 |
|
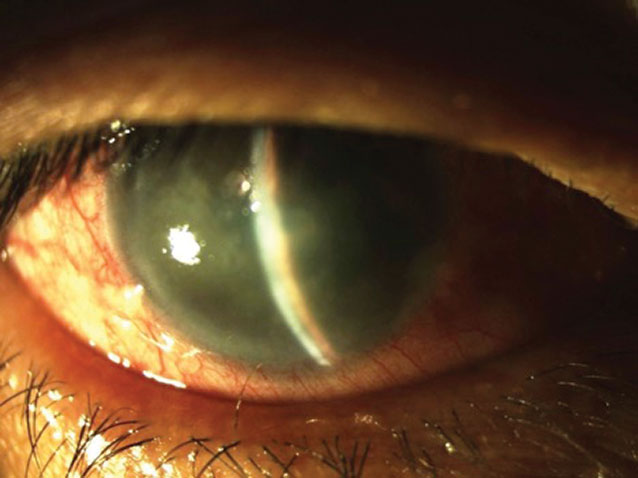
Several new drugs are suspected to have an association with angle closure in light of recent research. Photo: Joseph W. Sowka, OD. Click image to enlarge. |
A large new body of research has recently analyzed instances of acute angle-closure (AAC) glaucoma in tandem with medication usage. The retrospective study was conducted in Korea. Patients diagnosed and treated for AAC between 2013 and 2019 were identified, then all drugs prescribed from the period of one to 180 days before onset were extracted. One to 30 days before instance onset was considered the hazard period while 91 to 180 days prior was deemed a control period.1
The JAMA Ophthalmology study found 61 of the 949 analyzed drugs to be associated with AAC, from a population of 13,531 patients. The medications that displayed the highest odds of development after use included sumatriptan, with the highest association, followed by topiramate and duloxetine. Also included were new findings of drugs not associated with AAC prior to this study, including lactulose (for constipation) and metoclopramide (for nausea).1
While these are certainly important findings, an invited commentary in the same issue by other authors contends the study in question “highlights some of the challenges in using large sets of routinely collected data.”2 Of these challenges, one presented in the study was the authors’ lax approach to inclusion criteria. The study does not distinguish between, as the commentary points out, primary angle closure and primary angle-closure glaucoma.
Additionally, the commentary includes that, in the study, of the 37% of patients taking at least one of the 61 drugs found associated with AAC, 47% of those patients were taking three or more medications and 9% took six or more medications.1 As such, multiple systemic morbidity in these AAC patients may complicate the interpretations the authors made in the results.
To put the study into perspective, the commentary makes apparent that absolute risk to the general population of AAC patients is very small, at around one in 500 people over one year doubling their risk of AAC development from use of the meds in question.
Because of this low rate, the authors of the commentary disagree with the original authors’ viewpoint that “clinicians should be aware of the increased risk, provide appropriate explanations to the patient, conduct ophthalmic examination, and take precautionary measures when other risk factors for AAC are present.”1 They believe the authors may be inflating the prevalence and oversimplifying the issue, advising against barring these medications because of the important benefits they provide patients.
Instead, they propose that “in the context of low overall risk of drug-induced AAC, we suggest clinicians and patients should not be alarmed about the possible risks of a new medication precipitating AAC. A pragmatic approach would be for the prescribing physician to warn of the very small possibility of AAC, to explain the symptoms, and to instruct the patient to seek urgent ophthalmological care if these occur.”2
Both the original study and the commentary provide information that should be thoroughly considered by practitioners. To incorporate both viewpoints, it is likely good to be aware of the most common medications that were associated with AAC, but for the most part, the risk is probably not pertinent to most patients.
1. Na KI, Park SP. Association of drugs with acute angle closure. JAMA Ophthalmol. September 22, 2022. [Epub ahead of print]. 2. Foster PJ, Luben R, Khawaja AP.Association, risk, and causation—examining the role of systemic medications in the onset of acute angle-closure episodes. JAMA Ophthalmol. September 22, 2022. [Epub ahead of print]. |

