12thAnnual Pharamaceuticals ReportCheck out the other feature articles in this month's issue: |
Patients presenting with ocular burning, stinging and sometimes painful symptoms of dry eye disease (DED) are becoming more common in today’s optometric practice. More than 16 million Americans are coping with this disease, which can have a significant impact on quality of life.1-3 In addition, DED is a growing problem for both mature patients and young adults.1,3
Although DED is widely encountered in eye care practices, the number of pharmaceutical agents available to combat it pales in comparison with other chronic diseases.1-3 Fortunately, new and upcoming dry eye medications may change the landscape of DED management for the better. Here is a comprehensive look at new dry eye drugs and a preview of treatment options of the future.
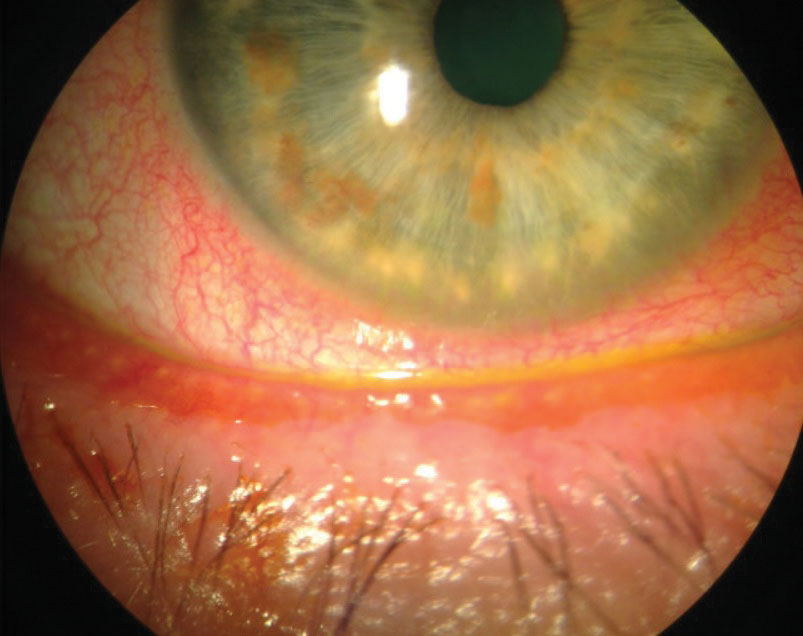 |
| Fig. 1. Conjunctival hyperemia is an excellent marker of ocular surface inflammation. A new topical steroid may help reduce this DED sign. Click image to enlarge. |
Primary Target: Inflammation
Several new therapeutic options are tackling this age-old problem associated with DED:
Cyclosporine (CsA). The last decade has seen a tremendous growth in our understanding of the pathophysiology of DED, and treatment options have shifted from solely artificial tear supplementation to disrupting the underlying disease processes such as inflammation.4 Many new and future dry eye drugs target the cycle of inflammation on the ocular surface that perpetuates this disease.5
Recently, Cequa (CsA ophthalmic solution 0.09%, Sun Pharma) joined Restasis (CsA 0.05% ophthalmic emulsion, Allergan), an immunomodulatory agent, and Xiidra (lifitegrast ophthalmic solution 5%, Takeda-Shire), an LFA-1 antagonist, as the third DED prescription drug approved by the FDA in 15 years. Like Restasis, Cequa is indicated for the treatment of keratoconjunctivitis sicca with the familiar BID dosage and supplied in single-use vials.6
Cequa is a first-in-class topical formulation with the highest FDA-approved concentration of cyclosporine and a novel nanomiceller technology, which overcomes the longstanding challenges of cyclosporine’s delivery to ocular surface tissues.6 The high hydrophobicity of the lipophilic CsA impedes the drug’s ability to penetrate the aqueous layer.6,7 Cequa’s nanomiceller formulation overcomes this barrier by forming small micelles or aggregates of amphiphilic molecules where the water-loving polar heads form an outer shell that faces the aqueous, while the non-polar, water-hating tails are sequestered inward to form a hydrophobic core.6
Through this delivery system, high concentrations of CsA can penetrate the ocular surface tissues and potentially provide better patient outcomes. Evidence suggests that improvements in signs and symptoms are seen earlier with Cequa than with other cyclosporine formulations available in the United States.
