 In 2003, a 63-year-old white female presented to the office as a new patient for a comprehensive evaluation. She complained of dry eye syndrome, which had been diagnosed previously, worse in her left eye than her right.
In 2003, a 63-year-old white female presented to the office as a new patient for a comprehensive evaluation. She complained of dry eye syndrome, which had been diagnosed previously, worse in her left eye than her right.
Best-corrected visual acuity was 20/20 O.U. through moderately strong hyperopic astigmatic prescription. Pupils were equal, round and reactive to light, with no afferent pupillary defect.
Slit lamp examination of her anterior segments supported the diagnosis of dry eye syndrome. The examination also revealed a decreased tear break-up time, the existence of a fine, diffuse superficial punctate keratitis (SPK) over the inferior half of each cornea, positive lissamine green staining in the same areas, and a scant tear prism. All findings were more pronounced in the left eye than the right. Guttatae were found centrally in both eyes.
Her internal examination was completely unremarkable bilaterally, showing 0.50 x 0.50 cup-to-disc ratios, healthy, plush, and well perfused neuroretinal rims, and normal macular and peripheral retinal evaluations. The patient was taking Benadryl (diphenydramine, McNeil) for seasonal allergies. I explained that Benadryl could worsen her dry eye, and asked her to begin using artificial tears to alleviate her symptoms.
 Diagnostic Data
Diagnostic Data
The patient did not present again until April 2007, when she complained of significant discomfort in her left eye upon awakening that day. She described the pain as significant (5 on a 0-to-10 pain scale), and characterized by foreign body sensation and photophobia.
Her current medications included Refresh Liquigel (Allergan), Crestor (rosuvastatin, AstraZeneca), Zyrtec (cetirizine HCl, Pfizer), Asmanex (mometasone furoate inhalation powder, Schering-Plough), and fluticasone. The patient reported severe allergies to sulfa drugs, oxycontin and hydrocodone.
Best-corrected visual acuity was 20/30-1 O.D. and 20/30-2 O.S. Pupils were normally reactive and symmetric. Extraocular motilities were full in all positions of gaze. Refraction was deferred, as the visit was acute.
Slit lamp examination of her anterior segments was remarkable for moderate corneal endothelial cell loss centrally O.U. There was a moderate amount of fine punctate staining centrally O.U., which was more concentrated in the left eye. Both anterior chambers were clear and quiet. Van Herrick angle estimation was grade 1 O.U., consistent with her moderately hyperopic status. Intraocular pressures were 16mm Hg O.D. and 15mm Hg O.S. Central corneal thickness was 613m O.D. and 632m O.S. The limbal episclera O.S. was slightly injected compared to the O.D.
I diagnosed Fuchs endothelial dystrophy in both eyes as well as chronic dry eye syndrome, which was worse in the left. Narrow angles were also noted. I prescribed FML Forte (fluorometholone 0.25%, Allergan) q.i.d. for the first five days and b.i.d. for the next five days. The patient was asked to return in seven to 10 days for further evaluation.
 When the patient returned for follow-up, she reported that the FML Forte provided almost instantaneous relief, and she said that she complied with the prescribed tapering schedule. On follow-up anterior segment evaluation, her corneas were not staining, and the episcleral injection O.S. had cleared.
When the patient returned for follow-up, she reported that the FML Forte provided almost instantaneous relief, and she said that she complied with the prescribed tapering schedule. On follow-up anterior segment evaluation, her corneas were not staining, and the episcleral injection O.S. had cleared.
The patient also reported that the application of Refresh Liquigel q.i.d. was sufficient to keep her dry eye symptoms under control.
However, over the past two years, she noted that her left eye would flare up intermittently and become painful. The discomfort could last anywhere from several hours to 36 hours. The pain intensity varied, and would clear with increased use of the tear supplement.
The patient denied any increased frequency of this pain in darker environments. Although her clinical signs were more noticeable on the left side compared to the right, her symptomatology seemed to be out of proportion to the clinical picture of dry eye syndrome. Therefore, I ordered a full glaucoma evaluation, which was performed on the same day as the follow-up examination. Her IOP was 16mm Hg O.U. Her central corneal thicknesses were 624m O.D. and 616m O.S.
Gonioscopic examination of her anterior chamber angles demonstrated grade 1 angles O.D. and slit-like angles O.S. The anterior chambers were quiet. Threshold visual fields were normal O.U., and reliability indices were high.
I dilated the patient with two drops of 2.5% phenylephrine O.U. Post-dilation, IOP was 19mm Hg O.D. and 18mm Hg O.S.
