 |
A 19-year-old Jamaican male presented with “cloudy vision” in the left eye for nearly two months. He was diagnosed with toxoplasma chorioretinitis in his home country and treated with Bactrim DS (sulfamethoxazole-trimethoprim, Roche Pharmaceuticals), but his symptoms did not improve. Lab tests for toxoplasmosis came back negative, and the patient was referred for a second opinion given the lack of improvement.
On examination, his unaided visual acuity was 20/20 OD and 20/800 OS with no improvement with pinhole. A relative afferent pupillary defect was detected in the left eye. His extraocular motilities were full in both eyes and his left eye had a mild peripheral constriction on confrontation visual fields. The intraocular pressures (IOP) measured 14mm Hg OD and 7mm Hg OS. The anterior segments were unremarkable. Fundus photos and corresponding fluorescein angiograms (FA)—early and late phase—reflect the posterior segments findings (Figures 1 to 3).
 |
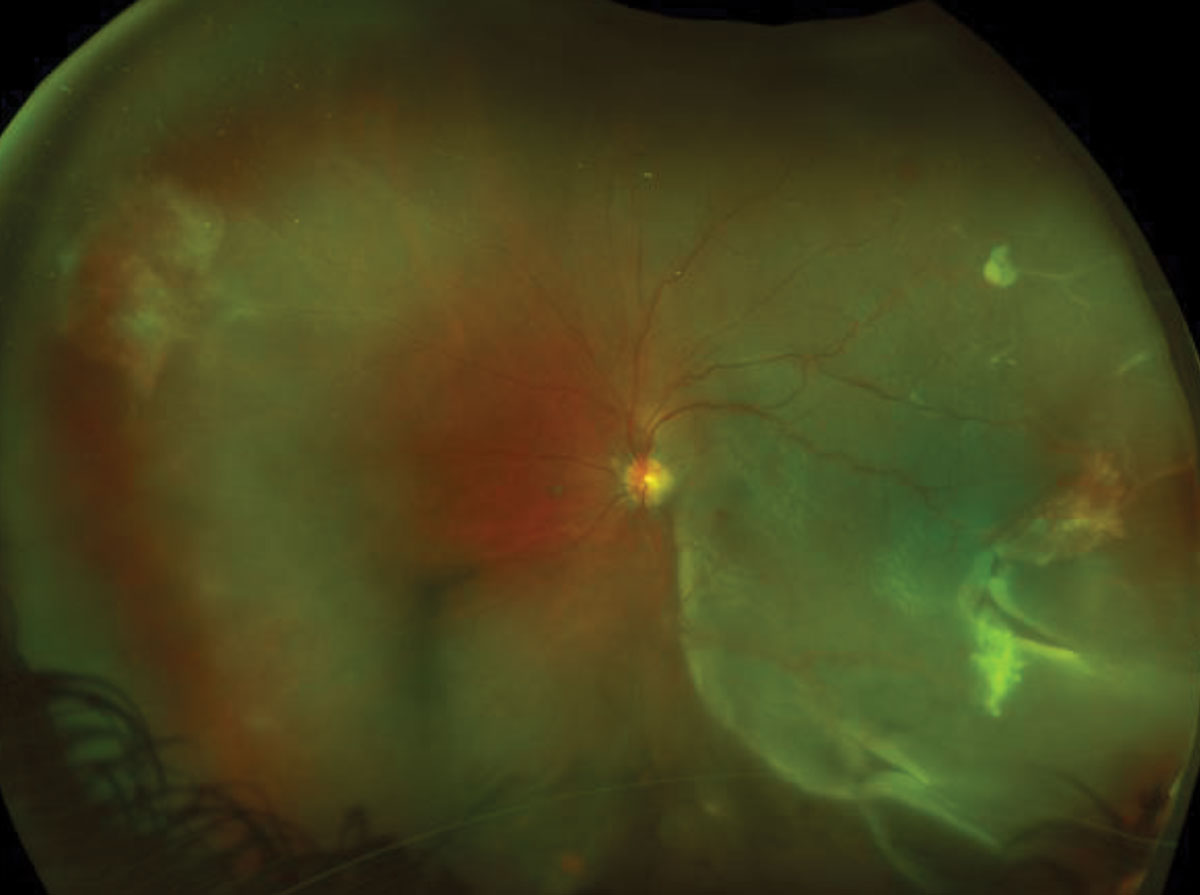 |
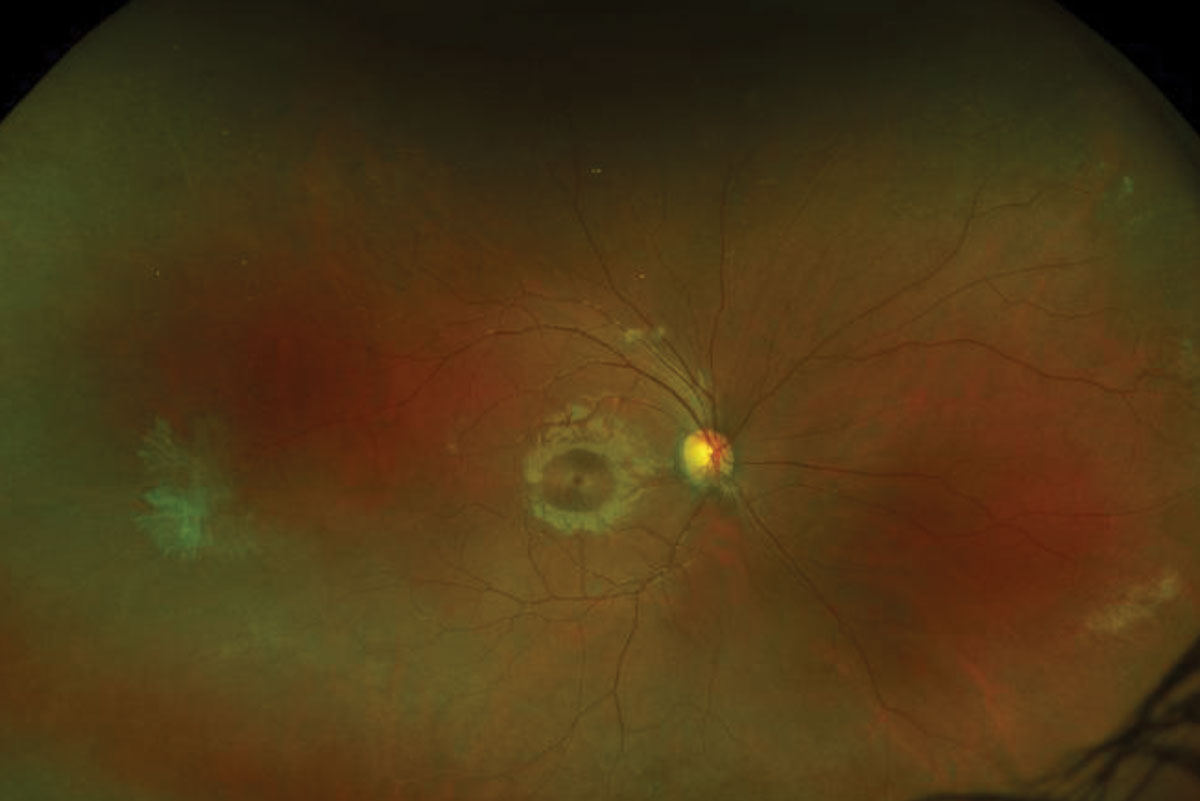
| Fig. 1. These ultra-widefield fundus photos show our young patient’s right (at top) and left eyes. What do you notice that can explain his blurry vision? Click image to enlarge. |
Take the Retina Quiz
1. How would you characterize the peripheral retinal changes in the right eye?
a. Multiple retinal tears.
b. Choroidal neovascularization.
c. Retinal neovascularization.
d. Retinal capillary angiomas.
2. How would you characterize the peripheral retinal changes in the left eye?
a. Rhegamtogenous retinal detachment.
b. Retinal neovascularization.
c. Retinal capillary angiomas.
d. Both a and b.