In a Phase III trial of Cequa, there were statistically significant improvements in Schirmer’s score at three months and ocular surface staining at one month.6 These improvements are earlier than the six months until improvement in Schirmer’s tear strip wetting and corneal staining seen with the lower concentration cyclosporine, Restasis, and earlier than the three month improvement in corneal staining seen with Xiidra.8,9 While a promising new drug, one medication won’t work for everyone, especially for a heterogeneous condition such as DED.
Although the reported mild to moderate adverse reactions of these DED medications are similar, responses to a drug, whether suboptimal or optimal, depend on the individual.6
Klarity-C (cyclosporine/chondroitin sulfate 0.1% ophthalmic emulsion, Imprimis Pharmaceuticals) is a new compounded non-preserved topical BID eye drop used for DED management. It combines the active ingredient CsA 0.1% with lubricants in a chondroitin sulfate emulsion. Its unique benefits are the higher concentration of cyclosporine and the chondroitin sulfate vehicle. Chondroitin sulfate is a lubricant that also has mild anti-inflammatory properties and can minimize instillation irritation, which is a common patient concern.10,11 Both Klarity-C and Restasis increase tear production and improve tear film instability and ocular surface staining, although Restasis has a better impact on goblet cell density.11
Ikervis (CsA 0.1%, Santen) and CyclASol (CsA 0.1%, Novaliq) may be future cyclosporine options in the United States. Ikervis, available in Europe, is a once-daily preservative-free formulation.12 It is a cationic emulsion containing CsA that leads to the long-lasting presence of CsA in the tear film and a higher bioavailability than the anionic CsA formulation of Restasis.12,13 As a result, with only once-daily dosing, Ikervis is well tolerated and significantly improves symptoms and signs of patients with severe DED by six months.12,14
CyclASol is a promising cyclosporine that uses a non-aqueous perfluorobutylpentane technology based on semifluorinated alkanes. This formulation may improve bioavailability of CsA and onset of efficacy. In a Phase II study, CyclASol showed a significant reduction in corneal and conjunctival staining compared with both the vehicle and Restasis with an onset of effect at two weeks.7
These formulation breakthroughs, in conjunction with the invalidation of patents for Restasis, have opened the door for more, and even generic, CsA options.
 |
 |
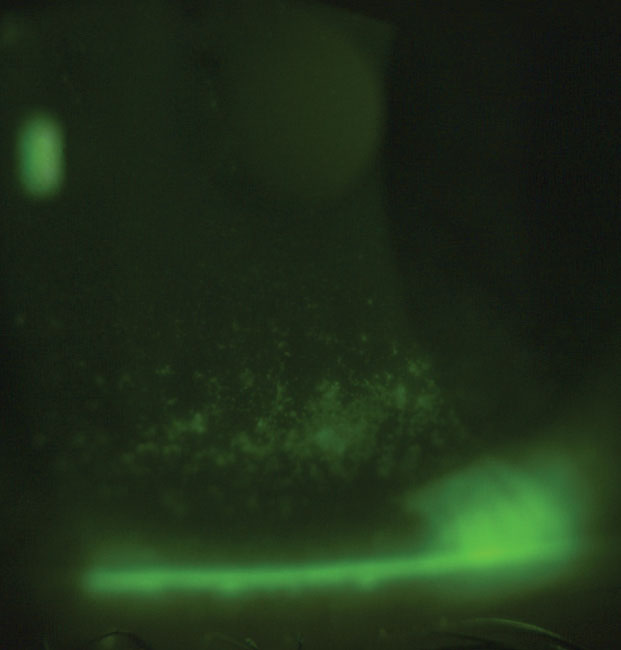
| Fig. 2. Moderate-to-severe ocular staining is best managed with anti-inflammatories. Artificial tears with lubricants with anti-inflammatory-containing properties such as trehalose could be beneficial in these cases. |
Corticosteroids. Soft topical steroids are valuable for dry eye management, especially in acute flare-ups that necessitate rapid relief. However, the potential side effects of elevated intraocular pressure (IOP) and cataracts limit their use. KPI-121 (0.25% loteprednol etabonate ophthalmic suspension, Kala Pharmaceuticals) could be the first FDA-approved product for the short-term, two-week treatment of DED.
