Some patients experience profound decompensation of the respiratory system requiring airway protection. While the thought of intubation is not pleasant, it is warranted in emergent cases such as acute trauma; septic shock; acute and chronic respiratory difficulties; respiratory compromise secondary to cerebrovascular accident, respiration failure secondary to neurological diseases (i.e., Guillain-Barre); respiratory distress following an asthmatic event; and acute hemorrhagic event (i.e., ruptured aneurysm).
In cases like these, patients may be intubated to protect the vulnerable airway from collapse or interruption.1 Intubation of the airway is accomplished via use of a laryngoscope blades to position plastic tubing into the trachea, through which respiration can be automatically driven by a positive pressure instrument known as a ventilator.2,3 All ventilators can be adjusted to create specific tidal volumes—volume of air per breath—in the setting of adjustable positive pressure (i.e., inflow push) and frequency of respiration (i.e., respirations per minute).4 Ventilators breathe for the patient to protect and preserve the air pathway, as well as removing the muscular responsibility from individuals who lack the strength or neurologic control to complete respiration.4,5
The process works by creating positive pressure within the lungs.5,6 Ventilators may be set to two modes: volume mode and pressure mode.5 Volume mode permits the titration of each breath, and is referred to as tidal volume.5,7 Pressure mode regulates the force of air introduced into the lungs.5,6
Respiration frequency can also be adjusted to control respirations per minute. The combination of these three variables can be used to control the peak airway pressure and inspiratory flow, generating a flow pattern that is customizable for each individual and situation.4,7
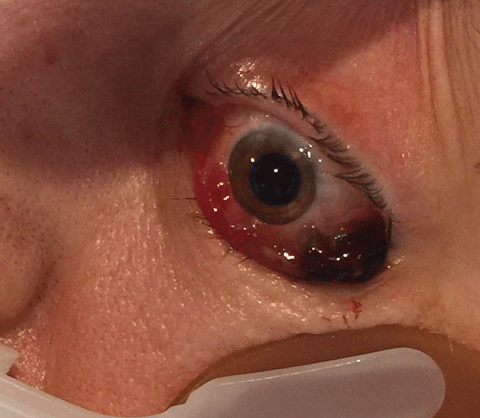 |
| Fig. 1. After introduction of the ventilator, this patient developed acute and intractable bilateral proptosis.Click image to enlarge. |
The mode of respiration can also be defined. Triggered Respiration is selected to permit respiration based on an inspiratory effort by the patient within a time boundary. Cycled Respiration is selected to deliver breaths following the reaching of a set tidal volume.6
To intubate the trachea, the base of the neck must be aligned so that the three principle axes are located in their proper positions:3,4
• The oral axis line—the plane of the tongue and the hard palate.
• The posterior pharyngeal—which defines the pharyngeal axis.
• The tracheal axis.3,4
When a patient is supine, the oral axis is perpendicular to the surface of the bed, the pharyngeal axis sits on an angle from the posterior wall of the pharynx to the larynx and the tracheal axis is positioned at downward angle from the larynx.3,4 Pillows and pads along with techniques designed to preserve the safety of cervical spinal alignment are used to bring the head and neck into position for placement of the tubing.3,4
To bring the oral axis into alignment, a jaw-thrust technique preserves neutral head position as the supportive aids stabilize the pharyngeal and tracheal axes. A clear line of sight permits the passage of the intubation tubing into the airway.3,4
Ventilator-induced Ocular Complications
The circulatory system is cyclic, owing its efficiency to an un-inter-rupted circle of movement. Whenever alteration occurs to the physical pathway (blood vessel impingement by thrombosis, blood cell pathology, restriction or compression) or to the pressure within the circulatory system, perfusion can be altered.8,9
