 |
A 70-year-old Caucasian male presented with an acute onset of diplopia, which he noted for the past two weeks. He described the diplopia as constant, vertical and worse when looking to the left. His medical history was remarkable for hypertension, high cholesterol, GERD and a history of prostate cancer, which was in remission for a number of years. Other than the new onset of double vision, he felt to be in very good heath with good control of his vascular issues and no other systemic symptoms.
Evaluation
On examination, his best-corrected visual acuity was 20/25 in the right and left eyes. His extraocular muscle movements appeared full with no restriction. There was no afferent pupillary defect noted. His intraocular pressures were normal at 12mm Hg OD and 14mm Hg OS. Cover testing showed a right hypertropia worse on left gaze and right head tilt. His retinal exam was remarkable only for two small choroidal nevi, which had been noted on prior exams and were stable.
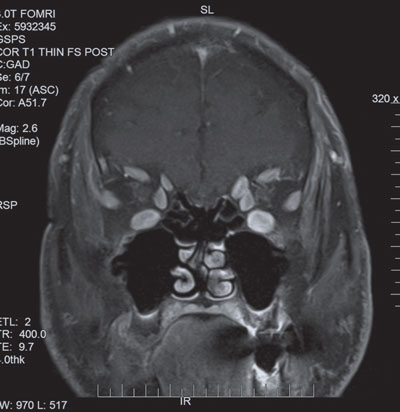 |
| MRI of the orbits showed significant enlargement of the extraocular muscles. |
The pattern of his cover test was consistent with a right fourth nerve palsy. Given his age, the most likely cause of a fourth nerve palsy is microvascular; however, due to his prior history of prostate cancer, an MRI of his brain was obtained, which was normal. Although the MRI was of his brain, his orbits and extraocular muscles were also well visualized and looked normal. Over the next two months, his diplopia continued to worsen, and his right hypertropia worsened significantly.
Although the cover test findings were consistent with a fourth nerve palsy, the progression was not typical for a microvascular nerve palsy. Subsequent testing included brain MRA, myasthenia labs, ESR and CRP, all of which were normal. A neuro-ophthalmology second opinion agreed with the fourth nerve palsy diagnosis, but with no definitive etiology. The neuro-ophthalmologist was considering seronegative myasthenia gravis mimicking a fourth. As the patient’s presentation didn’t seem consistent with myasthenia, a brain MRI was reordered, as well as an orbital MRI, given his continued progression of the hypertropia.
The Path to Orbital Radiation
The follow-up MRI of his brain was still normal, but the orbit MRI now showed significant enlargement of his extraocular muscles with sparring of the tendinous insertions. These findings were highly suspicious for thyroid associated orbitopathy, which was micmicking a fourth nerve palsy, and he was sent for thyroid labs, which showed an extremely low TSH and elevated TSI, thyroglobulin and thyroperoxidase.
Endocrinology diagnosed him with Grave’s disease and started the patient on prednisone and methimazole. His thyroid levels began to normalize; however, in spite of being on oral prednisone, his hypertropia and diplopia continued to worsen. He now showed moderate muscle restriction.
His orbitopathy was unresponsive to oral prednisone, so he was referred to oculoplastiscs for further management. The patient was treated with intraorbital steroid injections that didn’t have any effect so it was recommended that he have orbital radiation. He then received a total of 20Gy distributed over a two-week period, at which point seemed to stop the progression of his hypertropia and restriction.
It wasn’t until approximately one year after his diagnosis that his findings had stabilized, and he underwent muscle surgery to correct the residual ocular deviation. Fortunately, the surgery had completely resolved his double vision, and he has done well since.
