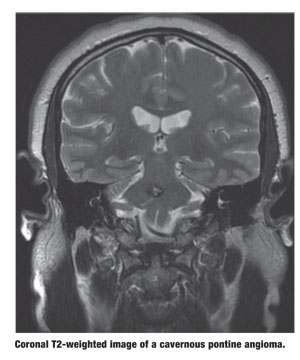
 |
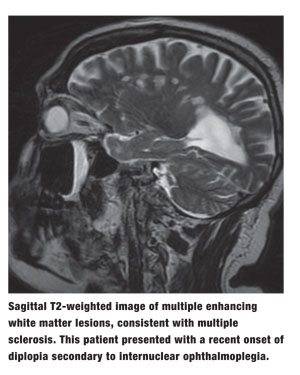 |
Here, we’ll review some of the most common neuro-ophthalmic emergencies and describe the role that optometrists play in the referral and comanagement processes.
Neuro-Ophthalmic Emergencies
Neuro-ophthalmic emergencies are conditions that pose a sudden threat to visual system function and/or systemic wellbeing.1 Some examples include acute painful Horner’s syndrome, painful cranial nerve palsies, optic disc edema, acute painless vision loss and severe headaches.
Once uncovered, time is of the essence. As a matter of reference, emergencies are classified into one of five triage levels:2
- Immediate
- Emergent (required evaluation within one to 14 minutes)
- Urgent (treatment required within 60 minutes)
- Semi-urgent (required evaluation within one to two hours)
- Non-urgent (a delay of up to 24 hours would make no appreciable difference in the overall clinical management of the condition)
It is generally accepted that the hallmark of a true eye care emergency is the potential for loss of sight (or even life) within 48 hours. By comparison, an ocular urgency is regarded as a condition that may result in significant morbidity or progress to an emergency at a later time (likely greater than 48 hours).3
The literature widely substantiates good decision making among optometrists. In a retrospective review of optometric malpractice payments from 1991-2008, researchers showed that less than 2% of payments stemmed from a failure or delay in the referral/consultation process.4
While knowledge of the condition and the associated standard of care can raise awareness of how rapidly the condition should be referred, consultation with a specialist via personal contact or phone conversation is the best method of determining the proper timetable.
• Giant cell arteritis (GCA) is the quintessential medical emergency in eye care, because blindness is imminent without proper disease recognition and prompt treatment.5 According to Sohan S. Hayreh, MD, there is no definitive “safe intervention period” that will ensure visual preservation between the time of GCA diagnosis and the initiation of corticosteroid therapy.6 Nevertheless, permanent sequelae from GCA are reported to occur in 30% to 60% of patients who do not receive prompt treatment.6
Once a patient has lost vision in one eye, the risk of GCA-related visual loss in the fellow eye is highest within hours to days.5 Contralateral eye involvement occurs in up to 75% of untreated patients.7 Timely corticosteroid use reduces the percentage of devastating visual loss by 14% to 20%.6 When patients present with any associated vision loss, the primary treatment goal is to retard the process––namely, reducing the risk of disease progression in the affected eye and preventing any vision loss from occurring in the fellow eye.
• Pituitary tumor apoplexy is a rare neurosurgical emergency that results from an acute ischemic or hemorrhagic infarction within pre-existing pituitary adenomas.8 It is one of the most serious, life-threatening complications of pituitary adenoma.
The term “pituitary tumor apoplexy” should be reserved for cases that feature an abrupt onset of the most common signs and symptoms, including headache (92%), nausea and vomiting (54%), oculomotor paresis (54%), visual disturbances (50%) and/or altered mental state (42%).9 In more than 60% to 80% of documented cases, pituitary tumor apoplexy occurs spontaneously in previously asymptomatic patients or in those with undiagnosed adenomas.10,11
The extent of neurological and neuro-ophthalmological improvement following surgical intervention was reported to be higher when decompression was performed within one week of symptom manifestation.11
Mortality occurs in up to 12.5% of those with pituitary tumor apoplexy. Patients with signs/symptoms must be referred for an immediate neuro-ophthalmic work-up.