KPI-121 uses a mucus-penetrating particle drug delivery system to enhance penetration of this familiar steroid into ocular tissues.15 Thus far, two Phase III trials show mixed results regarding ocular discomfort. One shows statistically significant improvement in ocular discomfort, while the other does not demonstrate a significant improvement.15 Both studies show a consistent statistically significant reduction in conjunctival hyperemia, with IOP elevations similar to the placebo (Figure 1).15,16 The results of the third Phase III trial investigating the temporary relief of signs and symptoms of DED with KPI-121 are expected some time in the fourth quarter of 2019.15
Thymosin β-4. RGN-259 (Thymosin β-4, RegeneRx Biopharmaceuticals) promotes ocular surface healing by stimulating corneal epithelial cell migration and decreasing inflammatory cytokines.17 In a study investigating the effects of RGN-259 topical treatment on DED in a mouse model, 10 days of treatment with RGN-259 led to the recovery of mucins and goblet cells, improved corneal integrity, increased tear production and reduced inflammation.18 The investigators suggested that RGN-259 was comparable with or even better than Xiidra in improving the functions of the cornea, conjunctiva and lacrimal glands of mice.18 Thus far, Phase II trials show positive results for improving symptoms and signs of DED and demonstrate a strong safety profile with fewer side effects compared with products currently on the market.19
Brimonidine tartrate. More commonly known for its IOP-lowering effect, this also has anti-inflammatory properties.20,21 Ocugen is working toward FDA approval of a twice-daily preservative-free drop of 0.2% brimonidine ophthalmic solution for the treatment of DED called Ocu310. It is a nanoparticle formulation of 0.2% brimonidine designed to increase the drug’s efficacy by prolonging retention time on the ocular surface.22 Data from recent Phase III clinical trials is expected in the second half of 2019.22
Unconventional Targets
With the growing need for dry eye therapeutics, pharmaceutical companies are looking at several novel targets for dry eye relief, many of which are making their way through the FDA approval process:
Mucin secretogogues. Mucins play a vital role in stabilizing the tear film and helping the tear film adhere to the ocular surface. Tavilermide (MIM-D3 ophthalmic solution, Mitogen Pharmaceuticals/Allergan) is a peptidomimetic that stimulates mucin-like production by activating mitogen-activated protein kinase systems involved with mucin production, which can also potentially aid in corneal healing.23 In clinical trials, this topical medication improved symptoms and signs of DED in 28 days and had a favorable safety profile.23 Thus far, the drug seems promising.
