When we think of conditions that require long-term management, diabetes, hypertension and glaucoma often come to mind. Like these chronic conditions, dry eye disease (DED) also requires both ongoing management for lasting positive outcomes and an awareness that success lies in the long-term collaboration of the practitioner and patient. Ultimately, successful DED management necessitates a shift in perspective of the doctor and patient from short-term treatment goals that result in temporary relief to a comprehensive chronic care plan for a more permanent resolution. Armed with the right strategies and approaches, clinicians can give their dry eye patients a lifetime of relief.
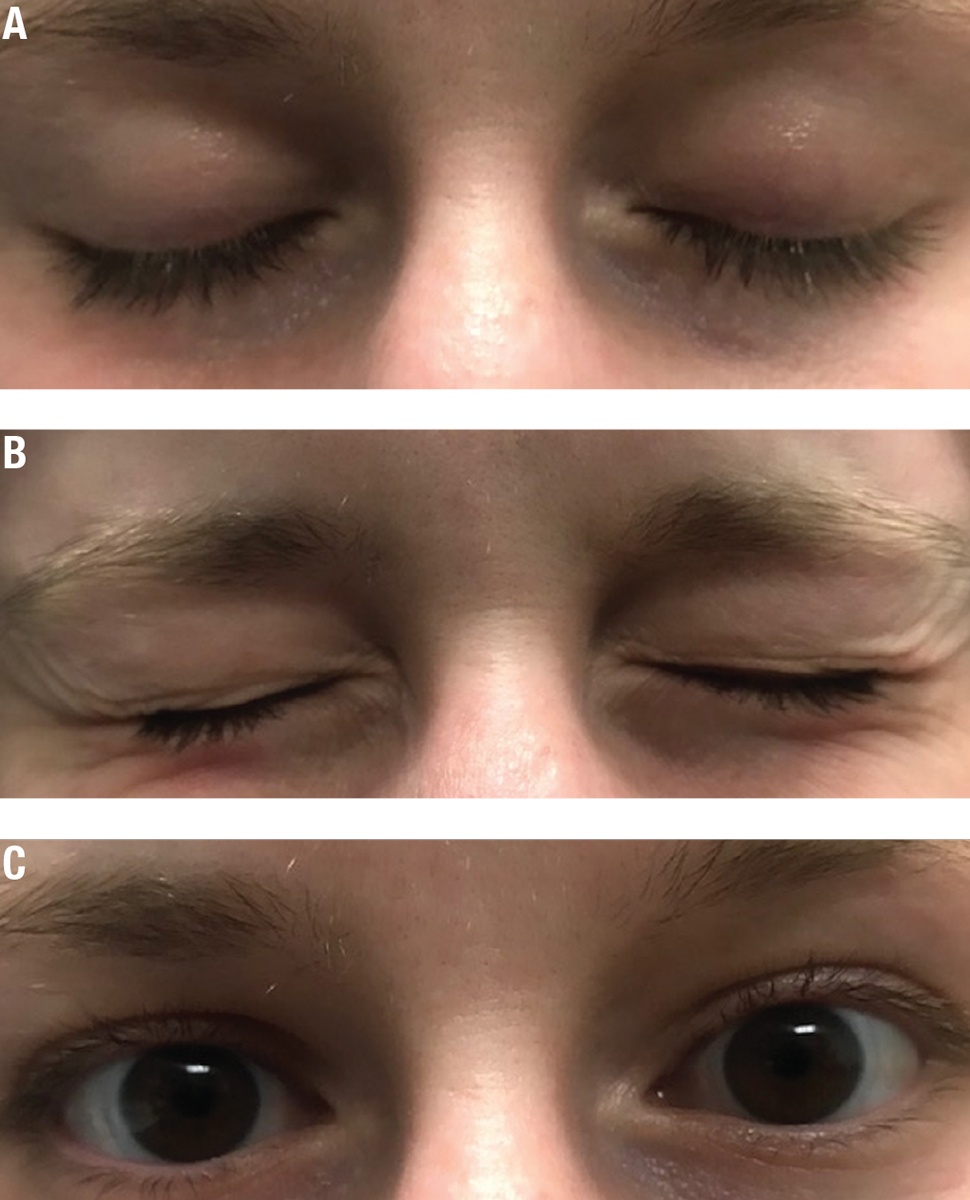  |
| Fig. 1. Blinking exercises will help make proper complete blinking a habit. The blink sequence: (A) Close normally for a count of two and open. Close normally for a count of two again. (B) Squeeze the eyelids together for a count of two. (C) Open to complete the blink sequence. Click images to enlarge. |
Know Your Target
The ultimate aim of DED management is to restore the homeostasis of the ocular surface by offering long-term options that prevent the resurgence of symptoms.1 Achieving this goal goes beyond identifying the root cause of DED, such as aqueous deficiency or meibomian gland dysfunction (MGD), but also involves getting to truly know your patient and the modifiable contributing factors that can lead to life-long wellness. Even with medicinal management, DED symptoms can be exacerbated by lifestyle and environmental factors. When these factors aren’t identified and addressed properly, patients can become discouraged by the belief that the treatment isn’t working. Often, this results in patients discontinuing therapy or departing from the practice. Like an effective weight loss plan, lasting results in DED management often require a permanent lifestyle change.