• Cervical carotid artery dissection is the underlying cause of approximately 2.5% of all strokes. More specifically, it accounts for 10% to 25% of ischemic cerebral events in patients younger than 45 years old.12 More than a quarter of patients with stroke secondary to cervical artery dissection develop relevant disability, while almost half report a decreased quality of life.13
Headache is reported in up to 69% of affected patients, and neck pain has been found in up to 49% of patients––typically in the ipsilateral upper anterolateral cervical region.14 Additionally, Horner’s syndrome may be found in up to 50% patients with carotid artery dissection.14 Patients who present with sudden-onset, painful, unilateral Horner’s syndrome should be suspected of carotid artery dissection until proven otherwise.12-14
Because most carotid artery dissections heal spontaneously, timely treatment is aimed at limiting neurological deficits by preventing thromboembolic complications and restoring blood flow.14 Meta-analyses that compare antiplatelet to anticoagulant therapy conclude that the former should be first-line treatment in these cases. Remember that hyperacute management of ischemic stroke restricts the use of antiplatelet medications to eligible candidates to within 4.5 hours of the event.15 Immediate referral is the standard of care.12-15
• Papilledema refers to swelling of the optic disc as a consequence of increased intracranial pressure (ICP). This is in contrast to papillitis, where disc edema is associated with inflammation or infection. In either case––visual loss occurs secondary to neuronal dysfunction after axoplasmic stasis. Typically, visual prognosis correlates to optic disc appearance during the acute presentation.16
Increased ICP with subsequent papilledema is observed in many conditions, including idiopathic intracranial hypertension, intracranial tumors, shunt obstruction, subdural hematoma, intracranial inflammation and subarachnoid hemorrhage.20 Clearly, the sequelae from these processes can threaten vision and signal the development of fatal disease. Observation of the condition requires an emergent response, including transfer of care to a neurology subspecialist. In some instances, an emergency department visit may be required to first establish the patient in that facility’s health system. The emergency physician can then obtain a consultation from the appropriate specialist.
• Aneurysm of an undiagnosed origin has been reported to cause mortality in up to 20% of affected individuals within 48 hours of onset. The incidence of third nerve palsy associated with internal carotid artery-posterior communicating artery (ICA-PCoA) aneurysm has been reported to be as high as 56%.18
Recent studies have shown that posterior circulation aneurysms (specifically, those of the posterior communicating artery) are more likely to rupture than others.18-21 Treatment of unruptured intracranial aneurysms in the setting of third nerve palsy is aimed at preventing rupture.
However, in patients with subarachnoid hemorrhage secondary to aneurysmal rupture, aneurysm treatment is intended to prevent a rebleed.19
Early endovascular management (within one week following CN III symptom onset) likely is the strongest positive predictor of clinical outcome.18 One study indicated that 50% of patients with ruptured PCoA and 48% of patients with unruptured ICA aneurysms had incomplete third nerve palsy at admission.20 Painful third nerve palsy must be considered to have an aneurysmal etiology, and always requires emergent referral.21
How to Inform the Patient
Prior to transferring a patient’s care, it is necessary to educate them regarding their condition. Inadequate physician-patient communication can result in poor compliance, as well as increased morbidity and mortality.22
The literature also recommends assessing the patient’s frame of mind, along with his or her ability to understand the alternatives and choices.22 If the individual is of sound mind, a patient-oriented approach can be used. In fact, “patient-centered care” is now the gold standard in Europe and the United States, and widely embraces the notion that “there should be no decision about me, without me.”23
When educating a patient, the standard informed consent process requires full disclosure of findings, a clinical justification for referral to another provider or facility, and a recommendation regarding the timeliness of that referral.24 Even in cases where the diagnosis isn’t firm, the potential issues must be discussed and the reason for referral must be explained.