Intubation Technique1. The endotracheal tube is held in the provider’s right hand and introduced into the right side of the patient’s mouth along with the laryngoscope blade.2. The curve of the endotracheal intubation tube is directed anteriorly. 3. It is advanced toward the glottis from the right side of the mouth so as not to obscure visualization of the glottic opening. 4. Once the proximal end of the tube’s cuff is 1cm to 2cm past the vocal cords, placing the distal end of the tube midway between the vocal cords and carina the laryngoscope blade is removed from the patient’s mouth. 5. Once in place, the cuff of the endotracheal tube is inflated with air to create a seal against the tracheal mucosa. This seal facilitates positive-pressure ventilation of the lungs and decreases the likelihood of aspiration of pharyngeal or gastric contents, protecting the airway. 6. Upon correct placement (confirmation made via end-tidal CO2, auscultation for bilateral breath sounds, ballottement of cuff in the suprasternal notch), the endotracheal tube is secured in position externally with tape. 7. Use of the minimum volume of air in a low-pressure high-volume cuff will typically prevent leaks during positive ventilation pressure (20cm to 30cm H2O). It also minimizes the likelihood of mucosal ischemia resulting from prolonged pressure against the tracheal wall. 8. Serious complications attributable to prolonged or excessive endotracheal cuff pressures include tracheal stenosis, tracheal rupture, tracheoesophageal fistula, tracheocarotid fistula and formation of tracheoinnominate artery fistula. |
Ventilation in the setting of ex-cessive positive pressure, excessive volume per breath or increased frequency of respiration has the potential to increase cardiopulmonary congestion, as efficient communication between the heart and lungs is altered. When this occurs, blood can be prevented from filling the right ventricle, with the net effect of increasing systemic venous pressure.8,9
Additional circumstances that increase net systemic venous pressure include poor air exchange, in which the lungs cannot expel carbon dioxide efficiently, and cases when the jugular veins are mechanically occluded by the tie back used to externally stabilize the intubation apparatus.8,9
Case Report
A 58-year-old woman was admitted to the critical care unit of the hospital secondary to septic shock caused by candidiasis. She obtained the infection from leg sores that had developed as a result of poorly controlled non-insulin dependent diabetes mellitus. As the condition worsened, she developed respiratory compromise necessitating protection of the airway through intubation. Three days following introduction of the ventilator she developed acute and intractable bilateral proptosis (Figure 1). Following neuroimaging that demonstrated normal intracranial structures and vasculature, an ophthalmic consult was requested by the medical team. At the time of the consult, the patient’s diabetes and hypertension had been controlled and the systemic sepsis was declining. The chart demonstrated no allergies.
The bedside consult found the patient’s eyes covered with gauze pads that had been saturated with Polymyxin B/bacitracin ophthalmic ointment. Her best-corrected visual acuities measured 20/200 using a calibrated near-point card and her near vision spectacles. Extraocular muscle motilities were restricted in all fields of gaze in both eyes. The confrontation visual field test was normal. No afferent pupillary defect existed. No ability existed to retropulse the eyes and there was limited movement upon forced duction testing. Portable biomicroscopy revealed diffuse-exposure keratoconjunctivitis without frank corneal abrasions in both eyes. No evidence of iris neovascularization or iritis was present. Tonopen (Reichert) intraocular pressures (IOP) measured 28mm Hg in both eyes. Dilated fundus examination found 0.3/0.3 round optic nerves with no evidence of notching or disc edema. No choroidal folds or peripheral pathologies were present and there was no evidence of Candida retinitis. The patient had mild non-proliferative diabetic retinopathy without macular edema in both eyes.
Without evidence of neuro-opthalmologic disease (e.g., tumor, hemorrhage, orbital cellulitis, varicocele, carotid cavernous fistula, venous sinus thrombosis or acute proptosis from thyroid storm), a hypothesis was proffered implicating the pressure effect on the venous system exerted by the ventilator.
An acute strategy of topical antibiotic ointment patching was continued. With permission from the medical team, a topical anti-glaucoma agent (brimonidine 0.15%) TID in both eyes was added to lower the IOP to protect the nerves.