3. Which of the following best describes the FA findings?
a. Early hyperfluorescence with late staining.
b. Early hyperfluorescence with late leakage.
c. Early phase is normal with late pooling.
d. Hyperfluorescence associated with window defect.
4. What is most likely diagnosis consistent with the patient’s clinical presentation?
a. Eales’ disease.
b. Familial exudative vitreoretinopathy.
c. Proliferative diabetic retinopathy.
d. Proliferative sickle cell retinopathy.
5. Based on the Goldberg classification, the patient’s left eye would be categorized as:
a. Stage II.
b. Stage III.
c. Stage IV.
d. Stage V.
For answers, see below.
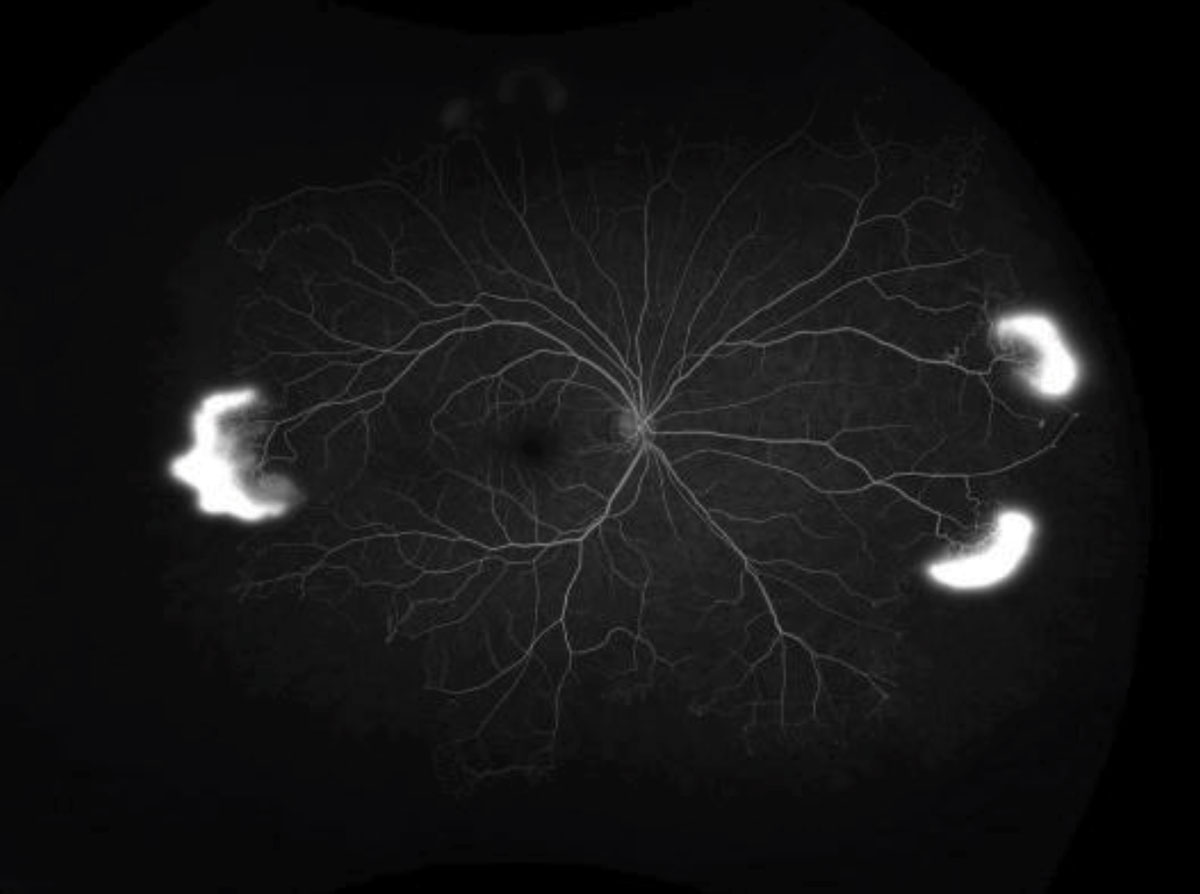 |  |
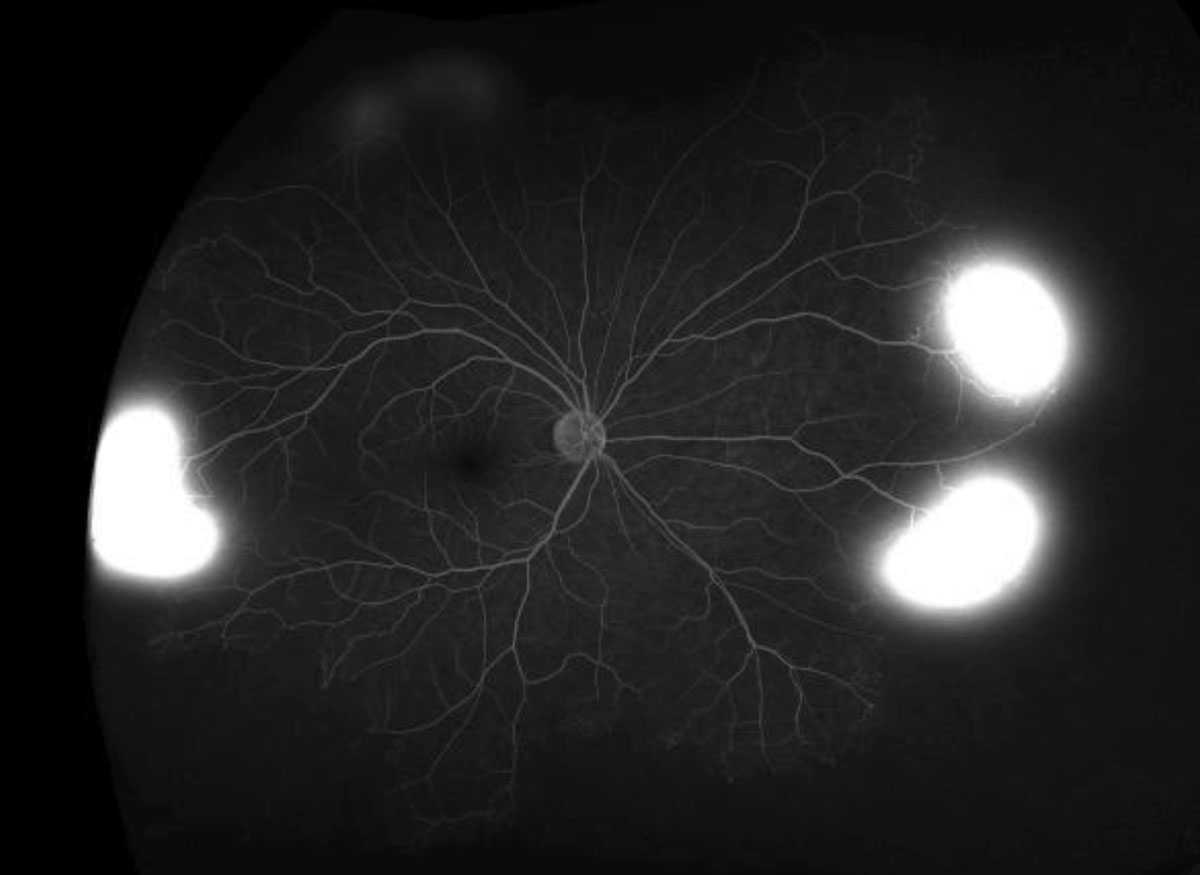
| Fig. 2. The ultra-widefield FA shows our patient’s right eye in mid phase, at left, and left eye in the early phase. Click images to enlarge. | |
Diagnosis
It was evident that our patient has a large macula-off retinal detachment in the left eye. We were able to see multiple retinal tears, one of which can be seen in the fundus photograph at 4:00. But that’s not all. Superior to the retinal tear, he also had an area of active neovascularization in the shape of a sea-fan. The neovasculation was not just in the left eye but also in the peripheral retina of the right eye. The widefield FA in the left eye highlights where we can see hyperfluorescense in the early phase and leakage (via increased hyperfluorescence with blurring of margins) in the late phase (Figure 2). The FA in the right eye also shows large areas of capillary nonperfusion anterior to the areas of neovascularization (Figure 2).
