Because the term traumatic brain injury (TBI) encompasses a plethora of situations that range from mild to severe, clinicians often look for greater precision in the definitions and categorization of TBI. Mild traumatic brain injury (mTBI) and concussion are somewhat interchangeable terms used to define “a complex pathophysiologic process affecting the brain, induced by traumatic biomechanical forces.”1,2 The difference between mTBI and moderate-to-severe TBI can be determined using the Glasgow coma scale (Table 1). A 2010 study found that 75% of an estimated 1.7 million people treated for brain injuries in the United States had mTBI.3 Recently, head injuries among athletes and military service personnel—the two most common patient populations at risk for TBI—have shone a spotlight on the consequences affecting individuals with mTBI.1
| This demonstration of oculomotor training shows techniques taught to patients dealing with traumatic brain injury-related vision disorders, notably convergence issues. Here, a patient is asked to locate a stimulus. |
This article provides an overview of common visual problems associated with TBI, guidelines for diagnostic assessment and management recommendations. An understanding of potential visual and ophthalmic changes following TBI is essential in allowing the primary care optometrist to properly care for affected patients.
Presentations
Clinically, a TBI patient can have any combination of signs and symptoms that may include, but are not limited to, visible trauma, confusion, headaches, memory loss or muscle function problems. Visual disturbances common in mTBI patients can include photophobia, blurred vision, reading difficulties, eyestrain, diplopia, visual field defects, color vision changes and vestibular dysfunctions. Visual symptoms can impair rehabilitation and create significant restrictions with occupational, educational and other activities of daily living.4-14
TBI-related damage to the afferent pathway may include the optic nerve, optic tract, chiasm, optic radiation or occipital cortex. Damage to any part of this pathway may decrease visual acuity, which may result in reduced visual function and an inability to perform daily activities. In the worst-case scenario, there may be such significant loss that the individual ends up legally blind.
| Table 1. Glasgow Coma Scale | |||
| Behavior | Response | Score | |
| Eye opening response | Spontaneously To speech To pain No response | 4 3 2 1 | |
| Best verbal response | Oriented to time, place and person Confused Inappropriate words Incomprehensible sounds No response | 5 4 3 2 1 | |
| Best motor response | Obeys commands Moves to localized pain Flexion withdrawal from pain Abnormal flexion to pain (also termed decorticate) Abnormal extension to pain (also termed decerebrate) No response | 6 | |
| TBI Classification | Loss of Consciousness | Glasgow Coma Total Score | |
| Mild (mTBI) | Less than 30 minutes | 13 and up | |
| Moderate TBI | Greater than 30 minutes, but less than 6 hours | Between 9 and 12 | |
| Severe TBI | Greater than 6 hours | 8 or less | |
Photophobia
Increased sensitivity to light is a common visual symptom following TBI.4-8 One study found that photophobia was the most common complaint of the TBI population studied.6 Photosensitivity may be present in all types of lighting or it may be more pronounced in specific conditions, such as indoor fluorescent lighting or outdoor sunlight. The underlying cause of photophobia remains somewhat unclear but some evidence points to an alteration in the visual system’s ability to adapt to prolonged light or darkness.4,6 In addition, abnormal critical flicker fusion frequency—the minimum light flicker frequency for an individual to perceive a steady (non-flickering) presentation of light—may be related to discomfort with fluorescent lighting in some patients.4,5,7,8,15
Effective management begins with a thorough history that accounts for the patient’s experience of the specific lighting conditions associated with their symptoms. Be sure to rule out any possible ocular inflammatory processes, such as uveitis, that may develop following trauma. Photophobia is typically addressed with filters, visors and attention to proper illumination. Management may also include changing the room lighting from fluorescent to incandescent or LED lightning indoors, employing a light tint in or outdoors and wearing sunglasses outside.
Blurred and decreased vision may occur following TBI as a result of refractive error, or structural ocular changes or both.4,7,9 Refractive changes may occur with trauma to any refractive ocular structures, including the cornea and crystalline lens, as well as with accommodative dysfunctions, which can follow mTBI. Any ophthalmic examination following TBI must include a meticulous objective and subjective refraction so that any refractive errors can be precisely corrected.