The Ocular Surface Disease Index (OSDI) questionnaire is an excellent tool that identifies activities and environmental factors that contribute to DED, such as driving, computer use and windy, air-conditioned environments.2 When daily activities and environmental factors known to exacerbate dry eye (DE) are identified as targets, patients can be educated on lifestyle modifications that can have a meaningful impact on DED symptoms in the long-term. Visually demanding tasks, such as digital device use and driving, reduce the blink rate and increase the rate of incomplete blinks, leading to tear film instability.3,4 Proper blinking habits that support a stable tear film can be achieved through daily blinking exercises (Figure 1).5 If patients have difficulty incorporating blink exercises into their daily schedule, free computer applications are available such as the Donald Korb Blink Training App (TearScience) and EyeLeo.5,6 The applications encourage proper blinking with reminders and proper pacing.5,6 As an alternative, patients also can make proper blinking a habit by blinking during activities that they perform frequently such as sending an email. Blinking fully can be an impactful lifestyle change for DE patients, especially during sustained visual tasks.
Other adjustments in daily lifestyle, such as pointing air vents away from the face, make for simple environmental modification that can be easily used by patients long-term if air-conditioned environments are identified as drying for a patient (see “Simple Lifestyle Changes for Long-term Relief").
An assessment of systemic conditions and medications that contribute to DED is important as well. As long as patients continue to use drying systemic agents or have poor control of systemic conditions such as diabetes or autoimmune disease, they may appear to have a limited response to DED pharmaceutical management. A simple change in medication to one that is less drying or education on the importance of good control of a underlying DED-associated systemic condition can have a lasting impact on keeping DED symptoms to a minimum. However, keep in mind that your patient’s family physician, internist or psychiatrist may have labored to find the right medicine. That’s why it’s necessary to have a comprehensive picture of your patient’s lifestyle. By identifying key targets, doctors can prescribe effective medical therapy and lifestyle changes that help prevent the resurgence of symptoms. Once these targets are identified, a customized plan can be created.
Simple Lifestyle Changes For Long-term ReliefDaily activities and environmental factors can contribute to dry eye disease. Here are some lifestyle modifications that can keep dry eye symptoms at bay. • Increasing water consumption. Drinking more water and decreasing caffeine, a diuretic, can help alleviate dry eyes. Sufficient water is vital for overall health, and you should encourage DED patients to drink the recommended eight to 10 glasses of water every day. • Avoid blowing air. Constant air flow is known to dry the eyes. Advise DED patients to not sleep under ceiling fans and to point air vents away from them in the home and car. Wrap-around sunglasses or moisture chamber glasses can benefit DED patients who work in windy environments. • Alter the digital work space. Upgaze position can increase the palpebral fissure size, exposing the ocular surface, consequently destabilize the tear film.1 A stabilized tear film can result when the palpebral fissure is narrowed in downgaze.1 Educate patients to set up their work space so that their eyes are positioned slightly downgaze when viewing digital screens. Raising chairs and lowering computer monitors so that the eyes are positioned downgaze should help minimize computer-related dry eye symptoms. • Stop smoking. Tobacco smoke is known to exacerbate dry eye.2,3 It causes tear film instability and increases ocular surface staining.2 Smokers are nearly twice as likely to have dry eyes.3 Encourage smoking cessation and avoidance of smoke.
|
Develop a Customized Chronic Care Plan
Since DED is multifactorial, the clinician and patient should recognize that one treatment will not completely address this chronic condition and that no single treatment plan works for everyone. A customized chronic care plan that suits the individual patient will be most effective for long-term positive outcomes.
