 A 63-year-old Hispanic female presented with a chief complaint of decreased visual acuity in her left eye that had persisted for nearly a year. She began having trouble reading and noted that the vision in her left eye was blurry.
A 63-year-old Hispanic female presented with a chief complaint of decreased visual acuity in her left eye that had persisted for nearly a year. She began having trouble reading and noted that the vision in her left eye was blurry.
She reported that her acuity had been much worse when she first noticed the problem, but slowly improved to the current level. Her medical history was significant for hypertension, for which she was properly medicated.
Her best-corrected visual acuity measured 20/20 OD and 20/70 OS. Confrontation fields were full to careful finger counting OU. The pupils were equally round and reactive to light, without evidence of afferent defect. The anterior segment examination was un-remarkable OU.
Dilated fundus exam showed small cups with good rim coloration and perfusion OU. The macula, vessels and peripheral retina in the right eye were normal (figure 1). However, we documented retinal changes in the left eye (figure 2). We also obtained a spectral domain optical coherence tomography (SD-OCT) scan of the left eye (figure 3).
Take the Retina Quiz
1. Based on the clinical appearance, what is your diagnosis?
a. Macular telangiectasia.
b. Retinal vein occlusion (RVO).
c. Valsalva retinopathy.
d. Ocular ischemic syndrome.
2. What does the SD-OCT scan reveal?
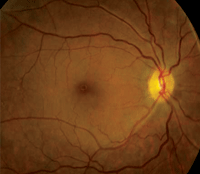
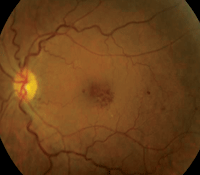
1, 2. Fundus images of our patient (OD left, OS right). Note the retinal hemorrhages located around the left macula.
a. Normal macula.
b. Vitreomacular traction (VMT) syndrome.
c. Serous retinal detachment.
d. Cystoid macular edema (CME).
3. What additional testing might provide the most useful information?
a. Visual fields.
b. Fluorescein angiogram.
c. Blood pressure.
d. Carotid artery evaluation.
4. How would you manage this patient?
a. Laser photocoagulation.
b. Intravitreal anti-VEGF injection.
c. Observation.
d. Endarterectomy.
For answers, see below.
Discussion
We detected focal areas of intraretinal hemorrhages located around the macula of her left eye, which seem to be more localized inferotemporally. On clinical exam, we noted the presence of CME and a large, centralized cyst in her fovea. This finding was confirmed on the SD-OCT scan.
So, why does she have CME in her left eye? It’s not blatantly obvious, but there are several clues. The caliber of the retinal vessels may be one important indicator. In the right eye, she has normal retinal vessels, with an artery-to-vein (A/V) ratio of about three-to-four (3/4). In the left eye, however, the veins appear more dilated and tortuous, with an A/V ratio more like 3/5. This difference suggests that the CME is vascular in origin.
Two additional clues include the presence of a large blot hemorrhage located temporally in the left eye, and the evidence of subtle microvascular changes seen on clinical examination.
Given this information, we concluded that our patient likely had a nonischemic RVO in her left eye. It’s difficult to determine if this was a branch or a central retinal vein occlusion (CRVO); however, considering the dilated and tortuous retinal veins, we assumed that this was a largely resolved CRVO. Nonetheless, the associated intraretinal hemorrhages and increased vascular permeability around the macula catalyzed the development of CME.
So, how should we manage this patient? Until the last five to 10 years, laser photocoagulation was considered the standard of care for macular edema associated with RVO.1 While laser treatment improves macular edema on fluorescein angiography, significant visual improvement is rarely achieved.1 Fortunately, more effective treatments have emerged.
In the early 2000s, intravitreal Kenalog (triamcinolone, Bristol-Myers Squibb) became a popular and successful treatment for CME associated with RVO.2 Despite its efficacy, many clinicians became concerned with the side effects of Kenalog injection, including IOP spikes and cataract development.
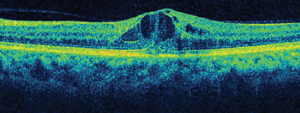
3. SD-OCT image through the macula of her left eye.
More recently, anti-VEGF agents emerged as a good—if not better—treatment option, without the side effects of steroids. In June 2010, Lucentis (ranibizumab, Genentech/Roche) became the first agent to receive FDA approval for the treatment of macular edema secondary to RVO.
Then, in September 2012, the latest anti-VEGF agent, Eylea (aflibercept, Regeneron Pharmaceuticals), was approved for the treatment of macular edema associated with CRVO (
see “Eylea Snares Second Approval,” December 2012).
Additionally, most retinal specialists prefer to use off-label intravitreal Avastin (bevacizumab, Genentech/Roche), because of its significantly lower price tag.
In several clinical trials, Lucentis, Eylea and Avastin have each shown good results in the treatment of macular edema secondary to RVO due to their unique ability to decrease capillary permeability.3-8
We sent our patient to a retina specialist for an opinion on the most appropriate treatment option. At that visit, the patient received an intravitreal Avastin injection and was asked to return for follow-up in one month.
At the one-month follow-up, she demonstrated visual improvement. But, because complete CME resolution was not achieved, she received a second Avastin injection. We will continue to monitor her for signs of improvement.
1. The Central Vein Occlusion Study Group M report. Evaluation of grid pattern photocoagulation for macular edema in central vein occlusion. Ophthalmology. 1995 Oct;102(10):1425-33.
2. Park CH, Jaffe GJ, Fekrat S. Intravitreal triamcinolone acetonide in eyes with cystoid macular edema associated with central retinal vein occlusion. Am J Ophthalmol. 2003 Sep;136(3):419-25.
3. Varma R, Bressler NM, Suner I, et al. Improved vision-related function after ranibizumab for macular edema after retinal vein occlusion : Results from the BRAVO and CRUISE trials. Ophthalmology. 2012 Oct;119(10):2108-18.
4. Campochiaro PA. Safety and efficacy of intravitreal ranibizumab (Lucentis) in patients with macular edema secondary to branch retinal vein occlusion: The BRAVO Study. Paper presented at: Retina Congress 2009. October 4, 2009; New York.
5. Brown DM. Safety and efficacy of intravitreal ranibizumab (Lucentis) in patients with macular edema secondary to central retinal vein occlusion: The CRUISE Study. Paper presented at: Retina Congress 2009. October 4, 2009; New York.
6. Boyer D, Heier J, Brown DM. Vascular endothelial growth factor Trap-Eye for macular edema secondary to central retinal vein occlusion: six-month results of the phase 3 COPERNICUS study. Ophthalmology. 2012 May;119(5):1024-32.
7. A randomized, double-masked, sham-controlled Phase 3 study of the efficacy, safety and tolerability of repeated intravitreal administration of VEGF Trap-Eye in subjects with macular edema secondary to central retinal vein occlusion (CRVO). Clinical trial identifier: NCT01012973. Available at:
http://clinicaltrials.gov/ct2/show/NCT01012973?term=edema+trap-eye&rank=7. Accessed January 16, 2013.
8. Epstein DL, Algvere PV, von Wendt G, et al. Bevacizumab for macular edema in central retinal vein occlusion: a prospective, randomized, double-masked clinical study. Ophthalmology. 2012 Jun;119(6):1184-9.
Answers
1. b
2. d
3. b
4. b

