You’re probably using ophthalmic drugs for off-label uses (those not approved by the FDA) more often than you realize.
In fact, about half of medications prescribed routinely in eye care are likely used off label.1 For example, topical antibiotics are generally approved only for treating bacterial conjunctivitis—but are even more valuable when used off label against bacterial keratitis.
Not only is this permissible, it’s good medicine.
“The FDA acknowledges that physicians may prescribe any legally marketed product for an off-label use, as long as it is in the best interest of the patient,” according to a position statement made by the Alliance of Specialty Medicine.2 “Off-label use is often the standard-of-care in the community. Not using medicines off label could be considered malpractice in many circumstances.”
So, why don’t drug companies pursue these indications “on label”?
There are several reasons, says Louis Catania, OD, an innovator in off-label uses of drugs in eye care. Not the least of these is the added costs to the pharmaceutical company to sponsor clinical trials to pursue additional labeling, and the many years required for the FDA approval process.
Thus, off-label drug usage may not only be effective, but more expeditious. Indeed, in the days before optometrists had therapeutic prescribing privileges, Dr. Catania advocated the use of over-the-counter Polysporin ointment for eyelid and conjunctival infections and inflammation—a legal and legitimate therapy.
These days, better treatments are available and ODs have the license to use them. But the FDA approval process is still slow and narrowly focused, and many treatment needs remain unmet. “Thus, it’s imperative for practitioners to keep up with the drug literature and research, and to recognize legitimate clinical studies, trials, results and potential off-label uses,” Dr. Catania says.
To that end, here are the best off-label applications chosen by some of optometry’s therapeutic experts.
Restasis for Uveitis
“As an immunosuppressive agent, cyclosporine has enormous value in all forms of inflammation—well beyond that caused from dry eye,” Dr. Catania says.
Specifically, it’s a great off-label treatment for uveitis, he suggests.
While Restasis (cyclosporine A 0.05%, Allergan) is known to take weeks (or sometimes months) to be effective in patients with chronic dry eye, that’s not the case in patients with an acute inflammatory condition such as uveitis—it works quickly. “The literature is extensive (from as far back as the 1990s) regarding the efficacy and response time of topical and oral cyclosporine (effects within days) vs. steroids (sometimes weeks) for uveitis,” Dr. Catania says.3
He points to a recent research poster that showed that topical cyclosporine hastened improvement when used adjunctively in a small group of patients with recurrent anterior uveitis. “The patients had statistically significant fewer episodes of anterior uveitis, shorter duration of episodes and fewer total days of inflammation per year while on topical cyclosporine A 0.05%,” the authors reported.4
Barring contraindications, “any immunomodulator that can begin to reduce the extensive use of topical steroids in eye care should be considered by clinical optometric practitioners as both a primary and adjunctive therapy for most external and uveitis inflammation,” Dr. Catania says.
Start Restasis with the recommended BID dosage, Dr. Catania says. “However, dosages may have to be increased based on the response to BID and/or the severity of the condition.”
Advil for Anterior Uveitis
To shorten or eliminate the use of oral or high-dose topical steroids for anterior uveitic inflammation, try oral ibuprofen, Dr. Catania says.
“As an anti-inflammatory, I found its use in dosages as high as 2,400 to 3,000mg/day (given standard contraindications) to be valuable as a substitute for steroids or used as adjunct therapy.”
He adds, “I even found, when used prodromally in certain forms of recurrent uveitis—Fuchs’ being notorious for patients’ prodromal awareness of an acute attack—ibuprofen proved to be effective in reducing the recurrences and/or the intensity of the recurrence.”
Lotemax for Dry Eye
Lotemax gel (loteprednol 0.5%, Bausch + Lomb) is FDA approved for treating inflammation and pain following ocular surgery—but it’s routinely used for a variety of anterior segment inflammatory conditions, such as allergic conjunctivitis, rosacea, superficial punctate keratitis (SPK), herpes zoster keratitis, iritis, cyclitis and others.
Scott Ensor, OD, MS, who teaches systemic pharmacology at Southern College of Optometry in Memphis, Tenn., says there are two main circumstances when he uses Lotemax for dry eye.
“The first is when there are visible signs of inflammation—when the disease has progressed to the point where there is visible hyperemia and/or SPK, and the usual lubricant drops are much less effective. The anti-inflammatory properties of Lotemax help decrease those signs and hopefully give the patient a feeling of relief,” he says.
In such cases, the dosage depends on the severity of the inflammation, but is usually BID to QID, Dr. Ensor says. Because loteprednol is less likely than other steroids to cause a rise in intraocular pressure, a duration of six months or even longer is usually safe (although he monitors patients’ progress during this time). Also, be sure to measure IOP before treatment to compare it to the post-treatment IOP.