To ensure that the patient is adequately informed, you should make a reasonable effort to overcome problems associated with intellect, language and/or impairment associated with illness.25
Further, you must always be considerate of any unique wishes articulated by individual patients. For example, some may prefer to use a local doctor, while others may prefer a doctor who is a member of a certain hospital network. Once the decision to refer occurs, or is denied against medical advice, that informed decision must be meticulously documented in the medical record.
Necessary Information
Previously established relationships with various generalists, specialists and subspecialists will help you more effectively navigate the entire neuro-ophthalmic comanagement process. Knowing the expectations and abilities of local experts can help streamline the referral process.
Extraneous diagnostic data takes time to gather and has the potential to confound the problem. For example, it is unnecessary to send an optical coherence tomography image of retinal nerve fiber layer thickening the patient’s PCP in a case of a presumed optic neuropathy. That image cannot be interpreted by the physician and may, in fact, create more questions. A fundus photograph would be a far superior choice. Additionally, a suspected diagnosis, recommended systemic workups and specific testing protocols should be included in any referral letter to a specialist.
An Efficient Referral Letter
Today’s technology permits multiple ways to communicate with other health care providers. In non-urgent cases, writing a letter or report is still common. In emergent cases, personal or phone communication with an accompanying letter is preferred.
When generating calls and letters, it is commonplace to communicate with more than one care provider. In cases where both primary and specialty care is necessary, two calls and/or letters can be generated to briefly describe the discovery and outline the suggested testing strategy for each participant on the comanagement team (see “Referral Letter Goals: Be Tailored, Swift,” below). Phone calls allow a more direct question-and-answer approach, with the primary benefit of immediate feedback. However, written communication often will include more detailed diagnostic information, and can be invaluable in the rare event of medicolegal action.
In either case, your communication should be specifically detailed, yet brief. The longwinded, four-paragraph narrative––with the classic introduction, the findings stanza, the treatment and prognosis section and the obligatory, “if you have any questions, do not hesitate to call,” closing––is passé.
It takes a lot of time to listen to or read, and often provides information that is already known by the recipient or has no relevance to them because he or she is unfamiliar with the terminology. Simply stated: the person on the receiving end wants to get a brief report about the patient’s condition and what you want him or her to do.
For simple referrals, one or two lines may be sufficient to describe the problem and what is necessary to complete all aspects of care. For more complex cases, a larger volume of information will be necessary. Be sure to include photographs of key examination elements. Such imagery will help illustrate the clinical severity of the presentation, as well as provide supplementary documentation.26 Also, write the patient’s identity and the date of service at the top of the page.
In some instances, your referral letter to the internist may include recommendations for additional testing and/or a specialist’s opinion.26 It is ideal for the patient’s internist to have a relationship with any specialist involved in the comanagement process.
However, if the patient’s insurance won’t cover a visit to that specialist, or the patient is uncomfortable with that individual or the practice’s location, alternatives must be handy.
If the case is non-urgent, the letter can be dictated or written during call-back time and sent via fax or traditional mail. Written reports should be copied and kept with the chart. Patients sent for immediate referral or treatment can transport the note to the specialist or internist directly.
Use the Phone for Emergencies
A phone conversation is the fastest way to communicate your findings to a consulting doctor, and is considered mandatory in emergent cases. Phone calls should be documented for time of day, date and conversation content. Also, it is critical to note the names and titles of the individuals you speak with.
A research group from Australia sought to determine the impact of physician communication on emergent patient management during a one-month period.27 They concluded that telephone calls from referring physicians resulted in significantly decreased emergency room wait times and more efficient patient management.27
Interestingly, the researchers determined that written communication did not, in fact, decrease wait times or enhance patient management efficiency.27
Therefore, in cases of true ocular emergency, a phone call is highly recommended to accelerate and improve patient care.
Ordering Tests
Ordering laboratory and imaging studies are part of ophthalmic practice. However, because the optometrist often isn’t an expert in treating underling systemic etiologies or interpreting the results of the studies compiled, you may wish to consider asking the internist or specialist to order the appropriate tests. The subtleties of knowing how much a test costs, how fast the data will come back, the invasiveness and/or specificity of a study, and how to interpret its findings are all relevant considerations.