Studies associate TBI trauma with physical changes in various ocular structures that may result in vision blur or distortion.4,9,16 For example, an altered tear film composition may cause dry eye symptoms, including blink-related blur, distortion or foreign body sensation.
 | |
| This oculomotor training technique tests the patient’s ability to track a stimulus, in this case a ball. |
Corneal injury may result in keratitis or scarring, which can lead to loss of vision. The crystalline lens may develop a cataract or become dislocated in response to traumatic insult, causing vision distortion or diplopia. Vitreoretinal changes can include hemorrhages, detachments, floaters, maculopathies or other pathologies, any of which may lead to visual impairment.4,9,15,16
Careful examination of anterior and posterior segment ocular structures, including a dilated exam, is essential to identify and appropriately manage any anomalies associated with mTBI.
Accommodative Dysfunction
Mild TBI, like any damage to the accommodative pathways or the oculomotor nerve, may lead to accommodative dysfunction. Impaired accommodation can cause variable vision blur, visual fatigue, asthenopia and headaches, particularly with tasks at near distances, such as reading or computer work.4,7,9,10
The most common TBI-related accommodative disorder is accommodative insufficiency.10 Reference values for minimum amplitude of accommodation, typically measured using an accommodative target such as a near-point card with the patient wearing their distance correction, are measured by subtracting 15 from one quarter of the patient’s age (0.25[age]-15).
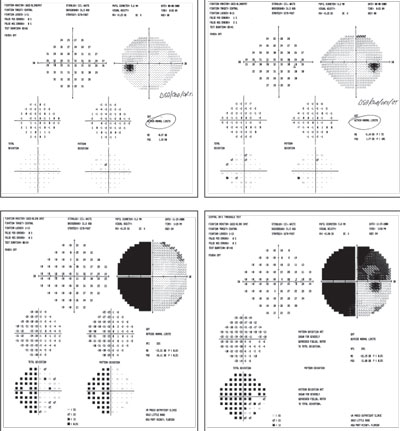 | |
| These fields are from the same patient. The clear fields, on top, were performed prior to those below (as the patient was a glaucoma suspect). Below you can see a visual field defect that was present after a severe TBI event (motor vehicle accident). |
Test each eye monocularly and binocularly. Inconsistent values between eyes may indicate localized pathological changes in one eye, although inaccurate refractive correction in one or both eyes may indicate dysfunction.
Other accommodative disorders may include accommodative infacility and pseudomyopia.10 Accommodative insufficiency may be treated with reading glasses or bifocal/progressive lenses. Both accommodative infacility and insufficiency may be improved with oculomotor rehabilitation.9
Oculomotor Dysfunctions
Both versional and vergence oculomotor dysfunctions can occur in TBI patients. Versions are conjugate eye movements and include pursuits, saccades and fixation.4,7,9,16-18 Versional eye movements should be assessed as part of the oculomotor evaluation. Anomalies of the visual fields or damage to various vision-mediating neurological structures can affect these eye movements.10,16 Dysfunction of versional eye movements may cause symptoms such as significant reading difficulties including reduced reading speed, loss of place when reading and re-reading text.4,7,9,10
The King-Devick (KD) test evaluates saccades through rapid number reading on a series of three testing cards, which become progressively more challenging due to variability in spacing between numbers. The test accurately and reliably identifies impaired saccadic function, which may occur following TBI.19
Vergence oculomotor functions refer to non-conjugate changes in eye position when viewing objects at varying distances.4,7,9,17,16 Vergence disorders may be non-comitant (strabismic but the deviation changes depending on the gaze), and may occur with damage to cranial nerves III, IV or VI, or comitant (strabismic and the deviation remains relatively constant). Symptoms of vergence disorders occur only under binocular viewing conditions and may include eyestrain, double vision and headaches.4,7,9,10,15