The second circumstance is when starting Restasis. “One of the drawbacks to Restasis treatment is the length of time that it sometimes takes for the patient to feel relief. This often leads to discontinuation of the medication and the feeling that it didn’t work,” Dr. Ensor says. “A short course of Lotemax to ‘jump start’ the process can give faster relief and encourage the patient to continue the Restasis.”5
Here’s his regimen: Start with Lotemax BID for two weeks, then use both Restasis and Lotemax BID for another two weeks. At that time, stop the Lotemax and continue with the Restasis alone. “I have much more success with Restasis when I follow this plan than when I use Restasis alone,” Dr. Ensor says.5
In either case, “patient education and follow-up appointments are essential,” he says. “Many patients are used to the simple artificial tears approach to dry eye treatment, and they don’t understand the difference in getting the corticosteroid,” Dr. Ensor says. “They must return to the office for IOP monitoring and proper discontinuation (i.e., tapering) of the medication.”
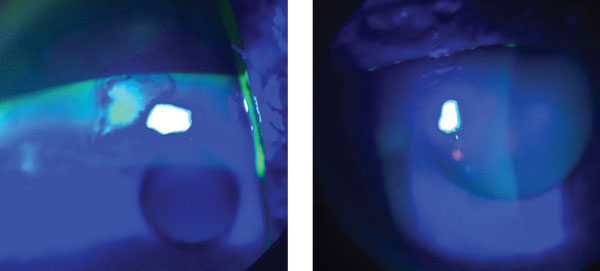
| |
| This patient presented with recurrent corneal erosion (left). After debridement, he began doxycycline 20mg BID for 2.5 months. He’s been symptom-free ever since (right). |
Doxycline for RCE
Riddle: When is an antibiotic not an antibiotic?
Answer: When it’s used in a sub-antimicrobial dose as an anti-inflammatory.
Indeed, when used in low doses (100mg or less), oral tetracyclines offer several anti-inflammatory benefits—not the least of which is downregulating the proinflammatory matrix metalloproteinases (MMPs).
In ocular surface conditions, notably recurrent corneal erosion (RCE), MMPs break down the epithelial adhesion complexes between the epithelium and the basement membrane, explains Jeffrey Varanelli, OD, who practices at Simone Eye Center in suburban Detroit.
“So, if we can decrease these inflammatory enzymes with doxycycline, then there’s the potential for stronger adhesions, less chance of the adhesions breaking down and ideally fewer recurrences of corneal erosions,” he says.6
For ophthalmic purposes, “low-dose” oral doxycycline means 20mg or 25mg. If the price of the 20mg tablets is too steep, prescribe the comparatively less expensive 50mg tablet and tell the patient to cut it in half, Dr. Varanelli says.
He uses a four-pronged combination approach for most patients with RCE. First, he debrides the cornea (by instilling tetracaine and then using a blade or cellulose sponge) to remove the damaged epithelium. Then, he applies an amniotic membrane (or a bandage contact lens) to cover the wound and begin the healing process. About a week later, when the membrane has dissolved, the patient begins low-dose doxycycline BID as well as a “soft” steroid (Lotemax) for two to three months.
“I had one patient who was symptomatic almost every day. He’d wake up and his eyes would be a little scratchy. And when he came into the office, he’d show early signs of recurrent erosion,” says Dr. Varanelli, who treated the patient with the combination approach. “That was back in March 2014, and he’s been symptom-free ever since.”
A Better ‘Get-the-Red-Out’
White eyes look bright and healthy, and that’s why people use “get the red out” drops like Visine (tetrahydrozoline, McNeil), says Marc Bloomenstein, OD, director of optometric services at Schwartz Laser Eye Center in Scottsdale, Ariz.
But as optometrists know (and many patients discover), “redness relievers” like tetrahydrozoline are only a short-term solution because they frequently lead to rebound hyperemia.
Instead, Dr. Bloomenstein provides patients with a dilution of the alpha agonist brimonidine to relieve red eyes. “When I have patients who have chronic red eyes, this drop comes in handy,” he says.
Interestingly, “selective alpha-2 adrenergic receptor agonists, when used at conventional doses of 0.1% or higher, are associated with a number of undesirable side effects, such as rebound hyperemia,” he says. “But the diluted version of this compound does not carry the same effects.”
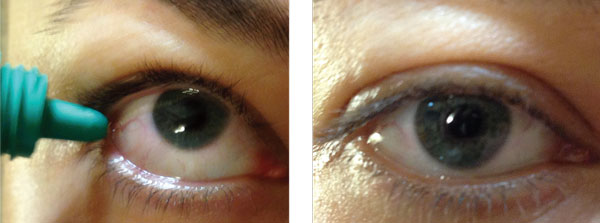
|
|
| Substitute patients’ tetrahydrozoline with a drop of diluted brimonidine to “get the red out.” Diluted brimonidine also improves comfort and won’t cause rebound hyperemia. |
|
How was this discovered? “In the late 1990s, we knew that brimonidine was a good vasoconstrictor, and thus it was applied before refractive surgery to reduce subconjunctival hemorrhage and hyperemia,” Dr. Bloomenstein says.7
More recently, a study of diluted brimonidine instilled before LASIK reduced subconjunctival hemorrhage and injection, and improved patient comfort after surgery.8
For Dr. Bloomenstein’s redness reliever, trial and error led to the dilution that offered the best effect. “I use the formula of two drops of brimonidine to every 1ml of low-viscosity tear solution,” he says. “The drop usually lasts about four to six hours, so I tell patients to use it when they feel they need it for the whitening effect.”