Discussion
Thyroid associated orbitopathy (TAO) is an immune-mediated inflammatory disorder that produces expansion of the extraocular muscles and fat within the orbit.1 Generally, you’ll see one of two types of TOA presentations. The first is fat-centric thyroid eye disease, where patients develop fat expansion, eyelid retraction and proptosis.2 This occurs in two-thirds of patients with TAO and typically progresses slowly and occurs in a younger, female population.2 The second type is muscle-centric thyroid eye disease, which was seen in our patient and causes enlargement of the extraocular muscles.2 This can lead to restricted ocular motilities and compression of the optic nerve at the orbital apex, which occurs in one-third of patients with TAO, typically progresses rapidly and occurs in a more balanced gender distribution.2
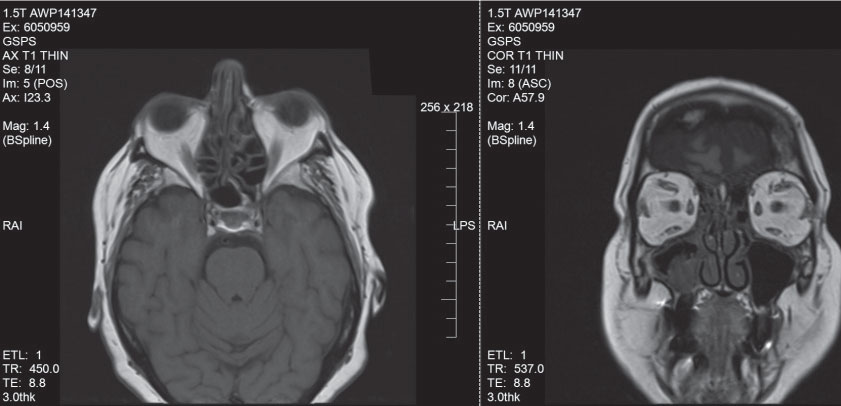 |
| Patient with fat-centric TAO. Note the fat expansion and proptosis. |
We will focus on management of muscle-centric TAO. This presentation will often follow an active phase that generally lasts anywhere from six to 18 months, followed by an inactive phase.2 During this active phase, treatment is initiated to help minimize the ocular complications of the inflammatory cascade. The major complications of muscle-centric TAO are diplopia as well as the possibility of vision loss due to optic nerve compression. There are various treatments one can initiate; however, depending on the severity, treatment doesn’t always reverse the orbitopathy but helps to achieve stabilization until it reaches an inactive phase, as seen in our patient.
Treatment
Corticosteroids are usually the first-line treatment; however, there is no general consensus on the optimum dose, interval, route of administration or duration of treatment.3 Oral, IV and local/intraorbital steroids are all used for treatment of TAO.3 Additionally, steroid-sparing agents, such as cyclosporine and rituximab, are being used either alone or in conjunction with corticosteroids.3
In patients who are either intolerant of steroid therapy, have reached a cumulative dose of more than eight grams of steroids or are inadequately controlled by steroid therapy, orbital radiation is another option.4 Generally, a total of 20Gy in 10 fractions over two weeks is recommended.3-5 Lastly, in patients who develop optic nerve compression, orbital decompression is used as an urgent intervention.
The goal of orbital decompression is expand the orbital volume to decrease the pressure within the orbit. The orbital expansion will relieve pressure on the optic nerve and restore function in cases of compressive optic neuropathy. Orbital decompression in the setting of compressive optic neuropathy must concentrate on bony removal of the orbital apex, where the optic nerve is most susceptible to compression.3,6
Except in the case of compressive optic neuropathy, surgical intervention for strabismus or eyelid retraction should be delayed until the inflammatory component of the orbitopathy has resolved and been stable for at least six months.
Our patient was challenging for various reasons, including his elusive presentation and difficulty in diagnosis TAO as well as his poor response to treatment. TAO can mimic other ocular motility disorders, so the clinician should be aware and consider this in atypical presentations. As mentioned previously, more severe cases of TAO can be unresponsive to certain treatments, and the goal at times is to try and stabilize the orbitopathy until it reaches an inactive phase.
Because diplopia from TAO can significantly impact a patient’s quality of life, all possible treatments should be considered to minimize this, as well as to help prevent further complications of compressive optic neuropathy. If stable and there is still double vision and muscle restriction present, patients can elect to undergo muscle surgery to help correct the deviation. Recurrences of TAO are not common (around 10%), yet patients should be continuously monitored for changes.2
|
1. Barrio-Barrio J, Sabater AL, Bonet-Farriol E. Graves’ ophthalmopathy: VISA versus EUGOGO classification, assessment and managment. J Ophthalmol. 2015;2015:249125 2. Dolman P. Grading severity and activity in thyroid eye disease. Ophthal Plast Reconstr Surg. 2018;34(4S):S34-40. 3. Kumari R, Chandra Saha, B. Advances in the management of thyroid eye disease: An Overview. Int Ophthalmol. 2018:38(5):2247-55. 4. Farach A, Carpenter L. Radiation therapy for orbitopathy of thyroid eye disease. Int Ophthalmol Clin. 2018;56(1):81-93. 5. Dolman, P, Rath, S. Orbital radiotherapy for thyroid eye disease. Curr Opin Ophthalmol. 2012;23(5):427-32 6. Fichter N, Guthoff RF, Schittkowski MP. Orbital decompression in thyroid eye disease. ISRN Ophthalmology. November 12, 2012. www.ncbi.nlm.nih.gov/pmc/articles/PMC3914264/. Accessed December 20, 2018. |