| Interventions: Vision Training Many TBI patients respond well to efforts to improve vision, cope with impairment, or both. In two studies, one research team shows that ocular motor rehabilitation has improved accommodative facility and amplitude, as well as vergence responsivity. This may be due to plasticity of the neural system and “oculomotor learning effects.” The improvement in accommodative amplitude “reflects an increase in neuronal firing (through recruitment) and/or better synchronization of the accommodatively based midbrain related neurons,” according to research.23,33,34 A short-term follow-up study shows persistence or delayed improvement of 62% of the clinical oculomotor parameters tested.29 A retrospective analysis concluded that their “current clinical sample exhibited either complete or marked reduction in their oculomotor–based symptoms and associated clinical signs with the maintenance of the symptom reduction and sign improvements at the two month to three month follow up.”26 Investigators noted that it was the combination of motor training with attention training that yielded the best prospect for improved vergence.35 Protocols developed for training to improve binocular vision included visual targets involving motor perceptual, memory and attention tasks. “Training allows a patient to have heightened attention to manifestation of visual deficits such as blurring of an image, corresponding motor response that compensates for the deficit,” say the authors. “With repetition it becomes reflexive.”35 In other words, the more that a patient has the aforementioned areas stimulated, the better the responses become. Oculomotor rehabilitation can take place either in office or at home—for instance, in the office first to customize the regimen to the patient’s specific needs, and then continued at home. Keep in mind that other, non-ocular dysfunctions may affect the outcome of rehabilitation. Many individuals who have experienced TBI may also have concurrent memory or cognitive issues, or both, which may make ocular motor rehabilitation more challenging. A review of studies for “evidence regarding the use of oculomotor based vision assessment to identify and monitor recovery from mild TBI” was able to identify “that limitations include small, inadequately described study populations; lack of clearly reported inclusion.”36 The review also mentioned “inadequate detail regarding study protocols and procedures, lack of detail regarding the diagnosis of mTBI and lack of blinded assessors and limited information on matching control participants.”40 |
The most common vergence dysfunction in TBI patients is a convergence insufficiency, usually occurring as a result of damage to the oculomotor nerve or the medial rectus muscles, causing symptoms to be most evident when reading or performing other near tasks.20-22 Patients with convergence insufficiency characteristically have exophoria at near distances greater than far, a reduced nearpoint of convergence and a reduced positive fusional vergence range.9,10,14 Positive fusional vergence range values that are less than double that of the exophoric posture are likely to be associated with symptomatic convergence insufficiency.9,10,14
Vergence testing is conducted using a phoropter, but you may obtain more reliable measurements using prism bars in free space.
Research shows convergence insufficiency often presents itself with other visual dysfunctions, such as accommodative disorders. Each condition may need to be addressed separately.28 Management of accommodative and vergence dysfunction may include visual therapy to increase fusional range, vergence facility, and accommodative amplitude and facility.3,21,24 Therapy may also target saccadic and pursuit dysfunctions.
Testing of these areas post-treatment along with subjective improvement of symptoms will indicate outcome and help the clinician develop a means of hopefully improving visual function.
Visual Field Defects
Various presentations of visual field defects may occur following TBI as a result of damage to any portion of the visual pathway from the visual cortex of the brain onward to the retina.4,7,25,26
Defects may include constriction of the fields and either isolated or multiple scattered defects throughout the fields, with or without a generalized decrease in sensitivity.4,7,25 Lateralized field defects such as homonymous hemianopias may also occur with or without neglect, in which patients are fully unaware of objects located in space within the visual field defects.
