Diagnosis is rarely an exact science. Patients suffering from a multitude of ailments might display strikingly similar signs and symptoms, and subtle distinctions between different classifications of disease types can easily go unrecognized. Optometrists today benefit from an ever-increasing sophistication in our understanding of the causes and presentations of ocular disease. Still, outside of laboratory settings, hard data is hard to come by; we often must rely on clinical instincts and expertise when making the call.
That clinical conundrum has given rise to various in-office testing services. These seek to bridge the gap between the lab and the clinic by delivering hard, measureable evidence that may yield a diagnosis in relatively short order. With the advent of point-of-care (POC) testing, the clinician is able to obtain a result in a timely fashion—essentially in real time, for many—avoiding the hassle of sending the patient to a different facility, risking incomplete or inaccurate testing.
|
Get CLIA Certified for POC Testing
By John Rumpakis, OD, MBA, Clinical Coding Editor Many tests performed at the point of care require that your office have a Clinical Lab Improvement Amendments (CLIA) waiver. What is CLIA? In 1988, Congress passed CLIA to establish quality standards for all laboratory testing to ensure the accuracy, reliability and timeliness of patient test results, regardless of where the test is performed.1 CLIA-waived tests were also defined as simple laboratory examinations and procedures that are cleared by the Food and Drug Administration (FDA) for home use, employ methodologies that are so simple and accurate as to render the likelihood of erroneous results negligible, or pose no reasonable risk of harm to the patient if the test is performed incorrectly. These regulations were further revised in 1997 to make it clear that tests approved by the FDA for home use automatically qualify for CLIA waiver. How do you become CLIA certified? Performing CLIA-waived tests, and obtaining reimbursments for them, require your office be designated as a CLIA-approved laboratory and one of your doctors be designated and approved as a clinical lab director. Apply with CMS to get your CLIA certification. Having this certification for both the office and the physician is absolutely critical to perform and bill for these point-of-care tests. 1. Centers for Medicare & Medicaid Services website. Clinical Laboratory Improvement Amendments (CLIA). Available at: www.cms.gov/Regulations-and-Guidance/Legislation/CLIA/index.html. Accessed February 17, 2015. |
While many of our most confounding differential diagnoses remain purely clinical, several conditions now allow us to turn to POC testing to provide a more comprehensive exam than ever before. Let’s look at its use in practice, with a general overview of what the literature documents on the available tests and a series of illustrative cases.
Adenoviral Conjunctivitis
Red eye is one of the top emergent reasons a patient will report to an eye clinic. A child with a red eye is often kept home from daycare or school until infectious “pink eye” is ruled out. Health care workers and individuals who work with the public also take the day for fear of spreading an infection. However, a patient presenting with acute-onset ocular redness can have any number of potential underlying etiologies. Clinicians prefer to treat these patients empirically.
• Pathogen screening. AdenoPlus (RPS) is the only currently available CLIA-waived point-of-care test that detects adenoviral infections. Incorporating the AdenoPlus test into a red eye work-up can help increase the accurate diagnosis of viral conjunctivitis, narrow down appropriate treatment options and help reduce the potential spread of the virus.
In a recent study, researchers looked at the specificity and sensitivity of AdenoPlus at detecting the presence of adenovirus in tear fluid as compared with both viral cell culture with confirmatory immunofluorescence assay (CC-IFA) and polymerase chain reaction (PCR).1 Viral culture uses a series of primary cell lines that allow the replication of a wide variety of clinically relevant viruses. Specimens are inoculated onto cell culture monolayers and monitored for changes that occur in response to viral infection.2 Immunofluorescence assay (IFA) uses fluorescent-labeled antibodies to detect specific target antigens and is used extensively scientifically and clinically to identify specific antigens.2 PCR allows for the synthesis of a new strand of DNA complementary to the offered template strand (e.g., adenovirus). DNA polymerase can synthesize DNA complementary to a presented target sequence, resulting in the production of billions of copies of the unknown virus.2
The study results demonstrated that when compared with CC-IFA, AdenoPlus showed a sensitivity of 90% and specificity of 96%. When AdenoPlus was compared only with PCR, it showed a sensitivity of 85% and specificity of 98%. When compared with both CC-IFA and PCR, AdenoPlus showed a sensitivity of 93% (27/29) and specificity of 98%.1
|
Adenoviral Conjunctivitis Case
The patient reported that his right eye became red a “couple of days ago” and he has noticed the eye feeling “tender,” but not itchy. He reported taking Claritin for allergies. He stated that he hasn’t been sick recently, is not a contact lens wearer and notices some excess watering, especially outside. He reports his wife and daughter recently developed red eyes and were being treated.
