 |  |
Macular holes are a well-known problem for older patients. While their incidence is relatively low, affecting just 0.02% of patients aged 40 or older, full-thickness defects can lead to central vision loss if not treated properly.1 Women are significantly more likely to present with a macular hole, with a 64% increased risk compared with men, and Asian-Americans have a whopping 177% increased risk compared with Caucasians.1
Tractional forces—such as vitreomacular traction (VMT) and adhesion (VMA)—are contributing factors to the formation of an idiopathic macular hole. Although the typical presentation is unilateral, a patient with a macular hole has about a 10% chance of developing a macular hole in the contralateral eye in the presence of VMA/VMT.
As is often the case with non-urgent retinal problems, most optometrists will encounter such patients at some point in their careers and will have to make the call: monitor or refer?
Set the Stage
Careful evaluation with OCT imaging can help you identify at-risk patients and stage the macular hole when present.
A stage 0 to 1 macular hole is described as a partial posterior vitreous detachment (PVD) with insertion of the posterior hyaloid into the fovea and no disruption of outer retinal layers. Partial-thickness (or lamellar) macular hole formation is a lateral or tangential tractional disorder with intact outer retinal layers. Many of these subtle contour changes are asymptomatic but should be observable, and clinicians can initially watch most lamellar holes closely.
Stage 0 to 1 cases that warrant heightened attention include those seen with Amsler grid changes, subjective vision changes and (often hyper-) autofluorescent changes at the foveal base, disruption to the contiguous line of photoreceptor segments overlying the retinal pigment epithelium and progression.
Stage 2 to 4 macular holes are described as full-thickness and typically require surgical intervention. Unlike lamellar macular holes, full-thickness macular hole formation is an anterior-to-posterior tractional disorder.
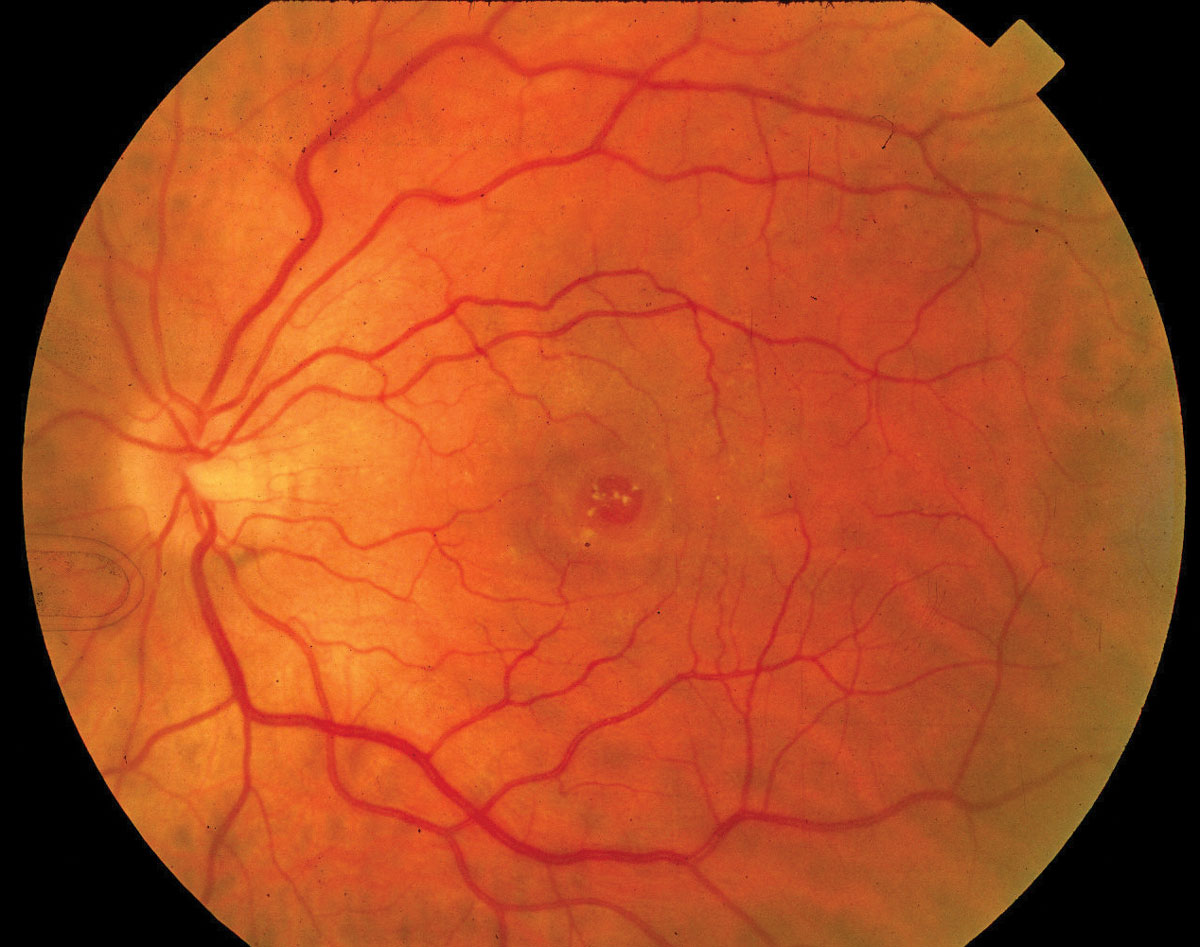 |
| Fig. 1. This patient’s three-month history of vision loss was due to a full-thickness macular hole. Click image to enlarge. |
Finding Closure
Most patients—about 90%—with later-stage macular holes can achieve closure with pars plana vitrectomy (PPV) and internal limiting membrane (ILM) peeling; still, 10% experience a failed procedure and remain at risk for vision loss.2
The goals of macular hole surgery include closure of the hole, improving visual function and minimizing future visual deterioration. It is imperative to refer and consider surgical invention promptly, considering delays in surgical repair once vision loss has occurred lead to sub-optimal visual improvement.
