A 61-year-old black female glaucoma patient, previously managed with beta-blockers, requires medical intervention for airway obstructive disease.

The patient initially presented to me in 1997 at age 54 with a diagnosis of open-angle glaucoma O.U., for which she was medicated with Betoptic S (betaxolol, Alcon) 1 drop qhs O.U. She had been on this regimen for about five years prior.
She also was on both Calan (verapamil, Pfizer) qd and Lasix (furosemide, Aventis) qd to control hypertension. She reported no allergies to medications. Family history was positive for open-angle glaucoma (father and maternal aunt), hypertension (parents), and diabetes mellitus (siblings).
Diagnostic Data
Best-corrected visual acuity was 20/20 O.D. and 20/25-2 O.S. Pupils were equal, round and reactive to light and accommodation with no afferent papillary defect. A slit lamp examination of her anterior segments was unremarkable.
Applanation tensions at 10 a.m. were 17mm Hg O.D. and 15mm Hg O.S.
Gonioscopic examination of the anterior chamber angles demonstrated grade 3 open angles, with a moderate amount of trabecular pigmentation inferiorly O.U.
Her crystalline lenses were clear O.U. Cup-to-disc ratios were 0.60 x 0.70 O.D. and 0.65 x 0.75 O.S. The neuroretinal rims showed inferior thinning. Threshold visual fields demonstrated a superior arcuate depression O.U., consistent with her optic nerve appearance and with her previous field studies. The macular evaluation in the right eye was unremarkable, while that of the left showed fine retinal pigment epithelium granulation and scattered drusen, consistent with the slightly reduced visual acuity.
Management
From her previous records and from my preliminary evaluation, this patients glaucoma was rather stable. Her initial pretreatment IOP, noted in her records, was 24mm Hg O.U. I instituted only one change: that she take the Betoptic-S in the morning rather than in the evening.
Her visual fields have remained stable during her entire seven-year history with me. But by mid-2001, her IOPs had gradually crept up to an average 19mm Hg O.D. and 21mm Hg O.S. I added Travatan (travoprost, Alcon) 1 drop qhs O.U., and the patients IOPs have averaged 13mm Hg O.D. and 14mm Hg O.S. Visual fields and neuroretinal rims remain stable. Pachymetry noted CCTs of 524m O.D. and 527m O.S.
In January 2004, I received a note from her primary-care physician asking to discontinue the Betoptic-S due to the development of obstructive airway disease. He instituted Proventil (albuterol, Schering-Plough) bid to manage the obstructive disease, but suggested she should avoid all beta-blockers.
After a washout period and stabilization of IOPs on Travatan qhs O.U. alone, I added Alphagan P (brimonidine, Allergan) bid O.U.
I continue to manage her with this combination therapy.
Discussion
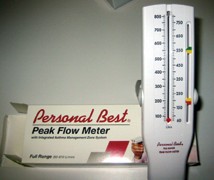
This low-cost device assesses pulmonary changes related to beta-blockers.
While more of us turn to prostaglandins as first-line therapy, many providers still consider topical beta-blockers the first choice in medical management of glaucoma. Beta-blockers can also provide a significant synergistic effect as combination therapy with prostaglandins.
But beta-blockers are not without risk. They increase the risk of asthma and obstructive airway disease, decrease libido, increase incidence of vivid dreams, increase the likelihood of depression and decrease blood pressure.1
Topical selective beta-blockers, such as Betoptic-S, have significantly fewer respiratory side effects and have been used more freely for patients with respiratory concerns. But a recent report noted significant respiratory side effects in glaucoma patients medicated with selective beta-blockers.2 This study determined that there was no significant difference in the risk of airway obstruction between non-selective and selective beta-blockers.
Does this mean we should abandon topical beta-blockers for glaucoma patients? Absolutely not. But we should always take a thorough medical history in our patients before prescribing them.
Also, we can employ pulmonary function tests (PFTs) to help determine the safety of topical beta-blockers in our patients. We can use PFTs to gather baseline respiratory information on a patient before administering any topical beta-blocker, and can then repeat them over subsequent visits to assess any decrease in pulmonary function attributable to the beta-blockers.
A commercially available peak flow meter is inexpensive, and can readily assess both forced expiratory volume (FEV) and forced vital capacity (FVC). FEV is the volume of air forced from the respiratory system in one second on forced expiration. FVC is the entire volume of air expressed from the respiratory system using maximum force in a minimum amount of time. It measures values in liters per minute.
In general, a decrease in FEV indicates airway obstruction (such as bronchiolar constriction), whereas a decrease in FVC indicates restrictive disease (such as interstitial diseases). Furthermore, the ratio of FEV to FVC is usually 0.80, and is lower in patients experiencing airway obstruction. Do not institute therapy below 0.80 or if the patient reports asthmatic tendencies in general. Discontinue therapy if this ratio falls below 0.80 or if the patient complains of any respiratory distress even if the value is above 0.80.
The symptoms associated with beta blockade appear gradually and increase over time; many patients may never notice symptoms. So for your patients on topical beta-blockers, gather preliminary PFT data on them now so that you can make a comparison over time. Also, for patients for whom you are considering topical beta-blockers, measure pre-treatment PFT values now to help you assess subtle respiratory side effects later.
1. Kirwan JF, Nightingale JA, Bunce C, Wormald R. Beta blockers for glaucoma and excess risk of airways obstruction: population based cohort study. BMJ 2002 Dec 14;325(7377):1396-7.
2. Kirwan JF, Nightingale JA, Bunce C, Wormald R. Do selective topical beta antagonists for glaucoma have respiratory side effects? Br J Ophthalmol 2004 Feb;88(2):196-8.
3. Berg DD. Handbook of Primary Care Medicine. 2nd ed. Philadelphia: Lippincott Williams & Wilkins, 1998.