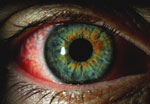
Testing. VA was measured at 20/25 uncorrected in both eyes with EOMs, pupils and visual fields unremarkable. A slit lamp examination (see image) revealed 2+ injection of the right eye and trace injection in the left eye. His cornea was clear, with no staining by sodium fluorescein or lissamine green, and trace papillae were noted on the upper lids. We performed the AdenoPlus test, collecting a sample from his right eye.
Diagnosis and Treatment. We diagnosed the patient with epidemic keratoconjunctivitis (EKC) secondary to the severity of the patient’s symptoms, his family members having a similar red eye and the intensity of the positive response on the AdenoPlus test. We treated him with betadine, prescribed a topical steroid to be used QID and informed him that he would not be able to return to work due to the potential infectious etiology of his condition. Educating him included advising him to limit contact with others, not share towels or washcloths and to wash his hands frequently. He was asked to return to the clinic the next day for follow up. Analysis. This case demonstrates the importance of being able to provide a patient with a concrete diagnosis, the appropriate management and patient education, in a timely manner. Until the AdenoPlus test confirmed a viral infection, the patient was convinced his symptoms were a result of allergies and dryness, and sought clearance to return to work. Performing the AdenoPlus test gave us an objective result indicating that his red eye was secondary to a viral infection and not allergies. The test prevented his coworkers and customers from exposure. 1. Sambursky R, Trattler W, Tauber S, et al. Sensitivity and specificity of the AdenoPlus test for diagnosing adenoviral conjunctivitis. JAMA Ophthalmology. 2013;131(1):17-22. |
Dry Eye
Numerous factors can predispose patients to dry eye, including increasing age, contact lens wear, meibomian gland dysfunction and autoimmune disease.3 However, diagnosis is challenging, as patients may report with significant symptoms but no obvious signs of ocular surface compromise, or vice versa.3
Dry eye patients often experience inflammation associated with an increase in matrix metalloproteinases (MMPs). These proteolytic enzymes are produced by compromised epithelial cells on the ocular surface. Investigators have consistently shown MMP-9 elevated in the tears of patients with dry eyes.4
|
Tear Osmolarity Case
Testing. Uncorrected VA at distance was 20/20 in both eyes. Entrance skills were unremarkable. Prior to performing a slit lamp exam, a TearLab test was performed. The right eye’s osmolarity was measured at 306mOsmol/L while the left was 347mOsmol/L.
Diagnosis and treatment. We shared our results with the surgeon and indicated that her osmolarity and staining results indicated a dry eye condition even though the Schirmer results weren’t significantly different. The surgeon recommended the patient begin Restasis (cyclosporine, Allergan) treatment prior to blepharoplasty surgery. Analysis. Although the Schirmer values were slightly lower in the left eye, they weren’t low enough to be considered significant. However, the elevated osmolarity and positive staining made it evident that the patient was experiencing dryness in her left eye, likely associated with contact lens use. Performing the tear osmolarity and combining that with the positive staining resulted in the patient beginning treatment for her dry eye and potentially averting a more serious condition post-blepharoplasty. |
• Measuring MMP. InflammaDry (RPS) samples tears for the presence of elevated MMP-9. Sambursky xet al. evaluated 143 patients with clinical signs and symptoms of dry eye, and 63 healthy individuals serving as controls. Participants were assessed as healthy controls or clinically diagnosed with dry eye using a combination of the Ocular Surface Disease Index, Schirmer tear test, tear break-up time, and corneal and conjunctival staining. InflammaDry demonstrated a sensitivity of 85% and specificity of 94% in diagnosing the dry eye cohort as compared with the healthy cohort.5
The ability to rapidly and reliably assess MMP-9 levels may lead to the clinician making an earlier diagnosis of dry eye and initiating management. Patients with MMP-9 levels less than 40ng/mL are thought to be in the “normal range” and not suffering from ocular surface disease, and levels above 40ng/mL is considered elevated and abnormal and indicative of some form of ocular surface disease.4 The determination of a patient having elevated MMP-9 levels, and therefore elevated inflammatory makers, may provide the clinician with an avenue of treatment that may have better efficacy, such as the use of anti-inflammatory agents (topical steroids, cyclosporine and other immunomodulators). An accurate measure of MMP-9 as an indication of inflammation on the ocular surface could be particularly important for the management of refractive or cataract surgery patients, where a stable tear film is important in post-operative outcomes.