Since macula hole formation is caused by abnormal adhesion to the foveal margins associated with impending PVD, the most critical step of surgical repair is complete PVD to remove all anterior-to-posterior traction at the site of the macular hole.
Much like repairing a bone fracture, whereby a cast or splint stabilizes the reset break, a gas bubble added at the end of surgery helps close the macular hole via two mechanisms: (1) surface tension (encouraging the edges of the macular hole to re-approximate) and (2) dehydrational force (assisting in maintaining hole closure).
Although most patients are advised to keep a facedown position for a week, many macular holes close within one to two days post-op, negating the need for facedown head positioning for a full a week. This is assuming the gas fill of the vitreous cavity is significant enough to contact the macular hole at that time.
By being as efficient as possible and limiting frequent re-entries into the eye via the vitrectomy ports, the surgeon can preserve the anterior hyaloid, thereby limiting the contact of gas with the posterior lens capsule. In Dr. Girschek’s experience, an extremely small percentage of patients undergoing fluid-gas exchange (and not being restricted to facedown head positioning) require cataract surgery during the first three months post-op. Additional facedown positioning is not required for full closure of the hole, in his estimation. However, clinicians should still advise patients to avoid a supine head position to prevent a buoyant gas bubble from shallowing the anterior chamber and potentially causing acute angle-closure glaucoma.
Macular hole surgery has an extremely high success rate, and the sooner surgical repair is performed after macular hole formation, the better the visual outcome. Although historically necessary, facedown head positioning may not be required to achieve successful surgical repair, assuming careful attention is paid intraoperatively to complete vitreous removal and fluid drainage. Efficient and expedient surgical repair may also reduce the risk for post-op cataract formation due to gas contact.
Patient in a Better Position
Case by Drs. Girschek and Shechtman
A 62-year-old white female was referred by an optom-etrist for macular hole evaluation in her left eye follow- ing vision loss for the past three months (Figure 1). Her best-corrected visual acuity measured 20/400 OS, and OCT revealed a full-thickness macular hole. Due to the clinical presentation, surgical intervention was recommended with PPV, ILM peel, indocyanine green (ICG) dye and fluid-gas exchange. Facedown positioning was not encouraged. When the patient returned a week later, her clinical exam findings and OCT images showed full closure of the hole. She was asked to return to the clinic in one month for follow up.