Following a conference with all of the members of the medical team, it was decided that extubation would commence when a determination of safety was made. Following extubation, the proptosis resolved and ocular motility, IOP and visual function normalized. The topical antibiotic ointments were reduced to in frequency from TID to BID and the topical anti-glaucoma agent was removed. The patient’s visual acuities returned to pre-disease levels (i.e., 20/40) and the untreated IOP was measured at 16mm Hg.
Differential Diagnosis of Proptosis1. MassAny type of mass in the orbit can cause proptosis. If the mass is in the intraconal fat, it causes a straight-forward proptosis; if it is in the extraconal fat, it causes proptosis induced at an angle. Examples: neoplasms resulting from lymphoproliferative disorders, leukemia or metastatic disease. 2. Thyrotoxicosis Thyrotoxicosis, also known as Grave’s disease, is the eye’s response to an autoinflammatory disorder of the thyroid gland. Women are affected four to five times more frequently than men and the symptoms range from mild to rapidly progressing. The extraocular muscles can be infiltrated with lymphocytes, macrophages, plasma cells, mast cells and mucopolysaccharides, resulting in proptosis, increased tendon-sparing extraocular muscle (EOM) mass, eye lid inflammation, swelling, eye lid retraction and EOM motility restrictions. 3. Carotid cavernous fistula A carotid cavernous fistula is defined by arterialization of the internal carotid artery and the cavernous sinus. This abnormal communication results in retrograde flow of arterial blood into the orbit from the ophthalmic veins. This fistula can occur spontaneously or secondary to trauma that tears the internal carotid artery. As arterial blood floods the venous sinus, pressure builds and proptosis occurs. 4. Orbital cellulitis Orbital cellulitis results from infection that has spread from the maxillary sinuses or ethmoid bone through the ophthalmic and facial veins to the orbital cavity. The walls of the sinus cavity are thin, and are incapable of serving as strong barriers against infection movement. When the pathogens reach the orbit, the orbital contents become inundated, infected and edematous, resulting in pain and loss of function. Proptosis results with hallmark decreased ocular motility. 5. Retrobulbar hemorrhage A retrobulbar hemorrhage can be accidental or a result of surgical trauma. It can develop very quickly or over minutes to hours. The blood can accumulate in several different locations. The signs and symptoms depend the volume and location of the hemorrhage. The rapid blood fixed against the rigid boundaries of the orbital cavity results in proptosis, pain, decreased ocular motility, nausea, vomiting and increased in intraorbital pressure with secondary elevation of intraocular pressure. |
Ocular Vascular Anatomy
The ocular venous system, like the systemic venous system, contains limited valves.10,11 The choroidal blood supply exits the eye via four to 12 venous voticosae, or vortex veins and the intraretinal blood supply exits the eye via the central retinal vein.
Ocular venous blood exits the eye depending on its anterior or posterior location:
• The majority of internal ocular venous blood exits the eye using the superior and inferior ophthalmic veins.
• The vortex veins, which drain blood from the choroid, join the superior and inferior ophthalmic veins depending upon their position.
• The inferior ophthalmic vein has an anastomosis with the inferior ophthalmic and infraorbital vein and generally drains blood from the adnexa and conjunctivae to the pterygoid plexus of veins in the face.10,11
• The superior ophthalmic and inferior ophthalmic veins drain into the cavernous sinus in the middle cranial fossa.10,12
• The central retinal vein can drain into the superior ophthalmic vein or drain directly into the cavernous sinus.10,12
• The pterygoid plexus of veins in the face return blood to the heart via the external jugular system.12
• The anterior muscular veins drain the extraocular muscles. They are variable in location and merge with one another, but ultimately connect with the superior and inferior ophthalmic veins.10,11
The Role Of The Improperly Set Ventilator
All ventilators maintain ventilation based upon three general principles: They supply a prescribed volume of air mixed with oxygen at an adjustable rate, pressure and frequency.6,16 Once intubation is established, the patient’s physiology is analyzed to calculate initial ventilation values.
The ventilator has the potential to directly disrupt the pressure gradient of pulmonary blood flow as the air mixture is forced into the lungs.5,6 When the values are set in such a way that the system cannot compensate, homeostasis is lost.