On a day-to-day basis, the patient is responsible for their own care. Thus, chronic care plan development should involve a regimen that addresses the patient’s unique presentation and can be realistically incorporated into their day-to-day lives. A barrier to effective use of medications includes complex regimens, which often incorporate numerous medications.7 The more medications prescribed, the less likely the patient is to comply with the treatment regimen.8,9 For lasting success, the management plan should include a targeted approach that includes only a few effective interventions that the patient will reasonably use long-term, rather than a random selection of a multitude of options in which the least effective management options are employed by the patient.
Thus, simplified DED treatment regiments that target the predominant cause of DED—whether it be evaporative dry eye (EDE) or aqueous tear-deficient (ADDE)—are ideal. In ADDE lacrimal function is reduced with normal evaporation.1 In EDE, we see excessive evaporation in the presence of normal lacrimal function.1 However, ADDE and EDE are not mutually exclusive categories. As DED progresses, eventually the characteristics of both ADDE and EDE will be present.1 In such cases, in addition to recommending a treatment that is specifically indicated for the predominant subtype, it is best to recommend a therapy with a target that is central to multiple aspects of the patient’s DED presentation. Since inflammation is a commonality in both EDE and ADDE, an anti-inflammatory therapy such as Xiidra (lifitegrast ophthalmic solution 5%, Shire Pharmaceuticals) or Restasis (topical cyclosporine 0.05%, Allergan) can be a complementary option that minimizes the need for numerous interventions.
Tear hyperosmolarity is a hallmark of DED that also occurs in both forms of the disease.1 While artificial tears are typically viewed as solely palliative, when artificial tears are needed, a hypo-osmolar lubricant, such as TheraTears (Akorn Pharmaceuticals) is a good choice, especially if multiple therapies are a concern either due to cost or adherence.
Unfortunately, long-term management of DED can be complex, and it does not lend itself to one precise algorithm that works for everyone.1 Nonetheless, DED can be successfully managed for a lifetime with a minimum treatment regimen that produces results. Simplified, customized, chronic care plans that target the cause of DED and are tailored to the patient’s lifestyle should maximize patient adherence and optimize patient outcomes.
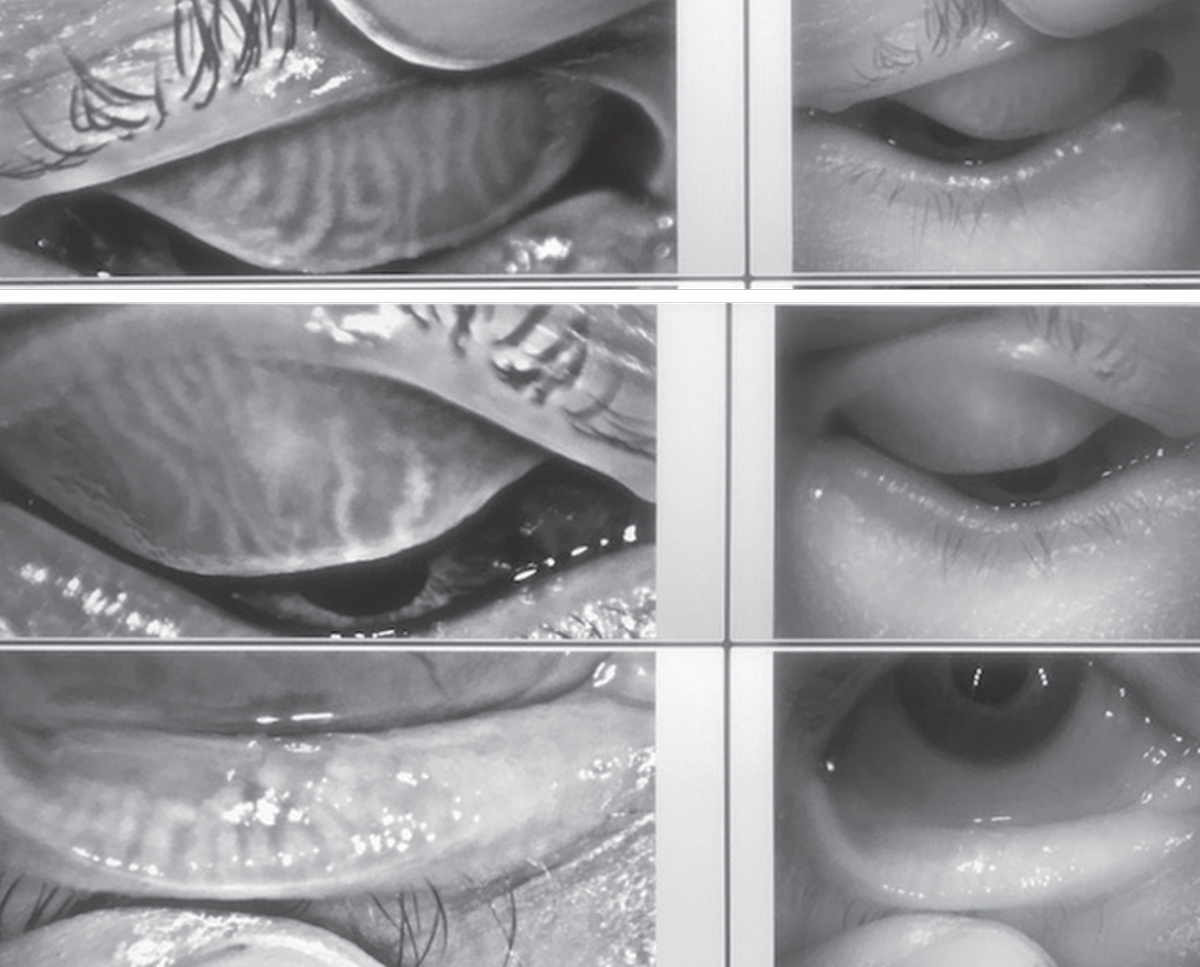 |
| Fig. 2. Advanced imaging, such as meibography, can help foster compliance. Click images to enlarge. |
As Time Goes By
Whether it is contact lens use, diabetes medications or glaucoma eye drops, getting patients to correctly and consistently adhere to treatment recommendations is an obstacle faced by most health care providers. DED therapy is no different. Understandably, as time goes on, DED patients can end up inconsistently or improperly follow treatment plans. Nonetheless, treatment adherence is an essential component of lasting DED management success. Eye care professionals are positioned to partner with patients to implement key compliance strategies that should help DED patients overcome adherence barriers and keep the condition well managed for the long haul.