Diquafosol ophthalmic solution 3% (Santen) and rebamipide 2% ophthalmic solution (Otsuka) are two other drugs that promote mucin secretion and, although both are available in Asia, further clinical trials in the United States have been terminated because they failed to meet clinical endpoints.24-28
 |
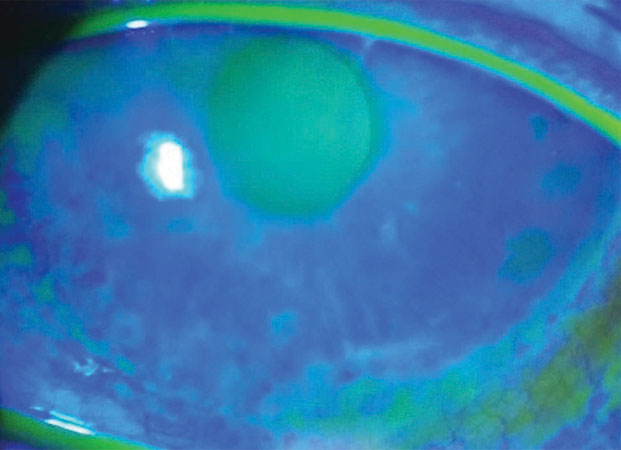
| Fig. 3. Patients with MGD-associated DED may soon have a new therapy option with NovaTears, a 100% perfluorohexyloctane lubricant that is a water- and preservative-free lipophilic liquid. Click image to enlarge. |
Sodium channel blockers. SHP-659 (formerly P-321, Takeda-Shire) is a Phase II investigational topical drug that may address DED by blocking ocular surface epithelium sodium channels. These play a key role in regulating the hydration of the ocular surface by controlling the reabsorption of tears through transport of sodium and water.29,30 As an epithelium sodium channel inhibitor, SHP-659 inhibits the absorption of sodium and water, thereby maintaining ocular surface hydration.30 In animal models, SHP-659 increased tear volume and was well tolerated in a study of dry eye patients.29,30 Future clinical trials should provide more information about its efficacy.
Lacrimal glands. Lacriprep (lacritin, TearSolutions) is another promising topical DED medication curently in clinical trials. It is a fragment of the lacritin protein, which is a glycoprotein released from the lacrimal glands. Research shows reduced levels of this protein are associated with dry eye signs.31-33 Lacritin can promote basal tearing in eyes of rabbits and rescue cultured human corneal epithelial cells from inflammatory cytokine stress in dry eye patients.33 A Phase II clinical trial of patients with Sjögren’s-associated DED is underway.34
Reducing dry eye symptoms and signs may be yet another application for Botox (botulinum toxin type-A, Allergan). While intraglandular injection of Botox is off-label, injection into the lacrimal gland and near the punctum can reduce lacrimal and tear drainage, respectively.35 In a study of intractable DED patients, Botox injection into the medial part of the eyelid improved symptoms and signs and reduced tear cytokine levels.36
Trigeminal nerve. OC-02 (Oyster Point Pharma) is a nasal spray of a nicotinic acetylcholine receptor agonist that stimulates the trigeminal nerve to increase natural tear production.37 In Phase II trials, tear production and symptoms improved significantly.37
The Dry Eye Device BoomWhile the proliferation of dry eye pharmaceutical agents has been sluggish, the dry eye device market has not. Optometrists have several effective instruments to manage their patients. Two recent device additions that have created a buzz are Allergan’s TruTear intranasal tear neurostimulator and Alcon’s iLux. TruTear is a handheld, patient administered intranasal neurostimulating device. The disposable tips are inserted into the nasal cavity to induce the production of natural tears through gentle neurostimulation of the trigeminal nerve, which innervates the goblets cells and both the lacrimal and meibomian glands.1 As a result, the device can potentially produce tears containing aqueous, lipids and mucin. Recent studies demonstrate that the device not only improves DED symptoms but can also cause a significant increase in tear volume with total lipid and protein concentrations equivalent to basal tears.2-4
