 A 63-year-old Hispanic female presented with blurred vision in her right eye that had persisted for approximately eight months. She reported that the visual distortion progressed over a period of several weeks, which now prohibited her from reading with her right eye.
A 63-year-old Hispanic female presented with blurred vision in her right eye that had persisted for approximately eight months. She reported that the visual distortion progressed over a period of several weeks, which now prohibited her from reading with her right eye.
She denied any history of ocular injury, surgery or infection. Her medical history was remarkable for controlled hypertension.
Best-corrected visual acuity measured 20/200 OD and 20/20 OS. Confrontation visual fields were full to careful finger counting OU. Pupils were equally round and reactive, with no evidence of afferent defect. Ocular motility testing was normal. The anterior segment examinations of both eyes were unremarkable. Intraocular pressure measured 14mm Hg OU.
The optic nerves in each eye were healthy, with good rim coloration and perfusion. Dilated fundus exam revealed a posterior vitreous detachment (PVD) in her right eye. The right macula exhibited obvious changes (figure 1). The vessels and periphery were normal. Additionally, we performed a spectral-domain optical coherence tomography (SD-OCT) scan (figure 2) OD. The left eye, however, was completely unremarkable.
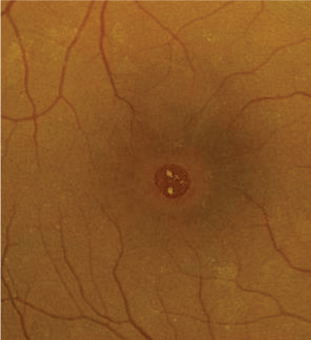 |
| 1. Our patient’s right macula exhibits
a peculiar lesion. |
1. Based on the clinical exam, what is the correct diagnosis?
a. Lamellar macular hole.
b. Cystoid macular edema (CME).
c. Stage III, full-thickness macular hole (FTMH).
d. Stage IV FTMH.
2. What does the SD-OCT scan reveal?
a. Epiretinal membrane with a pseudohole.
b. Lamellar hole with CME.
c. Neurosensory detachment with evident CME.
d. FTMH.
3. How should this patient be managed?
a. Observation.
b. Pars plana vitrectomy (PPV) with internal limiting membrane (ILM) peel, gas bubble injection and face-down positioning.
c. Intravitreal anti-VEGF injection.
d. Laser photocoagulation.
4. Which agent could be used to treat our patient’s condition?
a. Eylea (aflibercept, Regeneron Pharmaceuticals, Inc.).
b. Jetrea (ocriplasmin, ThromboGenics).
c. Avastin (bevacizumab, Genentech/Roche).
d. ACU-4429 (emixustat hydrochloride, Acucela).
5. How would you describe the finding illustrated in figure 3?
a. Normal macular structure.
b. Epiretinal membrane.
c. Foveal “pseudocyst.”
d. Incomplete inner/outer segment junction at the fovea.
For answers, see below.
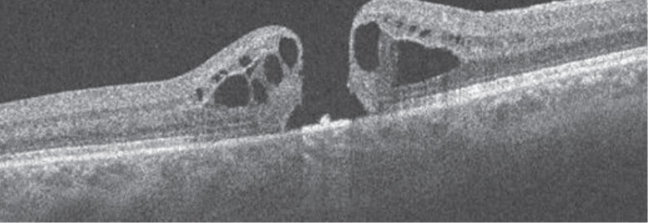 |
| 2. SD-OCT scan of her right macula. What do you notice? |
Our patient has a classic full-thickness macular hole in her right eye. On clinical examination, we documented a round, well-circumscribed, full-thickness retinal defect. Associated yellow deposits could be seen at the base of the hole, and an accompanying cuff of fluid located around the hole was indicative of a serous retinal detachment.
The presence of a PVD was slightly unusual, however. The existence of a PVD categorizes this as a Stage IV presentation, which occurs in approximately 25% of all FTMH cases. It is important to note that the vast majority of FTMHs are Stage III.
J. Donald M. Gass, MD, described the progressive stages of idiopathic macular hole formation based upon biomicroscopic observations.1 He postulated that macular holes developed as a result of cortical vitreous shrinkage, which resulted in tangential traction on the macula.
Indeed, this is still thought to be what occurs in patients with idiopathic macular hole development. However, with the development of OCT, we now have a much better understanding of the pathogenesis and clinical spectrum of macular hole formation:
• In a Stage I macular hole, there is loss of the foveal depression, which is seen as a yellow spot or ring in the center of the fovea. A foveal “pseudocyst” is evident on OCT, which represents a large, optically empty space within the retina. This presentation results from posterior cortical vitreous contraction.
• In a Stage II macular hole, the patient exhibits a full-thickness retinal defect that usually measures less than 400µm in thickness.
• Stage III macular holes are larger, measuring 400µm to 600µm, with a surrounding rim of retinal detachment that ranges between 1,000µm and 1,500µm. In some Stage III holes, there is no evidence of a cortical vitreous detachment. In these instances, the hole is caused by tangential traction. However, in cases where the macular hole manifests from an incomplete PVD, perifoveal vitreous detachment occurs. Here, the vitreous is attached in the center of the fovea but detached around the macula.
• A Stage IV macular hole occurs when there is an obvious PVD. Most of these holes develop as a result of trauma. In fact, it is rare to observe an idiopathic Stage IV macular hole––which is exactly what occurred in our patient.
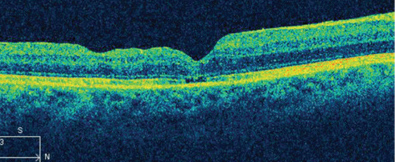 |
|
| 3. Three months after undergoing PPV, our patient experienced complete FTMH
closure. |
|
Jetrea is another potential treatment option. Ocriplasmin is a molecule created from plasminogen. It is a proteolytic enzyme that can induce a PVD by partially dissolving the protein structures contained within the vitreous. This action results in a pharmacologic lysis between the vitreous cortex and the retina. In the pivotal FDA trials of Jetrea, 40.6% of patients exhibited FTMH closure with a single intravitreal injection vs. just 10.6% in the placebo group.2 Unfortunately, the treatment is expensive––priced at nearly $3,000 per injection.
Our patient elected to undergo PPV. Three months after the surgery, her macular hole closed completely and her vision improved to 20/60 OD. However, there was some lingering disruption in the outer retinal layers at the level of the inner/outer segment junction. This likely explains why her vision wasn’t better than 20/60. Nevertheless, over time, we hope that the integrity of her outer retina will improve and yield further visual recovery.
1. Agarwal A. Macular dysfunction caused by vitreous and vitreoretinal interface abnormalities. In: Gass’ Atlas of Macular Diseases, 5th ed. Vol. 1. Philadelphia: Elsevier Saunders; 2012:628-712.
2. Stalmans P, Benz MS, Gandorfer A, et al. Enzymatic vitreolysis with ocriplasmin for vitreomacular traction and macular holes. N Engl J Med. 2012 Aug 16;367(7):606-15.
Answers
1. d
2. d
3. b
4. b
5. d