Symptoms of visual field defects include mobility issues (e.g., patients bumping into objects), reading difficulties and trouble locating items in tasks of daily living such as eating. Homonymous hemianopias create significant safety challenges, especially when associated with neglect and any activities that require an accurate awareness of one’s surroundings.4,7,25
Screening for gross field defects by confrontation testing is useful, but more detailed evaluation with automated or Goldmann perimetry is essential to accurately localize and quantify any suspected defects.
| Interventions: Low Vision Aids After performing refraction and BCVA assessment, consider low vision rehabilitation services and devices. For short duration distance-spotting tasks, such as seeing faces and signs, a hand-held or spectacle-mounted telescope apparatus might help. The patient may also find hand-held magnifiers useful for near-vision, such as reading price tags and labels. For more severe impairments, electronic hand-held magnifiers or closed circuit television may prove beneficial. Non-optical aids, such as large print or auditory devices (for instance, a talking watch), may be considered. Using tactile markers for the stove to help identify burner settings may assist with orientation. This can also be used for clothing to identify and coordinate colors and outfits. Today, low vision smartphone apps can help identify money and colors. If your office doesn’t have the resources to cater to low vision patients, refer them to a nearby specialist. Every state has an agency to provide services for the visually impaired. The individual may have already had low vision rehabilitation services, if the injury was longstanding. While long-term TBI patients may have already seen a low vision specialist, their needs and goals will change over time, as will available low vision technology. Afferent damage can also reduce contrast sensitivity, which a patient may refer to as “blurred vision.” Often, having proper illumination helps increase the contrast to allow for comfortable reading. The amount, type and intensity of illumination are subjective. Another common complaint from patients with contrast sensitivity loss is difficulty with steps and curbs. This typically occurs in severe cases. Refer these patients to an orientation and mobility specialist. |
Useful field of vision (UFOV) testing may also be helpful. This computer-based assessment of focused and divided visual function requires the patient to complete three subtests with variable numbers of objects and background noise.15,26 UFOV may serve as a more effective evaluation of one’s visual attention in an environment of distraction, or noise, and may better predict an individual’s visual function in real-world situations.
Post-chiasmal afferent damage can cause visual field loss, such as a homonymous hemianopia or a hemianopia of just one eye.27,28 Patients may complain of bumping into objects on the side of the field loss and a fear of traveling independently. Individuals with loss of inferior field may have greater problems with mobility, as they may have difficulty with stairs and curbs. Rehabilitation goals stress peripheral awareness and may include scanning and compensatory training, or large diopter Fresnel prism placed base out toward the field loss.7,8
Other lens options include peripheral prismatic lenses, such as the Gottlieb, Peli or Sectoral prisms. Specialty optical labs may make these devices. Further training with these lenses with a rehabilitation specialist is needed.
Individuals with hemianopic field loss may also have difficulty reading, with left-sided field loss preventing visualization of the first few words of a sentence or right-sided loss obscuring the last few words. Compensatory techniques such as scrolling and tilting the material away from field loss may help. A typoscope or line guide may help with tracking.
Since photosensitivity and glare are common complaints in our local population at the Philadelphia (Corporal Michael J. Crescenz) Veterans Administration Medical Center, we conducted a small study to evaluate responses to various tints and filters by 20 patients (age 24 to 64 years; 18 males and two females). Before prescribing a tint, indoor glare was tested with lights of various intensities affixed to the ETDRS chart, and outdoor glare under bright sunlight, intermittent sunlight and overcast conditions. To accomplish this, the patient stands with their back towards the sun and then turns and walks into the direction of the sun while trialing several tints. Patient preference for filters varied, with no clear associations noted. However, a wraparound prescription quality frame with the appropriate tint has been quite helpful for our patients. The results of this unpublished study show that the appropriate tint for each patient resulted in reduction of glare sensitivity for all patients. It is therefore important that the clinician consider multiple options to provide the best comfort possible as each patient may have differing success with different options. |
Visual-vestibular Disorders
TBI patients may experience visual-vestibular dysfunctions characterized by dizziness, vertigo, nausea, balance problems, photophobia in fluorescent lighting and increased motion sensitivity.4,10,29 Words may seem to move around while reading and computer tasks may be problematic due to monitor light flicker or sensitivity to scrolling motions. Visual-vestibular disorders likely occur due to abnormal function of the vestibular-ocular reflex (VOR) that controls gaze stabilization. The VOR stabilizes images on the retina during head movements by producing eye movements opposite that of the head movements.4,12,16,29-32
Dysfunction of the VOR may occur with damage to the semicircular canals in the ears, the oculomotor nerve (CNIII), the abducens nerve (CNVI), the acoustic nerve (CNVIII), or any neurological structures integrating these components.4,10,15,16,22,31,32 Detailed questioning regarding related symptoms in the patient history is especially useful in identifying and addressing potential visual-vestibular disorders that may result from TBI. For these patients, consultation with an ear, nose and throat specialist may be warranted to rule out other pathology. Also, a consultation with an occupational therapist may be beneficial for an evaluation and assistance for mobility purposes.
