathologies.
Clinical Approach
After diagnosing a patient with DED, clinicians must then determine whether it is ADDE, MGD or (as is common) a combination of the two. Next, determine which process is the most significant contributor to the presentation and direct your approach to therapy appropriately.
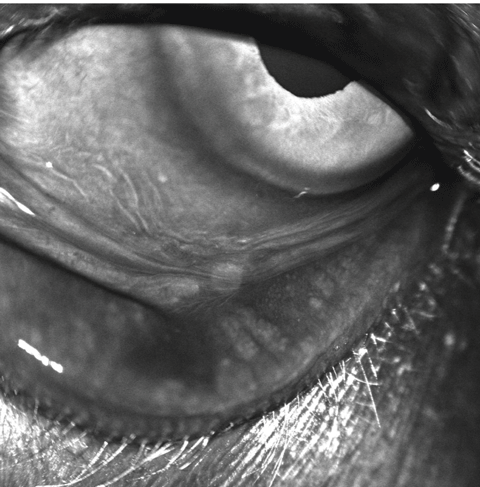 |
| This patient clearly demonstrates meibomian gland dropout. Click image to enlarge. |
Most patients who present with 2+ dry eye disease have minimal expression of oil from the meibomian glands, according to the DEWS report.3 Initially, an accurate differential diagnosis based upon staining and meibomian gland expression can be difficult to determine because the inflammatory process will create findings consistent with both ADDE and MGD. Too often, this can be misleading and start the practitioner and the patient on the wrong management path if the underlying cause is more significantly ADDE as opposed to MGD. Given that treatments for both of these diagnoses can take months to produce results, it is obviously essential to make the best initial diagnosis before long-term treatment is attempted.
The initial use of a topical steroid QID for two to four weeks can reduce the inflammation, allowing the clinician to make a more accurate initial diagnosis and initial treatment plan. The Report of the International Dry Eye Workshop states that, at least in some instances, periglandular inflammation contributes to MGD.3
In our practice, we typically initiate short-term use of a topical steroid to reduce the inflammation in and around the meibomian glands and help to restore the free flow of oil via digital expression. If this is successful, we’ll continue with reasonable confidence that treating the ADDE will improve the overall DED. Since this patient has ADDE, our medical regimen typically would be to taper the steroid over the first month’s treatment, introduce cyclosporine 0.5% OU BID and follow up in three months. These patients are given thorough education at diagnosis on the chronic nature of the disease and understand they will need to be treated and/or monitored routinely.
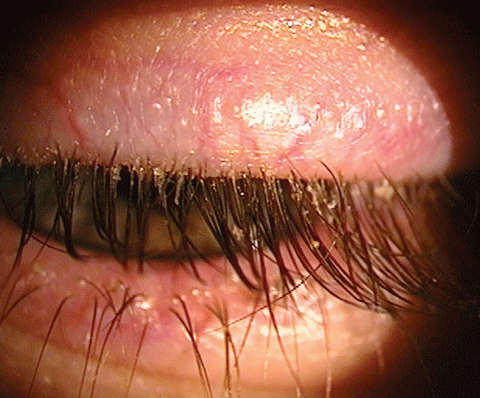 |
| Blepharitis is an inflammatory infection that can lead to dry eye disease. Several products on the market aim to treat this condition. Click image to enlarge. |
When reduction of the periglandular inflammation does not result in the desired clearance of meibomian gland blockage, we act more aggressively to improve the production of the lipid layer.
Many practitioners initiate treatment with both a steroid and topical cyclosporine. A 2012 study indicates that treatment with a combination of methylprednisolone 1% and cyclosporine 0.5% resulted in improved clinical findings at one month compared with patients who were treated with cyclosporine alone.14 This suggests that initial treatment with a steroid can help better address the underlying cause of the inflammatory process.
Lotemax gel (loteprednol etabonate, Bausch + Lomb) is particularly useful in this initial stage of treatment. An advantage of the drug’s gel form is that the suspension maintains an equal distribution of the active ingredient throughout the bottle without having to shake before using, which patients are apt to forget before instilling an eye drop. In non-gel drug delivery vehicles, this can result in a dilution of the active ingredient during the initial phase of the application and an oversaturation of the active ingredient toward the end of the treatment.