Ventilation settings resulting in excessively high pulmonary pressure have the potential to disturb the equilibrium of the circulatory system by increasing central venous pressure via congestion.6 Venous congestion begins in the pulmonary arteries and transmits itself through the entire cardiopulmonary system. Superior systemic blood flow backs up into the superior vena cava through the brachiocephalic vein, to the internal and external jugular veins, which drain all of the cranial contents.13
Increased central venous pressure affects the body system wide. It directly affects the ocular venous system and its ability to sustain equilibrium, reducing venous return and arteriole in-flow.13 While the ocular venous system is a limited valve network capable of tolerating bidirectional blood flow, in extreme cases compensation is not adequate, and back pressure is able to build.
When the ocular venous system does not drain appropriately, fluid and blood is trapped in ocular tissues and the orbit. This reduces egress of fluid and blood from the eye. Consequences of this include increased IOP due to reduced aqueous egress secondary to limited trabecular meshwork drainage and uveal scleral outflow-pathway restriction from interrupted episcleral venous flow. Conjunctival injection and edema, and proptosis can occur.10,17
The orbital contents consist of the globe, extraocular muscles, optic nerve, retrobulbar fat and the lacrimal gland. When back-pressure builds in the cavernous sinus, the rigid structures remain stable while the globe slips forward and proptosis occurs.17-24
Gravity’s effect on fluid pressure is another determinant in positive pressure ventilation and can change the vascular system’s performance. Changes in head height can produce pressure differences, affecting blood flow in the lungs as the body changes position.7,13 Head position can be used therapeutically to decrease blood flow to abnormal areas of the lung, such as in unilateral pneumonia, improving gas exchange, and in contradistinction can be used to increase flow.7,13
Lessons Learned
Ventilator-induced proptosis is a diagnosis of exclusion. Ventilator mechanics and cardiopulmonary physiology as they relate to ocular vascular flow create the potential for hemodynamic stasis and poor cardiac return, resulting in cases that range from the mild conjunctival fluid retention to the extreme, cavernous sinus congestion contributory to globe proptosis. Fortunately, the condition is rapidly reversible following either extubation or ventilator reset.
Ms. Lesniewski and Mr. Lilien are recent graduates of Salus University.
Dr. Gurwood is Co-Chief of Suite 3 at The Eye Institute and a professor of clinical sciences at Salus University.
Dr. Street serves as Assistant Professor at Salus University.
|
1. Kritek P, Choi A. Approach to the Patient With Disease of the Respiratory System. In: Longo DL, Fauci AS, Kasper DL, et al. Eds. Harrison’s Principles of Internal Medicine. McGraw-Hill, New York, NY 2012:2084-2086. 2. Epstein S, Hite R, Truwit J. Invasive Mechanical Ventilation. In: Epstein S, Hite R, Truwit J. Eds. Practical Guide to Mechanical Ventilation. John Wiley & Sons, Hoboken, NJ 2011:106-251. 3. Butterworth JF, Mackey DC, Wasnick JD. The Airway Management. In: Morgan GE, Mikhail MS, Murray MJ, Eds. Morgan & Mikhail’s Clinical Anesthesiology. McGraw-Hill, New York, NY 2013:108-141. 