The radiology literature reports an interpretation error rate ranging between 3% and 30%.28,29 If the ordering clinician cannot inspect the image and potentially detect an error, the patient’s life could be in jeopardy. Thus, a good rule of thumb: If you can’t interpret the testing results accurately, you should ask someone who can to order the study for you.
Keep in mind that it’s still reasonable (and helpful) to provide a list of potential differential diagnoses, as well as recommendations for supportive lab work. However, as a non-expert, basic medical ethics suggests that this part of the comanagement process should be left to those with specific training and experience.24,25,28,29
Scheduling a Follow-Up
Our primary goal in the neuro-ophthalmic management process is to prevent the loss of vision and life. Once this is accomplished, and confirmed via communication with our comanagement partners, ongoing ophthalmic management will remain essential for complete visual recovery.
Monitoring the progression of recovery also helps ensure that no new events or complications occur. So, it is advisable to schedule a follow-up visit based on the severity of the presentation:
• 24 to 48 hours for an ophthalmic emergency.
• One to two weeks for a systemic emergency with an ocular manifestation.
• Two to four weeks for a retinal or neuro-ophthalmic emergency.
• Two to four weeks after any visit with a specialist.
If it appears that the patient will not be compliant with a recommended follow-up schedule, you must send a certified letter that explains the condition as well as the need for medical and/or ophthalmic care. This provides medicolegal documentation of the quality care you provided, and officially establishes a paper trail regarding your concerns and attempted communications.
The primary care optometrist has a unique opportunity to both direct and participate in the care of patients with neuro-ophthalmic disease. Your level of involvement may vary depending on the practitioner’s location, setting and training, as well as the nature of the patient’s condition and his or her insurance.
Optometrists need to develop strong relationships within their medical community to provide prompt, effective care within a cohesive comanagement team. The influential role that we play in this partnership can be instrumental in preventing devastating visual compromise, or even mortality.
 |
 |
Dr. Rixon is an attending optometrist at the Memphis VA Medical Center in Tennessee.
Dr. Gurwood is a professor at Salus University in Elkins Park, Pa.
1. Khare GD, Symons RC, Do DV. Common ophthalmic emergencies. Int J Clin Pract 2008 Nov;62(11):1776-84.
2. Honigman LS, Wiler JL, Rooks S, Ginde AA. National study of non-urgent emergency department visits and associated resource utilization. West J Emerg Med. 2013 Nov;14(6):609-16.
3. Blaho K, Winbery S. How to prepare your office for medical emergencies. Available at: www.healio.com/optometry/practice-management/news/print/primary-care-optometry-news/%7B5f9b95b0-f1e6-4fdf-aeed-0b3864a3f344%7D/how-to-prepare-your-office-for-medical-emergencies. Accessed April 15, 2014.
4. Duszak RS, Duszak R. Malpractice payments by optometrists: An analysis of the national practitioner databank over 18 yrs. Optometry. 2011 Jan;82(1):32-7
5. Hayreh SS, Zimmerman B. Management of giant cell arteritis. Our 27-year clinical study: new light on old controversies. Ophthalmologica. 2003 Jul-Aug;217(4):239-59.
6. Hayreh SS, Biousse V. Treatment of acute vision loss in giant cell arteritis: Should we prescribe high-dose intravenous steroids or just oral steroids? J Neuroophthalmol. 2012 Sep;32(3):278-87.
7. Liu GT, Volpe NJ, Galetta S. Visual Loss: Optic Neuropathies. Neuro-ophthalmology: Diagnosis and Management, 2nd ed. Philadelphia: Saunders Elsevier; 2010:155-63.
8. Simon S. Neuro-ophthalmic manifestations and outcomes of pituitary apoplexy-a life threatening and sight-threatening emergency. N Z Med J. 2011 May 27;124(1335):52-9.