Long-lasting compliance is likely not going to come from simply telling a patient to follow a treatment plan; rather, you must identify the obstacle that prevent patients from complying. Since the cause of poor adherence can be multifaceted and individual-specific, identifying such barriers is the first step in success. Beginning the discussion by asking what is challenging for them can have a more meaningful impact than recapping a list of tasks they should implement. A simple question such as, “What is preventing you from using the drops?” will uncover specific barriers to adherence that you can focus on to incite change.
One barrier to adherence might be that the patient doesn’t clearly understand the chronic nature of DED and the necessity of taking a specific mediation for an extended period of time. We may think that we explained DED and its treatment well, but inevitably there are patients who truly don’t understand the condition and consequently discontinue treatment. Explaining the chronic nature of condition and why ongoing therapy is essential in clear and simple language can increase the likelihood of patient adherence. Patients may cease using an important treatment by assuming that the costly medication isn’t necessary or expecting it to be effective instantly. I’ve found that explaining how a particular treatment such as Restasis addresses a central cause of their dry eye symptoms and that continued use is needed to maintain relief goes a long way.
Some patients may not understand the seriousness of their condition and, as a result, do not place importance on the treatment plan. Advanced imaging technology such as meibography can impact the way patients understand the severity of their DED and encourage them to adhere to a treatment regimen. Patients are frequently more interested in how the disease is specifically affecting them than a general academic assessment of the condition. The easy-to-understand reports and imaging (Figure 2) allows patients to clearly visualize the effects of DED on their eyes and the importance of proper treatment compliance.
 |
| Fig. 3. Most DED pharmaceutical and eyelid hygiene therapies are BID. Streamlining the dosing regimen to BID should improve adherence. |
Complex medication dosing regimens are a significant barrier to patient adherence.10,11 If complexity is a barrier for the patient, simplify the patient’s treatment regimen by minimizing the number of treatment options and streamlining the dosing regimen. Since many DED treatments are BID, prescribing all therapies as BID if possible is convenient and easy for the patient (Figure 3). The BID regimen can be easily incorporated into their daily morning and evening hygiene routine.