Intervene Sooner
Comments by Dr. Haynie
Since its introduction in the 1990s, PPV has been a go-to management option for full-thickness macular holes (stages 3 and 4) in which the posterior hyaloid has separated from the retinal surface.3 An important yet often overlooked step in the surgery is ICG-guided peeling of the ILM. This not only aids in successful closure of the macular hole, but it also reduces potential failures.
We follow a management regimen in our practice similar to that of Drs. Girschek and Shechtman, with one key difference. While their group does not require facedown positioning for all patients, the surgeons in my group continue to comply with the recommended facedown positioning for 72 hours post-op.
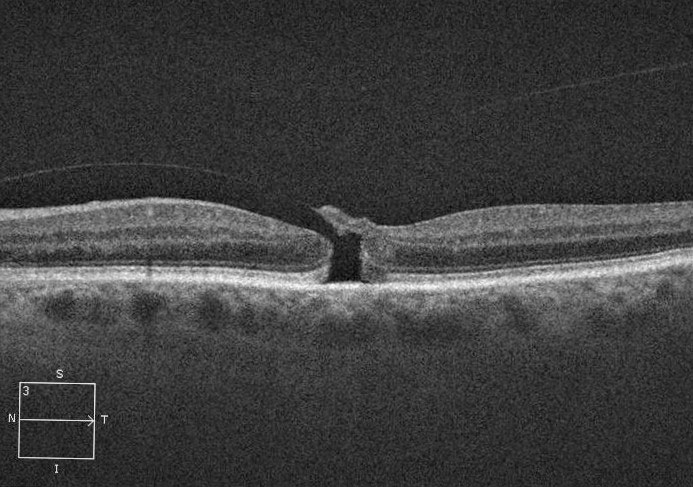 |
| Fig. 2. This stage 2 macular hole has an attached posterior hyaloid. Click image to enlarge. |
The case presented by Drs. Girschek and Shechtman highlights the classic clinical appearance and OCT images of a stage 4 macular hole pre- and post-surgical repair—something we are all familiar with. Less known is the management strategy necessary for a stage 2, or impending, macular hole, defined as a full-thickness defect whereby the posterior hyaloid is still attached (Figure 2). Here’s how we handle these cases in my practice:
A 66-year-old Caucasian female was referred for an evaluation with a complaint of blurred vision, a central scotoma and metamorphopsia for two months. Her best-corrected visual acuity measured 20/40-2 OD, and she missed letters during her acuity assessment. She was diagnosed with an impending macular hole (Figure 3). In the case of a stage 2 macular hole, the current treatment options can include close observation, intravitreal use of Jetrea (ocriplasmin, Thrombogenics), pneumatic vitreolysis or more traditional PPV with fluid-gas exchange.
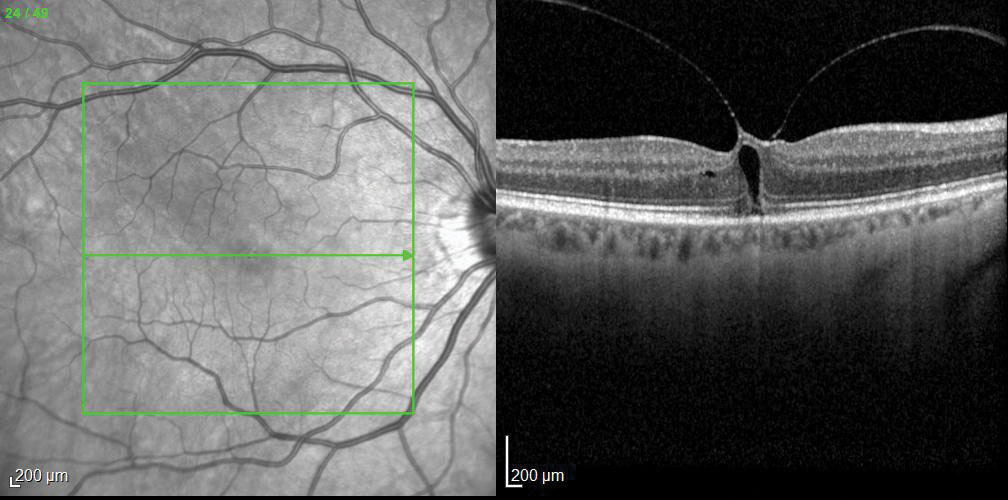 |
| Fig. 3. OCT imaging revealed an impending macular hole as the cause of this patient’s visual complaints. Click image to enlarge. |
Jetrea was approved by the FDA in 2012 for the treatment of symptomatic vitreomacular adhesion. The objective is to induce a PVD, and the medication has a reported success rate for all macular hole types of approximately 27%.4 Success with Jetrea for stage 2 macular holes specifically is approximately 40%.4
However, after Jetrea became more popular among retina specialists, an increasing number of complications were reported, including retinal detachment and permanent loss of central vision or scotoma development. Thus, its use has diminished, and our practice no longer offers this as a treatment option.