In our offices, we will all ultimately see patients who have had TBI, whether it be from an athletic or service injury, motor vehicle accident or fall. These patients often have visual concerns that range in complexity and severity. Keeping abreast of both the successes and limitations of recent research can help clinicians tailor a treatment plan to the individual’s needs.
Dr. Suhr practices at the Corporal Michael J Crescenz Department of Veterans Affairs Medical Center.
Dr. Shust practices at the Corporal Michael J Crescenz Department of Veterans Affairs Medical Center.
Dr. Prasad practices at the University of Pennsylvania Scheie Eye Institute and the Corporal Michael J Crescenz Department of Veterans Affairs Medical Center.
Dr. Wilcox practices at the Corporal Michael J Crescenz Department of Veterans Affairs Medical Center and works in the Advanced Low Vision Clinic.
Dr. Chronister practices at the Corporal Michael J Crescenz Department of Veterans Affairs Medical Center and works in the Advanced Low Vision Clinic.
1. Wiebe D, Comstock D, Comstock M. Concussion research: a public health priority. Inj Prev. 2011;17:69-70.2. McCrory P, Meeuwisse W, Aubry M, et al. Consensus statement on concussion in sport: the 4th International Conference on Concussion in Sport held in Zurich, November 2012. Br J Sports Med. 2013;47:250-8.
3. Faul M, Xu L, Wald M, Coronado V. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations and Deaths (2002-2006). Atlanta, GA: Centers for Disease Control and Prevention, National Centre for Injury Prevention and Control; 2010. http://www.cdc.gov/traumaticbraininjury/get_the_facts.html. Accessed March 20, 2015
4. Kapoor N, Ciuffreda K. Vision deficits following acquired brain injury. In: Cristian A. Medical management of adults with neurologic disabilities. New York, NY: Demos Medical Publishing; 2009;407-23.
5. Chang T, Ciuffreda K, Kapoor N. Critical flicker frequencyand related symptomsin mild traumatic brain injury. Brain Injury. 2007;21:1055-62.
6. Du T, Ciuffreda K, Kapoor N. Elevated dark adaptation thresholds in traumatic brain injury. Brain Injury. 2005;19:1125-38.
7. Kapoor N, Ciuffreda K. Vision disturbances following traumatic brain injury. Current Treatment Options. Neurology. 2002;4:271-80.
8. Schrupp L, Ciuffreda K, Kapoor N. Foveal versus eccentric retinal critical flicker frequency in mild traumatic brain injury. Optometry. 2009;80:642-50.
9. Hellerstein L, Freed S, Maples W. Vision profile of patients with mild brain injury. Journal of American Optometric Association. 1995;66:634-9.
10. Ciuffreda K, Kapoor N, Rutner D, et al. Occurrence of oculomotor dysfunctions in acquired brain injury: A retrospective analysis. Optometry. 2007;78:155-61
11. Neitz J, Neitz M. Colour vision: The wonder of hue. Current Biology. 2008;18:700-2.
12. Schlosser H, Lindenmann J, Vakkoczy P, Clarke A. Vestibulo-ocular monitoring as a predictor of outcome after severe traumatic brain injury. Critical Care 2009;13(6):R192. Epub 2009 Nov 30.
13. Miles F. The neural processing of 3-D visual information: Evidence from eye movements. European Journal of Neuroscience. 1998;10:811-22.