• Testing tear osmolarity. The Dry Eye Workshop consensus opinion published in 2007 included tear osmolarity changes in its dry eye definition. This is now seen as a hallmark sign, and a possible central mechanism, of the pathogenesis of dry eye.6 A study by Bron et al. showed that, in healthy subjects, tear osmolarity is maintained in a relatively narrow range, with an average value of approximately 302mOsmol/L. Also of note, the study showed no significant difference in osmolarity between eyes (average around 6.9mOsmol/L).6 The study shows that increased osmolarity has a predictive relationship with dry eye inflammation.
TearLab provides a POC testing unit able to measure a patient’s tear osmolarity in-office. The unit obtains a 50nL sample of tears from a patient’s tear film and analyzes the sample, providing a measure of the patient’s tear osmolarity.7 The process of acquiring the tears is brief and straightforward, typically obtained from the lower temporal canthal area, resulting in minimal stimulation of reflex tearing.
|
MMP-9 and Sjögren’s Case
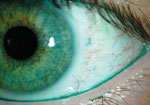
Testing. Slit lamp evaluation revealed 2+ MGD OU, 3+c onjunctival chalasis OU, and 1 to 2+ inferior SPK OU. RPS InflammaDry testing was positive OU. The results of the Sjö test was positive for the salivary protein (SP-1) EgA antibodies.
Diagnosis and Treatment. She was diagnosed with conjunctivochalasis, early Sjögren’s syndrome with keratoconjuntivitis sicca. She was placed on Restasis, warm compresses, and scheduled for LipiView. She was also scheduled for conjunctival resection with fornix rebuild.
|
|
In determining what “normal” osmolarity levels are for this protocol, 300 subjects were analyzed and a composite disease severity index was created from the following standard dry eye tests: TBUT, Schirmer I, corneal and conjunctival staining, meibomian grading, osmolarity and the Ocular Surface Disease Index (OSDI) for symptomology.7 Each patient was assigned a severity grade between 0 and 1, with 0 representing the absence of disease and 1 representing the most severe form of dry eye.7 TearLab correctly identified 88% of the normal subjects, 75% of the mild and moderate disease patients, and 95% of the severe dry eye patients. Investigators determined a value of 308mOsmol/L was the diagnostic cut off between “normal” and “mild dry eye.”7
According to colleagues who have incorporated TearLab into their practices, regional variability does exist for what is considered “normal” tear osmolarity. Geographical areas featuring lower humidity levels tend to show a higher baseline tear osmolarity. It may be important to evaluate your patient population and determine a normal range of osmolarity for your area.
Measurement of tear osmolarity is not only a diagnostic tool; it has potential for use in patient education and compliance. Patients who undergo treatment for dry eye and have repeated osmolarity testing will often ask for their “number” in hope that whatever treatment has been initiated has lowered their osmolarity score. You can use that concrete number, and patients’ interest in it, to spur adherence on their part.
• Testing for Sjögren’s. The autoimmune disease Sjögren’s syndrome affects the exocrine-producing glands throughout the body. Classically, patients who present with Sjögren’s suffer from dry eyes and dry mouth. Due to its multisystem involvement, this condition is often difficult to diagnose, as a patient may end up seeking care from multiple specialists, who may overlook the overall condition, as each specialist only addresses their particular area of expertise without putting the entire picture together. Two forms of Sjögren’s syndrome exist: primary Sjögren’s syndrome, which has no additional autoimmune disease associated, and secondary Sjögren’s syndrome, which can present in conjunction with an additional autoimmune disease (usually rheumatoid arthritis).8 Patients are often symptomatic with Sjögren’s syndrome for upwards of 10 years before being diagnosed with the condition.8 This can result in significant quality of life concerns and may also be life-threatening, as Sjögren’s syndrome is associated with a 16-fold increased risk of development of lymphoma.8
The Sjö (Nicox) test can help de-termine if a patient’s dry eye can be attributed to an underlying systemic etiology. A doctor or technician obtains blood on a collection card and sends it to a lab for analysis. A report is generated with results including rheumatoid factors, antinuclear antibodies, rho and la (Sjögren’s syndrome antibodies) and three biomarkers (salivary gland protein-1, carbonic anhydrase-6 and parotid secretory protein) unique to the test that the company says can pick up Sjögren’s earlier than the others.