4. Henderson J. Airway Management in an Adult. In: Miller RD, Eriksson LI, et al. Eds: Miller’s Anesthesiology. Churchill-Livingstone, Philadelphia, PA 2010:1573-1610. 5. Hall JB, Schmidt GA. Management of the Ventilated Patient. In: Hall JB, Schmidt GA, Wood LH, Eds. Principles of Critical Care. McGraw-Hill, New York, NY 2005:481-498. 6. Celli BR. Mechanical Ventilatory Support. In: Longo DL, Fauci AS, Kasper DL, et al. Eds. Harrison’s Principles of Internal Medicine. McGraw-Hill, New York, NY 2012:2210-2214. 7. Grasso S, Mascia L, Ranieri VM et al. Respiratory Care. In: Miller RD, Eriksson LI, Fleisher LA, et al. Eds: Miller’s Anesthesiology. Churchill-Livingstone, Philadelphia, PA 2010:2879-2998. 8. Eskaros SM, Papadakos PJ, Lachmann B. Respiratory Monitoring. In: Miller RD, Eriksson LI, Fleisher LA, et al. Miller’s Anesthesiology: Churchill-Livingstone, Philadelphia, PA 2010:1411-1442. 9. Prendergast TJ, Chesnutt MS. Pulmonary Disease. In: McPhee SJ, Hammer GD, Eds. Pathophysiology of Disease. McGraw-Hill, New York, NY 2010:1120-1345. 10. Snell RS, Lemp MA. The orbital blood vessels. In: Snell RS, Lemp MA. Clinical anatomy of the eye. Blackwell Science Inc, Malden, MA 1998:277-293. 11. Remington LA. Orbital Blood Supply. In: Remington LA, Ed. Clinical Anatomy and Physiology of Visual System. Butterworth-Heinmann, United States 2012:202-217. 12. Netter FH. Head and Neck. In: Hansen JT, Benninger B, Brueckner JK, Carmichael SW, Granger NA, Tubbs RS, Eds. Atlas of Human Anatomy. Saunders, Philadelphia, PA 2011:1-148. 13. Hedenstierna G. Respiratory Physiology. In: Miller RD, Eriksson LI, Fleisher LA, et al. Eds: Miller’s Anesthesiology. Churchill-Livingstone, Philadelphia, PA: 2010:361-392. 14. Barrett KE, Boitano S, Barman SM, Brooks HL. Introduction to Pulmonary Structure and Mechanics. In: Barrett KE, Boitano S, Barman SM, Brooks HL, Eds. Ganong’s Review of Medical Physiology. McGraw-Hill, New York, NY 2012:621-640. 15. Netter FH. Thorax. In: Hansen JT, Benninger B, Brueckner JK, et al. Eds. Atlas of Human Anatomy. Saunders, Philadelphia, PA 2011:175-239. 16. Fessler MB, Fessler MB, Welsh CH, Welsh CH. Mechanical Ventilation: Invasive and Noninvasive. In: Hanley ME, Welsh CH. Eds. Current Diagnosis & Treatment in Pulmonary Medicine. McGraw-Hill, New York, NY 2003:280-291. 17. Oyster CW. The orbit. In: Oyster CW. The human eye structure and function. Cinaur Associates Inc., Sunderland, MA 1999:111-131. 18. Kanski JJ. Orbit. In: Kanski JJ, Bowling B, Eds. Clinical Ophthalmology: A Systemic Approach. Elsevier Limited, Philadelphia, PA 2011:79-120. 19. Dutton JJ. Orbital diseases. In: Yanoff M, Duker JS. Ophthalmology. Mosby-Elsevier, St. Louis, MO 2009:1450-1464. 20. Rootman J. An approach to diagnosis. In: Rootman J. Ed. Disease of the Orbit: A Multidisciplinary Approach. Lippincott Williams and Wilkins, Philadelphia, PA 2003:92-103. 21. Rootman J. Inflammatory Diseases. In: Rootman J. Ed. Disease of the Orbit: A Multidisciplinary Approach. Lippincott Williams and Wilkins, Philadelphia, PA 2003:462-514. 22. Marotta TR, Graeb DA, Vascular Lesions. In: Rootman J. Ed. Diseases of the Orbit: A Multidisciplinary Approach. Lippincott Williams and Wilkins, Philadelphia, PA 2003:514-554. 23. Bauer DC, McPhee SJ. Thyroid Disease. In: McPhee SJ, Hammer GD, Eds. Pathophysiology of Disease. McGraw-Hill, New York, NY 2010: 2846-3976. 24. Rubenstein JB, Virasch V. Allergic conjunctivitis. In: Yanoff M, Dukerm JS. Ophthalmology. Mosby-Elsevier, St. Louis, MO 2009:259-263. |