Therapy Beyond the Bottle
Several new treatment options have come along recently with the purpose of eliminating the constant need for drops, and mainstays like punctal occlusion continue to play a role. However, even with the added control these interventions offer, DED patients need to be advised that they will likely be treated for life.
Unless you have a practice limited to dry eye, it is rare that any one practice would support purchasing all available options. These products are in various stages of FDA approval, but each shows some degree of promise for dry eye treatment, and MGD specifically.
Punctal plugs have of course been in use for years as a treatment for DED as a way to decrease tear drainage and maintain adequate tear volume. Dextenza (Ocular Therapeutix), a promising new development that’s similar in concept though not truly a punctal plug, is an intracanalicular drug depot that will time-release a tapered dose of dexamethasone over a four-week period. By avoiding the sizable variations in drug concentration typical of topically administered drugs, this sustained-release approach is believed to provide more consistent therapy. It is currently under FDA review.
BlephEx (Rysurg), an in-office procedure akin to an aggressive lid scrub, is another promising MGD intervention. Though blepharitis is not the same as MGD, its presence also can interfere with meibomian gland secretions. This treatment looks similar to an alger brush used for foreign body removal, but uses a microsponge instead of a diamond head. BlephEx is a procedure performed by the clinician to exfoliate debris from the lid margin, usually following application of a topical anesthetic. It takes about six to eight minutes.
Avenova (NovaBay) is a newer lid hygiene method to be used by the patient at home, although it does require a prescription. It contains a stable formulation of hypochlorous acid 0.01%—the same molecule used by white blood cells to inactivate pathogens—in saline said to be designed for daily use. Avenova has broad-spectrum activity against common lid microorganisms, including Serratia marcescens, Methicillin-resistant Staphylococcus aureus, Staphylococcus epidermis and Staphylococcus haemolyticus.16 The patient sprays Avenova on the surface of the lids with their eye closed, then gently massages it into the lid with their finger.
LipiFlow (TearScience), is a thermal pulsation treatment system to improve meibum mobility. The device delivers heat to the inner eyelid along with adaptive pressure that cleans the glands without putting pressure on the globe. The procedure is done bilaterally in the office and takes about 12 minutes to elicit an expression of the meibomian gland. It can be performed just once or repeated based on the patient’s response.
Intense pulsed light therapy (IPL) has been FDA approved for more than 15 years to treat rosacea and remove superficial skin lesions. Such a device is commonly found in dermatology offices.
| Above, significant meibomian gland structural loss in a patient with moderate to severe MGD. Below, the clinical appearance of the ocular surface and lid in the same eye. Click image below to enlarge. |
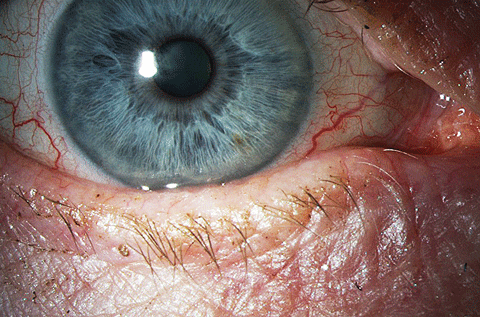 |
Ophthalmologist Rolando Toyos, MD, originated the idea of using IPL in eye care after a series of DED patients presented in his office demonstrating improved tear film characteristics without any change in their treatment regimen—all had been treated for rosacea with IPL.15 The device uses a xenon flash-lamp to emit wavelengths of light from 400nm to 1200nm. When placed on the light, a filter restricts the wavelength to the visible light range of 500nm. When applied to the skin, this band of light causes the blood cells in telangiectatic vessels to absorb the light, coagulate and, finally, to close the blood vessels.16 Some studies hypothesize that IPL treatment near the lid causes the abnormal blood vessels to close and note a positive effect on patients with MGD.
IPL is contraindicated in patients with darker pigmentation in that the pigment absorbs the light, making it ineffective. While IPL shows promise in treating MGD, scope of practice laws may hamper its widespread use by optometrists. However, numerous state optometry boards interpret the laws of that state as allowing this technology to be within optometry’s scope of practice. Prior to implementing any IPL treatment in-office, clinicians should check with their state board to make sure there is no prohibitive language that would prevent optometric use.