An early diagnosis of Sjögren’s syndrome is crucial for the long-term management of these patients.
|
Sjögren’s Syndrome Case
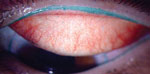
Courtesy of Dave Kading, OD A 38-year-old female presented with irritation and dryness. She noted the irritation had been increasing over the last few years. Dry eye evaluation noted a slight decrease in tear volume (through tear meniscus height evaluation), but what a decrease in the length of her meibomian glands was significant. A blink evaluation showed partial blinks in both eyes.
Testing. The lids were normal in appearance, but with pressure (using the TearScience Meibomian Gland Expresser) we noted a significant decrease in the flow of meibum. Her InflammaDry test was negative for inflammation. We performed Sjö testing. Staining was grade 1 corneal and positive on the lid wiper area (defined as that portion of the marginal conjunctiva of the upper lid that wipes the ocular surface during blinking) Diagnosis and Treatment. After reviewing the tests, our assessment was dry eye secondary to MGD and gland atrophy. Our treatment plan involved treatment with Lipiflow, lipid-based artificial tears and blink exercises. Analysis. At the patient’s follow up, she indicated improvement in symptoms. Clinically, we observed decreased lid wiper staining, normal meibomian gland secretion and resolution of the previous corneal staining. At the follow-up, we went over the Sjö test results; it indicated positive markers for Sjögren’s. This gave us the evidence we needed to begin the patient on a long-term Restasis (cyclosporine, Allergan) regimen, continue blink exercises and refer her to rheumatology for further evaluation and management. |
Diabetes
Diabetes is a major public health concern. Dealing with diabetes involves a complicated dance of managing blood sugar, blood pressure and cholesterol. Patients are asked to routinely check blood sugar levels, which gives them an indication of daily fluctuation. Additionally, an A1c (glycosolated hemoglobin) is a measure of a patient’s blood sugar over a three-month time period with normal values between 4% and 6%. A1c tests give the patient and practitioner an indication of the patient’s blood sugar control over an extended period of time.9
When reviewing diabetic patients’ medical histories, patients are often hard-pressed to give concrete numbers with respect to blood sugar control and, in particular, being able to recall A1c values.
• A1c testing units. In-office A1c testing units provide POC testing that can give doctors those concrete numbers. Several CLIA waived POC A1c testing units are available on the market. Some check only A1c. Others have a combined function and analyze a variety of lab tests.
Patients’ development of complications secondary to diabetes, such as diabetic retinopathy, kidney and heart disease, and peripheral neuropathy, is dependent on a combination of length of the disease state, blood sugar, blood pressure and cholesterol control.
The ability for an optometric physician to obtain a current A1c value provides valuable management information.
Spaeth et al. assessed the use of point-of-care A1c testing in a remote village in Australia and determined that the use of POC A1c testing improved patient A1c values as compared to laboratory A1c testing.10 On average the testing was able to lower A1c values by 1.5% by providing patients with a reliable and rapid indication of blood sugar control as compared with lab testing, where blood samples had to be sent off for analysis and results often took several days to weeks.10
|
Diabetes Case
Courtesy of Paul Chous, OD A 27-year-old white male presented for an eye exam. He has had Type 1 diabetes for 15 years. When asked what his last A1c value was, he said 120. He said his vision was fine, but his sister insisted that he get his eyes checked. The patient was taking Lantus and Humalog, his vision is correctable to 20/20 with an additional -1.25D (obvious myopic shift).
Testing. His dilated fundus exam showed mild nonproliferative diabetic retinopathy in both eyes with no observable diabetic macular edema; however, an OCT image revealed significant macular thickening and the presence of diabetic macular edema. Using the A1cNow test, his level was determined to be 12%. Diagnosis and Treatment. The patient returned with his spouse to discuss what was observed on the OCT and his current diabetes control. The patient was educated that his control is not at an acceptable level and he was referred to an endocrinologist. He was placed on an insulin pump with continuous blood sugar monitoring. His A1c dropped to 7% over the next year with no changes in his nonproliferative diabetic retinopathy or OCT over 23 months. He has since referred some 20 patients to the office for vision exams—patients with and without diabetes. Analysis. Optometric physicians can order other tests and even obtain the necessary sample from the patient to be analyzed, but these tests do not provide a result immediately and often require a day or two to obtain the results. |
AMD
Most patients with age-related macular degeneration are in the “dry” or non-exudative category where vision loss is modest and presents with retinal findings such as drusen, RPE clumping and potential atrophy. Patients who develop significant vision loss are typically in the “wet” or exudative stage, where patients have developed a choroidal neovascular membrane in the macular area, resulting in significant scarring.11
• Genetic tests. Risk factors for the development of AMD include increasing age, smoking, UV exposure, family history and genetic profile. Genetic tests may provide a sense of risk of developing AMD.12
Macula Risk PGx (ArcticDx) combines a patient’s current AMD status, genetic predisposition and non-genetic risk factors to determine the two-, five- and 10-year risk of developing advanced AMD, defined as either geographic atrophy or choroidal neovascularization (CNV).12
RetnaGene (Valeant) is a genetic test for Caucasian patients age 55 and older with early or intermediate AMD that evaluates genotype, phenotype, age and environmental risk (e.g., smoking status) and predicts a patient’s individual risk for disease progression to CNV within two-, five- and 10 years. RetnaGene predicts a patient’s individual lifetime risk at age ≥55 for developing advanced AMD.12
Macula Risk PGx and RetnaGene tests rely on a sample of epithelial cells obtained by a buccal (cheek) swab and sent for analysis.
