 Age-related macular degeneration (AMD) is a leading cause of vision loss in elderly, white Americans.1,2 Cumulative oxidative stress plays a role in the disease. So, it seems reasonable to suggest that dietary antioxidants and nutraceuticals will effectively combat the disease.
Age-related macular degeneration (AMD) is a leading cause of vision loss in elderly, white Americans.1,2 Cumulative oxidative stress plays a role in the disease. So, it seems reasonable to suggest that dietary antioxidants and nutraceuticals will effectively combat the disease.
During the last 20 years, several major studies have repeatedly demonstrated the benefits of nutritional supplementation in patients with existing AMD.3-12 Of these, AREDS exerted the most widespread, long-standing influence on clinical eye care and industry-based marketing. Can we expect a comparable impact from the long-awaited AREDS2 trial? Let’s take a look.
AREDS
The Age-Related Eye Disease Study was the first large-scale clinical trial to evaluate the efficacy of high-dose antioxidants on the progression of AMD. Its results showed that increased intake of antioxidants and zinc lowered the risk for disease progression by 25% in patients with intermediate or advanced AMD.6-8 Further analysis from AREDS suggested that higher dietary intake of lutein and zeaxanthin (L/Z), in addition to omega-3 fatty acids, decreased the risk of developing advanced AMD.10,11
Regarding adverse effects, subsequent research from AREDS found an increased incidence of genitourinary disorders associated with high-dose zinc supplementation.6 Additionally, research from other studies indicated that elevated intake levels of beta-carotene increased smokers’ risk for lung cancer.13 In addition to the dietary intake information from AREDS, the aforementioned safety considerations played a significant role in the testing parameters for AREDS2.
AREDS2
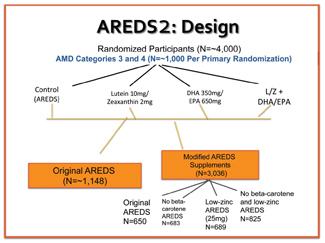
This five-year trial evaluated more than 4,000 subjects with intermediate AMD. Most participants were educated, well-nourished, elderly whites who already were taking a multivitamin. Sixty-five of the subjects had large bilateral drusen, while the remaining individuals had at least late AMD in one eye (either geographic atrophy or wet AMD).
The study’s primary goal was to determine if the addition of 10mg lutein, 2mg zeaxanthin and/or 1,000mg omega-3 long-chain fatty acids (350mg DHA and 650mg EPA) to the original AREDS formulation would further reduce the risk of progression to advanced AMD.
Additionally, the study evaluated the effects of refining the AREDS formula by eliminating beta-carotene and reducing the zinc doseage.12 Secondary outcomes included the effects of lutein, zeaxanthin and omega-3 fatty acids on the progression to moderate vision loss associated with AMD, as well as the prevalence of cataracts.12
The study design initially randomized approximately 1,000 subjects into four arms: original AREDS formulation (control); L/Z; omega-3 fatty acids; and a combination of L/Z with omega-3 fatty acids. A second classification further randomized the groups to receive a control or modified AREDS formula. The modified AREDS formula included either the presence or absence of beta-carotene, and either a high or low dose of zinc. (Note: There was no true placebo group, because all control subjects still took some form of an AREDS supplement. This was an ethical necessity, given that the AREDS formula was proven to be effective in the management of intermediate AMD.)
 What Did We Learn?
What Did We Learn?
The primary results of AREDS2 revealed that, when comparing the control group to the treated arms (L/Z; omega-3; L/Z and omega-3 combination), there was no significant reduction in the risk of progression toward advanced AMD. AREDS2 was an elobarate study with a complex design, involving secondary randomization and analyses. Some of the most pertinent statistical analyses are summarized below:12,14,15
• The addition of L/Z to the AREDS formula was associated with a 10% decrease in the risk of progression to AMD. This exploratory analysis evaluated the primary effects of taking L/Z vs. not taking L/Z.
When evaluating specific subgroups, it was determined that the addition of L/Z to an AREDS formula without beta-carotene yielded an 18% reduction in the risk of progression to advanced AMD.
• In another subgroup, patients in the lowest quintile of dietary intake for L/Z exhibited a 26% reduction in progression toward advanced AMD. Hence, the researchers determined that patients who would benefit the most from L/Z supplementation are those with the lowest dietary intake levels of L/Z.