The management of MGD in clinical practice remains challenging, as patient compliance with doctor-recommended self-administered therapies is notoriously poor.17 So, to provide more effective dry eye management, practitioners should investigate in-office options that may offer clinical improvement that is both immediate and independent of patient compliance (or lack thereof). Some manufacturers offer a trial period to ensure their patient demographics will support the added out-of-pocket expense and cover the initial overhead. Once you hone in on how to best treat DED in your particular population, you can begin improving the quality of their lives for years to come.
Dr. Moore is senior clinical instructor for the Kentucky College of Optometry in Pikeville, KY and is currently vice president of the Association of Regulatory Boards of Optometry.
|
1. Market Scope. 2011 Comprehensive Report on The Global Dry Eye Products Market. St. Louis, MO: Market Scope, November 2011. 2. National Eye Institute. Facts about dry eye. www.nei.nih.gov/health/dryeye/dryeye. Accessed April 7, 2016. 3. The definition and classification of dry eye disease: Report of the Definition and Classification Subcommittee of the International Dry Eye Workshop (2007) Ocul Surf. 2007;5:75–92. 4. Kim KW, Han SB, Han ER, et al. Association between depression and dry eye disease in an elderly population. Invest Ophthalmol Vis Sci. 2011 Oct 10;52(11):7954-8. 5. Lemp M, Crews L, Bron A, et al. Distribution of aqueous deficient and evaporative dry eye in a clinic-based patient population. Cornea. 2012 May;31(5):472-8. 6. The Gallup Organization, Inc. The 2012 Gallup Study of Dry Eye Sufferers. Princeton, NJ: Multi-Sponsor Surveys, Inc; 2012. 7. Doughty M, Fonn D, Richter D, et al. A patient questionnaire approach to estimating the prevalence of dry eye symptoms in patients presenting to optometric practices across Canada. Optometry and Vision Science. 1997;74(8):624-31. 8. Manaviat M, Rashidi M, Afkhami-Ardekani M, Reza Shoja M. Prevalence of dry eye syndrome and diabetic retinopathy in type 2 diabetic patients. BMC Ophthalmology. 2008 June 2;8:10. 9. Leung E, Medeiros F, Weinreb R. Prevalence of ocular surface disease in glaucoma patients. J Glaucoma. 2008 Aug;17(5):350-5. 10. Trattler W, Reilly C, Goldberg D, et al. Cataract and dry eye: Prospective Health Assessment of Cataract Patients Ocular Surface Study. 2011 American Society of Cataract and Refractive Surgery Annual Meeting, San Diego, Calif. 11. Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003 Aug; 136(2):318-26. 12. Creech J, Do L, Fatt I, Radke C. In vivo tear film thickness and implications for tear film stability. Curr Eye Res. 1998 Nov 17(11)1058-66. 13. Tomlinson A, Bron A, Korb D. The International Workshop on Meibomian Gland Dysfunction: Report of the Diagnosis Subcommittee. Investigative Ophthalmology & Visual Science. March 2011;52:2006-49. 14. Byun Y, Kim T, Kown S, et al. Efficacy of combined 0.05% cyclosporine and 1% Methylprednisiline treatment for chronic dry eye. Cornea, 2012 May;31(5):509-13. 15. Papageorgiou P, Clayton W, et al. Treatment of rosacea with intense pulsed light: Significant improvement and long-lasting results. Br J Dermatol 2008:159;3;628-32. 16. Toyos R, McGill W, Briscoe D. Intense Pulsed Light Treatment for Dry Eye Disease Due to Meibomian Gland Dysfunction; A 3-Year Retrospective Study. Photomed Laser Surg. 2015 Jan 1;33(1):41–6. 17. Geerling G, Tauber J, Baudouin C, et al. The International Workshop on Meibomian Gland Dysfunction: report of the subcommittee on management and treatment of meibomian gland dysfunction. Invest Ophthalmol Vis Sci. 2011; 52: 2050–2064. |