14. Padula W, Argyris S, Ray J. Visual evoked potentials evaluating treatment for post-trauma vision syndrome in patients with traumatic brain injuries. Brain Injury. 1994;8:125-33.
15. Greenwald B, Kapoor N, Singh A. Visual impairments in the first year after traumatic brain injury. Brain Injury. 2012;26(11):1338-59.
16. Leigh R, Zee D. The neurology of eye movements. 4th ed. New York:Oxford University Press; 2006.
17. Thiagarajan P, Ciuffreda K. Short-term persistence of oculomotor rehabilitative changes in mild traumatic brain injury (mTBI): A pilot study of clinical effects. Brain Injury. 2015;29(12):1475-9.
18. Sabates N, Gonce M, Farris B. Neuro-ophthalmological findings in a closed head trauma. Journal of Clinical Neuroophthalmology. 1991;11:273-7.
19. Galetta K, Barrett J, Allen M, et al. The King-Devick test as a determinant of head trauma and concussion in boxers and MMA fighters. Neurology 2011 Apr 26;76(17):1456-62.
20. Magone M, Kwon E, Shin S. Chronic visual dysfunction after blast-induced mild traumatic brain injury. J Rehabil Res Dev. 2014;51(1):71-80.
21. Thiagarajan P, Ciuffreda K. Effect of oculomotor rehabilitation on vergence responsivity in mild traumatic brain injury. J Rehabil Res Dev. 2013;50(9):1223-40.
22. Master C, Scheiman M, Gallaway M, et al. Vision diagnoses are common after concussion in adolescents. Clin Pediatr (Phila). 2015. Epub ahead of print.
23. Thiagarajan P, Ciuffreda K. Effect of oculomotor rehabilitation on accommodative responsivity in mild traumatic brain injury. J Rehabil Res Dev. 2014;51(2):175-91.
24. Ciuffreda K, Rutner D, Kapoor N, et al. Vision therapy for oculomotor dysfunctions in acquired brain injury: a retrospective analysis. Optometry. 2008;79(1):18-22.
25. Suchoff I, Kapoor N, Ciuffreda K, et al. The frequency of occurrence, types and characteristics of visual field defects in acquired brain injury: A retrospective analysis. Optometry. 2008;79:259-65.
26. Atkins E, Newman N, Biousse V. Post-traumatic visual loss. Rev Neurol Dis. 2008;5(2):73-81.
27. Dundon N, Bertini C, Làdavas E, et al. Visual rehabilitation: visual scanning, multisensory stimulation and vision restoration trainings. Front Behav Neurosci. 2015;9:192.
28. Goodwin D. Homonymous hemianopia: challenges and solutions. Clin Ophthalmol. 2014;8:1919-27.
29. Cohen A. The role of optometry in the management of vestibular disorders. Brain Injury/Professional. 2005;2:8-10.
30. Bronstein AM. Vision and vertigo: Some visual aspects of vestibular disorders. Journal of Neurology. 2004;251:381-7.
31. Ciuffreda K. Visual vertigo syndrome: A clinical demonstration and diagnostic tool. Clinical Eye Vision Care. 1999;11:41-42.
32. Schweigart G, Mergner T, Evdokimidis I, et al. Gaze stabilization by optokinetic reflex (OKR) and vestibule-ocular reflex (VOR) during active head rotation in man. Vision Research. 1997;12:1643-52.
33. Judge S, Cumming B. Neurons in the monkey midbrain with activity related to vergence eye movement and accommodation. J Neurophysiol. 1986;55(5):915-30.
34. Alvarez T, Kim E, Vicci V, et al. Concurrent vision dysfunctions in convergence insufficiency with traumatic brain injury. Optom Vis Sci. 2012;89(12):1740-51.
35. Barnett B, Singman E. Vision concerns after mild traumatic brain injury. Curr Treat Options Neurol. 2015;17(2):329.
36. Hunt A, Mah K, Reed N, et al. Oculomotor-based vision assessment in mild traumatic brain injury: A systematic review. J. Head Trauma Rehabil. 2015; Aug 19. Epub ahead of print.