The addition of point-of-care testing gives optometrists a greater variety of diagnostic tools to consider and reduces our reliance on the old “wait-and-see” approach. Using these tests can provide doctors with first-hand evidence of disease—yielding an advantage in time and confidence that we can use to deliver early, targeted treatment.
The tests aren’t foolproof, and hundreds of other conditions are not currently within their purview, but this newer approach is a welcome complement to a clinical work-up.
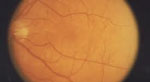
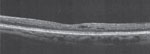
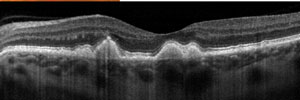
Courtesy of Jeffry Gerson, OD A 55-yea r-old female patient presented to the clinic concerned that she is “going to go blind” from AMD. The patient currently takes supplements. Testing. Evaluating her fundus and performing an OCT (see images) shows that the patient has a concerning phenotype with significant drusen in/surrounding the macular area. We discussed withher the potential role of genetic testing and that the costs would be covered by her medical insurance. She agreed to undergo testing. Analysis. The patient is still monitored every four months, but is much less worried because she knows the results of the genetic test. The genetic testing result eased the patient’s anxiety and helped her calmly live with her diagnosis. 1. Quick Reference Guide. AdenoPlus: An RPS Diagnostic Solution. www.rpsdetectors.com/en/wp-content/uploads/2013/08/FORM-MKT-014.2-AdenoPlus-QRG_Rev.pdf. Accessed 2/18/2015. | |||||||
2. Seal, DV, & Pleyer, U. Ocular Infection. New York: CRC Press; 2007.
3. Lin H, Yiu S. Dry eye disease: A review of diagnostic approaches and treatments. Saudi Journal of Ophthalmology: Official Journal of the Saudi Ophthalmological Society. 2014;28(3):173-81.
4. Sambursky R, Davitt W, Friedberg M, Tauber S. Prospective, multicenter, clinical evaluation of point-of-care matrix metalloproteinase-9 test for confirming dry eye disease. Cornea. 2014;33(8):812-8.
5. Sambursky R, Davitt W, Latkany R, et al. Sensitivity and specificity of a point-of-care matrix metalloproteinase 9 immunoassay for diagnosing inflammation related to dry eye. JAMA Ophthalmology. 2013;131(1):24-8.
6. Bron A, Tomlinson A, Foulks G, et al. Rethinking dry eye disease: a perspective on clinical implications. The Ocular Surface. 2014;12(2):S1-31.
7. Versura P, Campos EC. TearLab Osmolarity System for diagnosing dry eye. Expert Review of Molecular Diagnostics. 2013;13(2):119-29.
8. Goules A, Tzioufas A, Moutsopoulos H. Classification criteria of Sjögren’s syndrome. Journal of Autoimmunity. 2014;48-49:42-5.
9. Kovacic J, Castellano J, Farkouh M, Fuster V. The relationships between cardiovascular disease and diabetes: focus on pathogenesis. Endocrinology and Metabolism Clinics of North America. 2014;43(1):41-57.
10. Spaeth B, Shephard M, Schatz S. Point-of-care testing for haemoglobin A1c in remote Australian Indigenous communities improves timeliness of diabetes care. Rural and Remote Health. 2014;14(4):2849.
11. Solans-Laque R, Lopez-Hernandez A, Bosch-Gil J, et al. Risk, predictors, and clinical characteristics of lymphoma development in primary Sjögren’s syndrome. Seminars in Arthritis and Rheumatism. 2011;41(3):415-23.
12. Seddon J, Reynolds R, Yu Y, et al. Risk models for progression to advanced age-related macular degeneration using demographic, environmental, genetic, and ocular factors. Ophthalmology. 2011;118(11):2203-11.