iLux is also a handheld portable device with a disposable tip. However, this in-office instrument is primarily used to treat MGD-associated DED. A practitioner can treat MGD by using LED light energy to heat the inner and outer eyelids via disposable pads. While applying heat and manually controlled expression, the practitioner can view the eyelid margin with a magnifying lens to tailor the treatment for each individual. iLux treatment has produced clinically and statistically significant improvements in the symptoms and signs of MGD.5 A new comparison study demonstrated that iLux is equal in efficacy and tolerability to Lipiflow (Johnson & Johnson Vision).5 NuLids (NuSight Medical) is a new doctor-prescribed, home-use dry eye device that treats MGD. Analogous to an electric toothbrush, the electric “lid brush” is used for 60 seconds per day to remove collarettes and decap blocked meibomian glands.6 A company-sponsored study found it improved dry eye symptoms and signs, as well as meibomian gland function.6
|
Artificial Tears
Not long ago, DED was solely treated with artificial tears, which provided only temporary relief. Fortunately, we now have drugs that address the disease process of DED. Nonetheless, artificial tears remain an excellent economical first step or a complementary therapeutic. New formulations that offer more than simple tear supplementation may change the playing field for the artificial tear market:
Trehalose. This disaccharide with high water retention capabilities is naturally found in plants and animals and is both a bioprotectant and an osmoprotectant.38,39 It protects corneal cells from the harmful effects of desiccation and tear hyperosmolarity—a hallmark of DED—by fortifying cell membranes and preventing the denaturation of proteins in the absence of water.4,38,39 Research shows trehalose-containing artificial tears improve DED symptoms and signs, including the suppression of inflammation (Figure 2).40,41 Recently, three trehalose-containing artificial tears, Theratears Extra (Akorn), Refresh Optive Mega-3 (Allergan) and Thealoz Duo (Théa), entered the market with the potential to benefit DED patients beyond lubrication.
Perfluorohexyloctane. NovaTears (Novaliq) is a one-component eye drop containing 100% perfluorohexyloctane. This unique lubricant is a water- and preservative-free lipophilic liquid. NovaTears stabilizes the lipid layer and can improve signs of meibomian gland dysfunction (MGD), as well as symptoms and signs of DED (Figure 3).42-44 NovaTears is currently available in Europe and Australia in multidose bottles and is in Phase II clinical trials in the United States.45
As this once unappreciated condition garners more attention, the number of promising candidate therapies that should bring dry eye relief grows. They may one day overcome some of the frustrations clinicians face with today’s management strategies.
Dr. Mickles is an associate professor and the coordinator of the Dry Eye Care Center at Nova Southeastern University College of Optometry. She is a fellow of the American Academy of Optometry and the Scleral Lens Education Society. She lectures and publishes in the areas of ocular surface disease and contact lenses. She is also a principal investigator of dry eye and contact lens research investigations.
Disclosures: Dr. Mickles has received funding from Alcon. She is a speaker and consultant for Alcon and Johnson & Johnson Vision.
|