Other frequently cited reasons for nonadherence are cost and side effects.12,13 Clinicians can contribute to noncompliance by not discussing the necessity of the medication. Even if a patient can afford more costly medications, they may not fill the prescription if they don’t understand the need for the medication or the impact of not taking it.
Research shows Restasis (Allergan) can lead to fewer office visits, less time off work and decreased used of artificial tears.14 Using one costly yet effective medication can sometimes benefit the patient by saving time, reducing overall costs and, ultimately positively impacting quality of life. Taking the time to educate patients about the rationale for using a medication as it relates to quality of life can enhance compliance.
While we should never pre-judge patients’ ability to pay for medications, some clinicians may subvert their own efforts by not attempting to tailor the treatment plan to something affordable for the patient. While prescribing the most effective option is optimal, if cost is a concern, sometimes a similarly efficacious but less costly alternative is available. Pharmaceutical assistance programs offered by manufacturers such as Allergan and Shire and charitable organizations such as the Patient Access Network are also available to help uninsured patients with the cost of prescribed medications.15 BenefitsCheckUp is a user-friendly website that provides links to nearby patient benefit programs.16
Although dry eye medications can have adverse effects that are frequently non-serious and temporary, patients may discontinue a medication due to lack of awareness of potential side effects. Proactively alerting patients of these and providing strategies to minimize them work well to ensure compliance despite them.
Lotemax (topical loteprenol etabonate 0.5%, Bausch + Lomb) pretreatment is an effective strategy to minimize Restasis-associated stinging and subsequent discontinuation, for example. One study shows significantly less Restasis-associated stinging and discontinuation with loteprednol induction therapy.17
Anecdotally, punctal occlusion can reduce Xiidra-associated dysgeusia. Anecdotally, my patients have found that chewing gum after instillation of Xiidra may help reduced dysguesia, but no research has examined this.
Unfortunately, no matter how customized and cutting-edge a dry eye treatment plan is, it won’t work for patients who don’t follow it.18 Thus, clinicians must recognize the importance of patient adherence in long-term DED management success. Patient-centered compliance strategies can ultimately advance the possibility of long-lasting dry eye relief.17
 |
| Fig. 4. Lid margin debridement with a blunt golf spud or similar instrument can prevent or delay MGD-associated DED. |
Face Setbacks Head on
What do you if there is a resurgence of symptoms or a return of significant ocular surface staining? If there is a recurrence of symptoms or signs, increasing the level of pharmaceutical therapy isn’t always the answer. We shouldn’t assume that the current medicines and dosages aren’t sufficient to control the DED. Contributory DED extrinsic or intrinsic factors can arise at any time and become a culprit. When DED symptoms worsen or return, inquire about recent DED altering life changes such as increases in alcohol or caffeine consumption, changes in work or home environment, new diagnoses and medications that have a drying effect. Nonetheless, it is important for the patient and practitioner to realize that despite best efforts, DED is variant and symptoms will wax and wane.
If your treatment plan isn’t working, or results in only mild improvement in symptoms and signs, a formal dry eye assessment, including a detailed case history and a series of evidence based dry eye diagnostic tests, will likely lead to a correct treatment plan. Perhaps the patient may have concomitant disease that mimics dry eye that was overlooked. In this case, the DED component may have been effectively addressed, but symptoms and signs remain as a result of a masquerading condition.19 When only mild improvement in symptoms and signs occurs, practitioners should look for conditions that manifest with similar symptoms or signs to DED. Clinicians should look out for conditions such as mucin fishing syndrome, conjunctivochalasis, Demodex, and asthenopia from binocular vision disorders.19 There are also situations where there are chronic symptoms but limited signs that are refractory to treatment.1 When this occurs, neuropathic pain, rather than DED should be considered.1 Unless these conditions are addressed, the patient will continue to exhibit symptoms and become frustrated irrespective of a proper DED management plan.