9. Dubuisson AS. Classic pituitary tumour apoplexy: Clinical features, management and outcomes in a series of 24 patients. Clin Neurol Neurosurg 2007 Jan;109(1):63-70.
10. Muthukumar N, Rossette D, Soundaram M, et al. Blindness following pituitary apoplexy: timing of surgery and neuro-ophthalmic outcome. J Clin Neurosci. 2008 Aug; 15(8):873-9.
11. Nawar RN, AbdelMannan D, Selman WR, Arafah BM. Pituitary tumor apoplexy: a review. J Intensive Care Med. 2008 Mar-Apr; 23(2):75-90.
12. Rao A, Makaroun M, Marone L, et al. Long-term outcomes of internal carotid artery dissection. J Vasc Surg. 2011 Aug;54(2):370-4.
13. Sarikaya K. Antiplatelets versus anticoagulants for the treatment of cervical artery dissection: Bayesian meta-analysis. PLoS One. 2013 Sep 5;8(9):e72697.
14. Patel RR, Adam R, Maldjian C, et al. Cervical carotid artery dissection: current review of diagnosis and treatment. Cardiol Rev. 2012 May-Jun;20(3):145-52.
15. Song S. Hyperacute management of ischemic stroke. Semin Neurol 2013 Nov;33(5):427-35.
16. Zhang J, Foroozan R. Optic disc edema from papilledema. Int Ophthalmol Clin. 2014 Winter; 54(1):13-26.
17. Gray L. A clinical guide to third nerve palsy: How to spot the signs of oculomotor nerve compromise. Rev Optom. 1994 Jan;131(1):86-94.
18. Santillan A, Zink WE, Knopman J, et al. Early endovascular management of oculomotor palsy associated with posterior communicating artery aneurysms. Interv Neuroradiol. 2010 Mar; 16(1):17-21.
19. Matsukawa H, Fujii M, Akaike G, et al. Morphological and clinical risk factors for posterior communicating artery aneurysm rupture. J Neurosurg. 2014 Jan;120(1):104-10.
20. Guresir E, Schuss P, Seifert V, Vatter H. Oculomotor nerve palsy by posterior communicating artery aneurysms: influence of surgical strategy on recovery. J Neurosurg 2012 Nov;117(5):904-10.
21. Purvin V, Kawasaki A. Neuro-ophthalmic emergencies for the neurologist. Neurologist 2005 Jul;11(4):195-233.
22. Siegel AM, Barnwell AS, Sisti DA. Assessing decision-making capacity: A primer for the development of hospital practice guidelines. HEC Forum. 2014 Jan 14. [Epub ahead of print]
23. Kramer MH, Bauer W, Dicker D, et al. The changing face of internal medicine: Patient centered care. Eur J Intern Med. 2014 Feb;25(2):125-7.
24. Hammami MM, Al-Gaai EA, Al-Jawarneh Y, et al. Patients perceived purpose of clinical informed consent: Mill’s individual autonomy model is preferred. BMC Med Ethics. 2014 Jan 10;15:2.
25. Bush WH, Albright DE, Sather JS. Malpractice issues and contrast use. J Am Coll Radiol. 2005 Apr;2(4):344-7.
26. Wagman, L. How to write GP referral letters. GP Online. Available at: www.gponline.com/Education/article/655978/how-write-gp-referral-letters/. Accessed April 15, 2014.
27. Montalto M, Harris P, Rosengarten P. Impact of general practitioners’ referral letters to an emergency department. Aust Fam Physician. 1994 Jul;23(7):1320-1, 1324-5, 1328.
28. Raskin MM. Survival strategies for radiology:some practical tips on how to reduce the risk of being sued and losing. J Am Coll Radiol. 2006 Sep;3(9):689-93.
29. Lee CS, Nagy PG, Weaver SJ, Newman-Toker DE. Cognitive and system factors contributing to diagnostics errors in radiology. AJR Am J Roentgenol. 2013 Sep;201(3):611-7.