• Omega-3 fatty acid supplementation did not yield a statistically significant reduction in the progression of AMD.
• Regarding adverse effects, there were no differences in mortality across all groups. Additionally, a subgroup analysis revealed that there were no differences in genitourinary tract infection incidence between the low- and high-zinc groups. However, beta-carotene supplementation was shown to double the risk of lung cancer in former smokers.
• Overall secondary analysis of AREDS2 did not indicate any benefit of L/Z supplementation on cataract progression.
• Although serum levels of lutein increased two-fold among subjects in the L/Z groups, a subgroup analysis showed that the increase was tempered when L/Z was given simultaneously with an AREDS formula that contained beta-carotene.
The Initial Verdict
The AREDS2 results suggest that lutein and zeaxanthin have a role in AMD management, and should replace beta-carotene in the original AREDS formula. With regard to omega-3 fatty acids, perhaps eye care providers should evaluate individual needs before recommending supplementation. Further studies that more directly scrutinize the effects of these nutritional supplements will clarify their true impact on AMD progression.
It is important to remember that our role as eye care providers also should include a proactive approach, ensuring decreased disease prevalence and progression. It is our duty to take preventive measurements whenever possible. So, patient management should emphasize counseling with regards to appropriate diet and nutrition. Although this was not the assessed in AREDS2, the eye care provider should consider proper supplementation (such as carotenoids) for at-risk or early-stage patients when deemed necessary.
Further analysis of the AREDS2 data could provide more specific information and potentially signify the advent of a new era in personalized medicine. Going forward, decisions on proper patient management may be made on an individual basis––and may even include genetic testing.
1. National Eye Institute. Statistics and Data. Available at: www.nei.nih.gov/eyedata. Accessed November 14, 2007.
2. The Eye Disease Prevalence Research Group. Cause and prevalence of visual impairment among adults in US. Arch Ophthlamol. 2004 Apr;122(4):477-85.
3. Hogg R, Chakravarthy U. Age-related macular degeneration and micronutrients antioxidants. Curr Eye Res. 2004 Dec;29(6):387-401.
4. Allen MJ, Jarding JB, Zehner. Macular degeneration treatment with nutrients and micro current electricity. J Orthomolec Med. 1998;13:211-14.
5. Seddon JM, Ajani UA, Sperduto R. Dietary carotenoids, vitamins A, C, and E, and advanced AMD: Eye disease case control study. JAMA. 1994 Nov 9;272(18):1413-20.
6. AREDS Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with Vitamin C and E, beta carotene, and zinc for AMD and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001 Oct;119(10):1417-36.
7. AREDS Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with Vitamin C and E, beta carotene, and zinc for AMD and vision loss: AREDS report no. 9. Arch Ophthalmol. 2001 Oct;119(10):1439-52.
8. Clemons TE, Chew EY, Bressler SB, et al. Research group national eye institute visual function questionnaire in the Age-Related Eye Disease Study (AREDS): AREDS report no. 10. Arch Ophthalmol. 2003 Feb;121(2):211-7.
9. Richer S, Stiles W, Statkute L. Double-masked, placebo-controlled, randomized trial of lutein and antioxidants supplementation in the intervention of atrophic AMD: Veterans LAST study. Optometry. 2004 Apr;75(4):216-30.
10. SanGiovanni JP, Chew EY, Clemons TE, et al. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study: AREDS Report No. 22. Arch Ophthalmol. 2007 Sep;125(9):1225-32.
11. SanGiovanni JP, Chew EW, Argon E, et al The relationship of dietary omega-3 long-chain polyunsaturated fatty acid intake with incident age-related macular degeneration: AREDS report no. 23.Arch Opthlamol. 2008 Sep;126(9):1274-9.
12. Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA. 2013 May 15:309(19):2005-15.
13. Smigel K. Beta carotene fails to prevent cancer in two major studies: CARET intervention stopped. J Natl Cancer Inst. 1996 Feb 21;88(3-4):145.
14. The Age-Related Eye Disease Study 2 (AREDS2) Research Group. Lutein/Zeaxanthin for the treatment of age-related cataract: AREDS2 randomized trial report No. 4. JAMA Ophthalmol. 2013 May 5:1-7.
15. AREDS 2 Research Group. The Age-Related Eye Disease Study 2 (AREDS2): study design and baseline characteristics (AREDS2 report number 1). Ophthalmology. 2012 Nov;119(11):2282-9.