While this diluted compound is generally safe for most patients, the best candidates have no signs or symptoms of ocular pathology, he recommends.
Put a Plug in it
Off-label indications apply not only to drugs, but also to devices such as punctal plugs, says Walt Whitley, OD, MBA, director of optometric services at Virginia Eye Consultants in Norfolk, Va.
Like other medical devices, punctal plugs require FDA clearance before being marketed to treat dry eye disease.
But they have additional uses besides dry eye, Dr. Whitley says.
For instance, he uses temporary, collagen punctal plugs to intensify and prolong the effect of medications, particularly in conditions that require a high therapeutic dose. “In acute cases of recurrent corneal erosion, corneal abrasion or corneal ulcer—in which hourly antibiotic doses of topical fluoroquinolone are needed—I’ll insert a temporary plug in the lower tear duct, which evidently increases the medication’s residence time on the corneal surface,” Dr. Whitley says.
Anecdotally, he says, this seems to speed recovery and enhance compliance.
But it’s not just for acute conditions. In a chronic disease such as glaucoma, where compliance with daily drops is essential, punctal occlusion helps to lower IOP just a bit more.9
The next step, Dr. Whitley predicts, are drug-releasing punctal plugs. Some are already in Phase II clinical trials, including a punctal plug for post-cataract surgery that releases dexamethasone and one for glaucoma that provides a sustained dose of travoprost.10,11
Last but not least, a few words of caution from Dr. Varanelli, who lectures frequently on off-label usage: “Make sure the application is a prudent clinical decision that is based on a firm, scientific rationale and sound medical evidence,” he says. “Also, be sure to let the patient know that what you’re prescribing is an off-label indication, and make sure that you document the drug’s use and effects in the chart.”
1. Ask the Ethicist: Off-label Meds. EyeNet. 2011 May;15(5):55.
2. Alliance of Specialty Medicine. Physician Directed Applications (Off Label Use): A Position Statement of the Alliance of Specialty Medicine. October 14, 2011; Washington DC. Available at: www.specialtydocs.org/weblev/user_upload/Final_Alliance_offlabel_position_statement.pdf. Accessed January 5, 2015.
3. Kaçmaz RO, Kempen JH, Newcomb C, et al. Cyclosporine for ocular inflammatory diseases. Ophthalmology. 2010 Mar;117(3):576-84.
4. Michelotti M, Shtein RM, Prabhu SS, Cooney T. Topical cyclosporine A 0.05% for recurrent anterior uveitis. Poster presented at American Society of Cataract and Refractive Surgery Annual Symposium. April 25, 2014; Boston, MA.
5. Sheppard JD, Donnenfeld ED, Holland EJ, et al. Effect of loteprednol etabonate 0.5% on initiation of dry eye treatment with topical cyclosporine 0.05%. Eye Contact Lens. 2014 Sep;40(5):289-96.
6. Dursun D, Kim MC, Solomon A, Pflugfelder SC. Treatment of recalcitrant recurrent corneal erosions with inhibitors of matrix metalloproteinase-9, doxycycline and corticosteroids. Am J Ophthalmol. 2001 Jul;132(1):8-13.
7. Norden RA. Effect of prophylactic brimonidine on bleeding complications and flap adherence after laser in situ keratomileusis. J Refract Surg. 2002 Jul-Aug;18(4):468-71.
8. Pasquali TA, Aufderheide A, Brinton JP, et al. Dilute brimonidine to improve patient comfort and subconjunctival hemorrhage after LASIK. J Refract Surg. 2013 Jul;29(7):469-75.
9. Aritürk N, Oge I, Erkan D, et al. The effects of nasolacrimal canal blockage on topical medications for glaucoma. Acta Ophthalmol Scand. 1996 Aug;74(4):411-3.
10. Endl MJ, Levenson JH, Walters TR, Majmudar PA. Multi-center evaluation of safety and efficacy of sustained-release dexamethasone after cataract surgery. Paper presented at American Society of Cataract and Refractive Surgery Annual Symposium. April 27, 2014; Boston, MA.
11. ClinicalTrials.gov. Phase 2b study evaluating safety and efficacy of OTX-TP compared to timolol drops in the treatment of subjects with open angle glaucoma or ocular hypertension. Identifier: NCT02312544. Last updated Dec 4, 2014.