When treatment failure occurs, poor compliance frequently comes to mind, yet patients will report that they are following the treatment plan like clockwork. In this scenario, although their intentions are good, patients maybe missing the mark by administering the treatment incorrectly. In a study evaluating eye drop instillation technique with a tear substitute, for over a third of the patients, the artificial tear landed on the eyelid or cheek.20 It is easy to take for granted that patients know how to administer eye drops or use lid scrubs correctly. Using the American Medical Association’s teach-back method is effective in revealing where patients are going wrong.21 In-office and video demonstrations provided by pharmaceutical companies or found online are tremendously useful in subverting treatment failure and getting patients on the right path for treatment success. It is also essential that close follow-up ensues once treatment is initiated to ensure patients are successfully implementing the recommended plan.22
Nonetheless, poor technique and other extrinsic factors may not be the cause of treatment failure. Sometimes patients are just truly not responding to a given level of management and treatment plans will need to be adapted to replace or include an additional therapy. However, immediate adjustments to treatment plans should be avoided. While therapeutic response varies with the individual and therapy, with the exception of cyclosporine, therapeutic effect for most DED treatment options should occur anywhere from one to six months.22 Beyond this time period, for the majority of DED treatments, improvements are unlikely and modifications to the management plan are recommended.22 Awareness and education on expected onset of treatment effect is important in any DED management strategy.
DED treatment setbacks can be avoided when management objectives and the nature of the disease are clearly defined early and reinforced on a regular basis along the way. However, DED setbacks occur even under the best of patient management circumstances. Additionally, patients must recognize that once DED is controlled, achieving and maintaining an asymptomatic or minimally symptomatic state requires continuing effort and, sometimes, adjustments.
Develop a Maintenance Plan
Fortunately, there is light at the end of the tunnel on the DED journey. In most cases, eventually a patient’s DED will be controlled sufficiently for practitioners to reduce the number and frequency of supportive therapies such as artificial tears. However, a treatment maintenance strategy with follow-up at appropriate intervals and consistent care is essential in sustained relief. As with diabetes, patients should understand that regular maintenance and follow care for DED is important for long-term success. Home maintenance especially for MGD associated DED can be achieved with basic eyelid hygiene practices such as lid scrubs and warm compresses with eye masks. Like regular teeth brushing and face washing, these home eyelid hygiene care practices can be easily incorporated into their daily morning and evening hygiene routine and can help maintain control and possibly preventing MGD-associated DED.
Regular in-office maintenance treatments such as manual lid margin debridement are beneficial for MGD associated DED patients as well (Figure 4). Lid margin debridement can effectively remove accumulated debris and skin cells that obstruct the meibomian gland orifices.23 Similar to dental plaque, the material can re-accumulate over time.23 Thus treatments such as lid margin debridement should be performed on a regular basis. “Lid cleanings” at regular intervals can become standard practice for these patients as teeth cleaning.
Lipiflow (TearScience) and Mibo Thermoflo (Mibo Medical Group) are also in-office MGD treatments options. Both devices use heat and massage to unblock clogged meibomian glands. While Lipiflow can be efficacious for up to three years and Mibo Thermflo anecdotally for up nine months, concurrent home maintenance therapies such as warm compresses, lid hygiene and omega-3 supplements is critical for long-term success.24 Patients should recognize that neither of these devices are a cure for MGD and both regular in-office and home maintenance therapies are important. In the future, regular ocular surface maintenance with routine visits for DED patients may become as standard as annual diabetic exams and quarterly visits for glaucoma patients. The long-term efficacy of Lipiflow and Mibo are yet to be made clear in the academic literature.
Unfortunately, no one-size-fits-all approach can successfully solve dry. However, implementing effective long-term management strategies and elevating DED to the level of other chronic diseases in the mindset of the patient can optimize outcomes for a lifetime.
Dr. Mickles is an associate professor and the coordinator of the Dry Eye Care Center at Nova Southeastern University College of Optometry. She is a fellow of the American Academy of Optometry and a principal investigator of dry eye and contact lens research investigations.
1. Craig JP, Nelson JD, Azar DT, et al. TFOS DEWS II Report Executive Summary. Ocul Sur. 2017;15(4):802-12. |