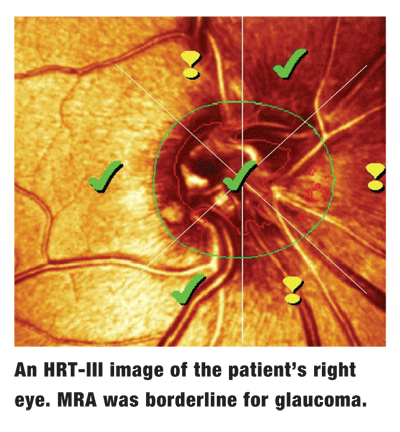
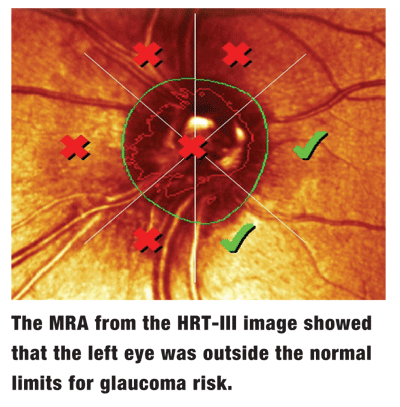
Stereoscopic evaluation of her neuroretinal rims demonstrated 0.55 X 0.55 cup-to-disc ratios in both eyes, and both neuroretinal rims adhered to the ISNT rule. Her macular, vascular and peripheral retinal examinations were normal. Baseline HRT-III scanning of both optic nerves was performed, which flagged asymmetric neuroretinal rim characteristics in the left eye.
This patient was diagnosed with subacute angle-closure glaucoma. She soon underwent bilateral laser peripheral iridotomies.
Discussion
I did not make the diagnosis of subacute angle-closure glaucoma based on the asymmetric right and left eye HRT-III findings. In fact, during her glaucoma work-up, the only significant abnormal finding was the HRT-III scans and, in hindsight, narrow anterior chamber angles. Rather, I diagnosed subacute angle-closure glaucoma based primarily on her history of recurrent, variable and longstanding pain in her left eye, along with clinical findings of narrowed but open angles. The dry eye syndrome complicated the clinical picture insofar as complaints of left-sided pain and discomfort, somewhat mimicking the complaints related to the sub- acute angle closure glaucoma.
While its management can be difficult and varied, acute angle-closure glaucoma is a relatively straightforward diagnosis. Acute angle-closure glaucoma occurs when the anterior chamber angle closes (or at least a significant portion of it does), causing a sudden increase in IOP. When the entire angle closes, IOP may rise to 45mm Hg or higher and cause serious painenough to induce debilitating headaches and nausea. Also, the cornea can become edematous, obscuring views of the anterior chamber and posteriorly beyond.
While the initial management of acute angle-closure is medical, subsequent management is typically surgical. Surgical options include peripheral iridectomy, lensectomy, laser peripheral iridotomy, and filtration surgery (if more than 50% of the angle is closed permanently by peripheral anterior synechiae).1
Subacute angle-closure is more difficult to diagnose and is often overlooked. Because both the anterior chamber angle in subacute angle-closure glaucoma only partially closes and the subsequent IOP rise is not as severe, the associated symptomatology can vary in presentation. The pain and blurred vision associated with subacute attacks may often be attributed to other issues, such as dry eye in this patients case. Subacute angle-closure glaucoma may be precipitated by physiological mydriasis (e.g., entering a dark movie theater) or anterior chamber shallowing (e.g., when tilting the head downward).
At this patients visits, she was not actively having an angle-closure attack. The diagnosis was presumptive, based upon her history and clinical findings of narrowed angles. Provocative tests, such as the dark room and/or prone tests, can be helpful for such patients. Though, in this case, such tests were unnecessary.
Because of both the low complication rate associated with YAG peripheral iridotomies and the increasing likelihood of a complete angle closure, I believed this patient was best served by bilateral iridotomies, aimed at treating the subacute angle closure glaucoma O.S. and to prevent a similar occurrence in the O.D.
Unless we see a patient with an acutely closed angle, we generally think of glaucoma as a symptomless disease. Remember, however, that patients with subacute angle-closure glaucoma do exist, and their symptoms are often attributed to other conditions or merely overlooked. By performing a careful physical examination and thorough patient history evaluation, this uncommon disease can be identified and treated.
1. Kanski JJ. Clinical Ophthalmology: A Systematic Approach. 5th ed.