1. Farrand KF, Fridman M, Stillman IO, Schaumberg DA. Prevalence of diagnosed dry eye disease in the United States among adults aged 18 and older. Am J Ophthalmol. 2017 Oct;182:90-98. 2. Paulsen AJ, Cruickshanks KJ, Fischer ME, et al. Dry eye in the beaver dam offspring study: prevalence, risk factors, and health-related quality of life. Am J Ophthalmol. 2014;157(4):799-806. 3. Asiedu K, Dzasimatu SK, Kyei S. Impact of dry eye on psychosomatic symptoms and quality of life in a healthy youthful clinical sample. Eye Cont Lens. 2018;44(Suppl 2):S404-09. 4. Craig JP, Nelson JD, Azar DT, et al. TFOS DEWS II report executive summary. Ocul Sur. 2017;15(4):802-12. 5. Novack GD, Asbell P, Barabino S, et al. TFOS DEWS II clinical trial design report. Ocul Surf. 2017;15(3):629-49. 6. Mandal A, Gote V, Pal D, et al. Ocular pharmacokinetics of a topical ophthalmic nanomicellar solution of cyclosporine (Cequa) for dry eye disease. Pharm Res. 2019;36(2):36. 7. Wirta DL, Torkildsen GL, Moreira HR, et al. A clinical phase 2 study to assess efficacy, safety and tolerability of CyclASol for treatment of dry eye disease (DED). Ophthalmology. January 28, 2019. [Epub ahead of print]. 8. Sall K, Stevenson OD, Mundorf TK, Reis BL. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. CsA Phase 3 Study Group. Ophthalmology. 2000;107(4):631-9. 9. Holland EJ, Whitley WO, Sall K, et al. Lifitegrast clinical efficacy for treatment of signs and symptoms of dry eye disease across three randomized controlled trials. Curr Med Res Opin. 2016 Jul 22:1-7. 10. Llamas-Moreno JF, Baiza-Durán LM, SaucedoRodríguez LR, Alaníz-De la O JF. Efficacy and safety of chondroitin sulfate/xanthan gum versus polyethylene glycol/propylene glycol/hydroxypropyl guar in patients with dry eye. Clin Ophthalmol 2013;7:995-9 11. Moon JW, Lee H-J, Shin KC, et al. Short-term effects of topical cyclosporine and viscoelastic on the ocular surfaces in patients with dry eye. Korean J Ophthalmol. 2007;21(4):189-94. 12. Leonardi A, Van Setten G, Amrane M, et al. Efficacy and safety of 0.1% cyclosporine A cationic emulsion in the treatment of severe dry eye disease: a multicenter randomized trial. Eur J Ophthalmol. 2016;26(4):287-96. 13. Daull P, Lallemand F, Philips B, et al. Distribution of cyclosporine A in ocular tissues after topical administration of cyclosporine A cationic emulsions to pigmented rabbits. Cornea. 2013;32(3):345-54. 14. Hoy SM. Ciclosporin ophthalmic emulsion 0.1%: a review in severe dry eye disease. Drugs. 2017;77(17):1909-16. 15. Kala Pharmaceuticals. Kala submits NDA for topical dry eye treatment KPI-121 0.25% [press release]. Healio. October 16, 2018. 16. Kala Pharmaceuticals. Focus on Eye Care. http://kalarx.com/technology/focus-on-eye-care. Accessed February 7, 2019. 17. Sonse G, Ousler GW. Thymosin beta 4 ophthalmic solution for dry eye: a randomized, placebo-controlled, Phase II clinical trial conducted using the controlled adverse environment (CAE) model. Clin Ophthalmol. 2015;9:877-84. 18. Kim CE, Kleinman, HK, Sosne G, et al. RGN-259 (thymosin β4) improves clinically important dry eye efficacies in comparison with prescription drugs in a dry eye model. Scientific Reports. 2018 July;8(10500). 19. RegeneRx. RGN-259. www.regenerx.com/RGN-259. Accessed February 8, 2019. 20. Bertino B, Blanchet-Réthoré S, Thibaut de Ménonville S, et al. Brimonidine displays anti-inflammatory properties in the skin through the modulation of the vascular barrier function. Exp Dermatol. 2018;27(12):1378-87. 21. Piwnica D, Rosignoli C, de Ménonville ST, et al. Vasoconstriction and anti-inflammatory properties of the selective α-adrenergic receptor agonist brimonidine. J Dermatol Sci. 2014;75(1):49-54. 22. Ocugen initiates phase 3 clinical trial of OCU310 for dry eye disease [press release]. Ocugen. October 1, 2018. 23. Meerovitch K, Torkildsen G, Lonsdale J, et al. Safety and efficacy of MIM-D3 ophthalmic solutions in a randomized, placebo-controlled Phase 2 clinical trial in patients with dry eye. Clin Ophthalmol. 2013;7:1275-85. 24. Keating GM. Diquafosol ophthalmic solution 3%: a review of its use in dry eye. Drugs. 2015;75(8):911-22. 