Our patient elected to have a pneumatic vitreolysis. Thirteen days post-treatment, OCT imaging confirmed release of the posterior hyaloid with macular hole closure, except for a small subretinal cleft—a finding that typically resolves over time (Figure 4).
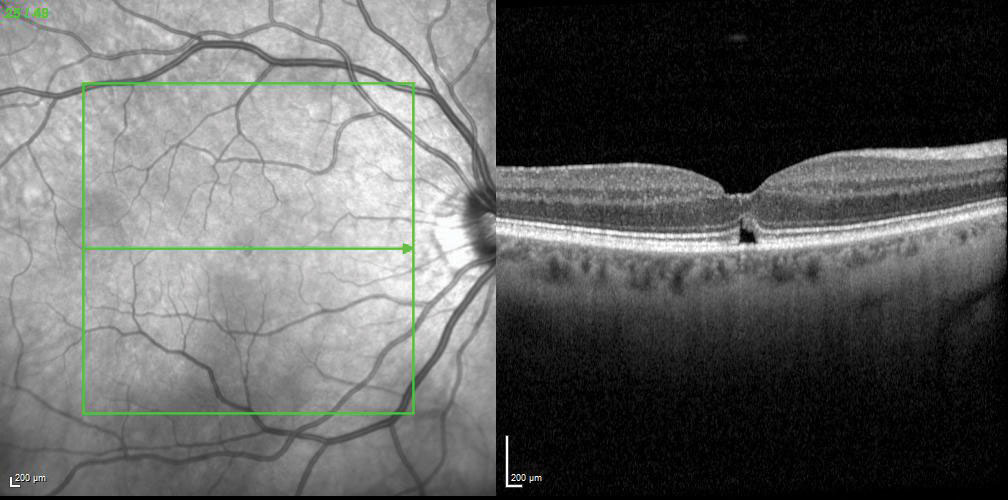 |
| Fig. 4. Pneumatic vitreolysis helped release the posterior hyaloid and close the macular hole. Click image to enlarge. |
Pneumatic vitreolysis is an in-office outpatient procedure. The surgeon injects C3F8 or SF6 gas into the vitreous cavity, followed by a therapeutic anterior chamber parancentesis. The patient is instructed to comply with facedown positioning for five days. This procedure comes with several advantages, including: its outpatient nature, lower cost compared with PPV and a reported success rate of 60% to 100%.5
Clinicians should consider pneumatic vitreolysis for selective cases with a stage 2 impending macular hole.
Dr. Girschek is a retina surgeon at Retina Macula Specialists of Miami.
| 1. Ali FS, Stein JD, Blachley TS, et al. Incidence of and risk factors for developing idiopathic macular hole among a diverse group of patients throughout the United States. JAMA Ophthalmol. 2017;135(4):299-305. 2. Mohammed OA, Pai A. New surgical technique for management of recurrent macular hole. Middle East Afr J Ophthalmol. 2017;24(1):61-63. 3. Kelly NE, Wendel RT. Vitreous surgery for idiopathic macular holes. Results of a pilot study Arch Ophthalmol. 1991;109:654-9. 4. Madi HA, Masri I, Steel DH. Optimal management of idiopathic macular holes. Clin Ophthalmol. 2016;10:97-116. 5. Chan CK, Mein CE, Crosson JN. Pneumatic vitreolysis for management of symptomatic focal vitreomacular traction. J Ophthalmic Vis Res. 2017;12(4):419-23. |