25. Shrivastava S, Patkar P, Ramakrishnan R, et al. Efficacy of rebamipide 2% ophthalmic solution in the treatment of dry eyes. Oman J Ophthalmol. 2018;11(3):207-12. 26. Townsend W. Not for our eyes: global ocular medications not available in America. Review of Cornea & Contact Lenses. 2011;148(6). 27. Novartis. US Securities & Exchange Commission 2017 form 20-F. www.novartis.com/sites/www.novartis.com/files/novartis-20-f-2017.pdf. Accessed February 9, 2019. 28. Chao W, Belmonte C, Benitez Del Castillo JM, et al. Report of the inaugural meeting of the TFOS i(2) ¼ initiating innovation series: targeting the unmet need for dry eye treatment. Ocul Surf. 2016;14(2):264e316. 29. Boyer J, Johnson MR, Ansede J, et al. P-321, a novel long-acting epithelial sodium channel (ENaC) blocker for the treatment of dry eye disease. Invest Ophthalmol Vis Sci. 2013 June;54:957. 30. National Center for Advancing Translational Sciences. Development of P-321 for chronic dry eye. October 26, 2018. https://ncats.nih.gov/bridgs/projects/complete/p-321-chronic-dry-eye. Accessed February 9, 2019. 31. Aluru SV, Agarwal S, Srinivasan B, et al. Lacrimal proline rich 4 (LPRR4) protein in the tear fluid is a potential biomarker of dry eye syndrome. PLoS One. 2012;7:e51979. 32. Nichols JJ, Green-Church KB. Mass spectrometry-based proteomic analyses in contact lens-related dry eye. Cornea. 2009;28:1109-17. 33. McNamara NA, Ge S, Lee SM, et al. Reduced levels of tear lacritin are associated with corneal neuropathy in patients with the ocular component of Sjögren’s Syndrome. Invest Ophthalmol Vis Sci. 2016;57(13):5237-43. 34. Lacripep in subjects with dry eye associated with primary Sjögren’s Syndrome. Clinicaltrials.gov. NCT03226444. 35. Alsuhaibani AH, Eid SA. Botulinum toxin injection and tear production. Curr Opin Ophthalmol. 2018;29(5):428-33. 36. Choi MG, Yeo JH, Kang JW, et al. Effects of botulinum toxin type A on the treatment of dry eye disease and tear cytokines. Graefes Arch Clin Exp Ophthalmol. 2019;257(2):331-38. 37. Novel nasal spray therapy for dry eye shows promise [press release]. Oyster Point Pharma. July 17, 2018. 38. Fariselli C, Giannaccare G, Fresina M, Versura P. Trehalose/hyaluronate eyedrop effects on ocular surface inflammatory markers and mucin expression in dry eye patients. Clin Ophthalmol. 2018;12:1293-1300. 39. McDonald MB, Fumuso PW. Trehalose: A novel treatment for dry eye. Ocular Surgery News US Edition. July 10, 2018. 40. Pinto-Bonilla JC, del Olmo-Jimeno A, Llovet-Osuna F, HernándezGalilea E. A randomized crossover study comparing trehalose/hyaluronate eyedrops and standard treatment: patient satisfaction in the treatment of dry eye syndrome. Ther Clin Risk Manag. 2015;11(11):595-603. 41. Fondi K, Wozniak PA, Schmidl D, et al. Effect of hyaluronic acid/trehalose in two different formulations on signs and symptoms in patients with moderate to severe dry eye disease. J Ophthalmol. 2018;2018:4691417. 42. Steven P, Augustin A, Geerling G, et al. Semifluorinated alkane eye drops for treatment of dry eye disease due to meibomian gland disease. J Ocul Pharmacol Ther. 2017;33:678-85. 43. Steven P, Scherer D, Krosser S, et al. Semifluorinated alkane eye drops for treatment of dry eye disease—a prospective, multicenter noninterventional study. J Ocul Pharmacol Ther. 2015;31:498-503. 44. Garhöfer G, Schmidl D, Werkmeister RM, et al. Influence of perfluorohexyloctane-containing eye drops on tear film thickness in patients with mild to moderate dry eye disease. Presented at the Association for Research in Vision and Ophthalmology, April 29-May 3, 2018; Honolulu, Hawaii. 45. Novaliq. NovaTears. www.novaliq.com/products/novatears. Accessed February 11, 2019. |