Given these findings, we concluded our patient had a traction retinal detachment secondary to proliferative sickle cell retinopathy that evolved into a combined tractional-rhegmatogenous retinal detachment (TRD/RRD).
Goldberg Stages of Sickle Cell Retinopathy1-6I. Peripheral arterial occlusions – Can have multiple simultaneous peripheral vascular occlusions and silver-wiring of arterioles. II. Peripheral arteriovenous anastomoses (hairpin loops) – Vascular remodeling at the junction of perfused and nonperfused retina that strives to shunt blood from occluded arterioles to adjacent venules. These lesions do not leak FA. III. Neovascular and fibrous proliferations – Defined by sea-fan lesions that are typically found in the peripheral retina. These lesions do leak on FA. IV. Vitreous hemorrhage – Defined by the presence of vitreous hemorrhage. Typically secondary to vitreous traction and can be focal overlying sea-fan lesions or diffuse. Chronic vitreous hemorrhage can lead to fibroglial membranes and vitreous strands that can induce traction. V. Retinal detachment – Defined by the presence of a TRD, usually secondary to chronic non-clearing vitreous hemorrhage. Can also develop combined TRD/RRD. |
Discussion
Sickle cell disease comprises a group of hemoglobinopathies characterized by intravascular hemolysis that ultimately results in defective oxygen transportation.1,2 Sickle cell disease is the most common inherited blood disorder and affects nearly 100,000 Americans.2,3 Approximately one in 12 black individuals carry the sickle cell trait and sickle cell disease affects ~1/365-500 African-American births and ~1/16,300 Hispanic-American births.1-3
The patient had a known diagnosis of hemoglobin (Hb) SC disease with no prior systemic crises of note. Hemoglobin SC disease is the second most common form of sickle cell disease, but is the most likely to cause sickle cell retinopathy.1-6 Hemoglobin SS disease is the most common form of sickle cell disease, but is associated with a higher rate of mortality.2,6,7
Hemoglobin SC results in hypercoagulability of blood and is more likely to compromise smaller blood vessels and cause chronic low-grade peripheral ischemia, while Hb SS disease causes larger thrombi that are more likely to affect large blood vessels and cause systemic crises.1,4,5
SC Retinopathy Findings1-6Nonproliferative • Salmon patch hemorrhages – Pinkish-orange hemorrhages between the retina and internal limiting membrane, typically found in the equatorial retina. • Iridescent spots – Small schisis cavities post-salmon patch hemorrhage or post-arteriolar occlusions that appear with multiple glistening, refractile iridescent spots. • Black sunbursts – Flat areas of hyperpigmentation thought to be secondary to intraretinal hemorrhages tracking into the subretinal space or representative of focal underlying choroidal ischemia or neovascular membrane formation. Proliferative • Sea-fan lesions – Neovascular fronds that typically form in the periphery at arteriovenous crossings secondary to peripheral arteriolar occlusions. |
Diagnosis is largely clinical and graded using a universal classification system (See, “Goldberg Stages of Sickle Cell Retinopathy”). According to the Goldberg classification, our patient is in Stage III OD and Stage V OS. The hallmark defining feature of Stage III disease is sea-fan neovascular fronds, and the defining feature of Stage V disease is traction retinal detachment.
Proliferative sickle cell retinopathy is unique from other proliferative vitreoretinal diseases in that the neovascular sea-fan lesions can autoinfarct in up to 60% of cases, therefore becoming inactive and no longer pose a threat to vision.1-7 For that reason, patients with proliferative sickle cell retinopathy are treated on a case-by-case basis and treatment may not be uniformly indicated. If intervention is indicated, options include laser, anti-vascular endothelial growth factor intravitreal injections and surgery (for non-clearing vitreous hemorrhage or retinal detachment). The ultimate goal of treatment is to prevent progression from Stage III to Stages IV or V when possible.
Ultra-widefield FA can provide great insight as to whether sea-fan lesions are active or inactive, which can help guide the decision to treat versus observe. Spectral domain OCT and OCT angiography can provide information regarding the foveal architecture and presence of ischemia.1,2,4
 | 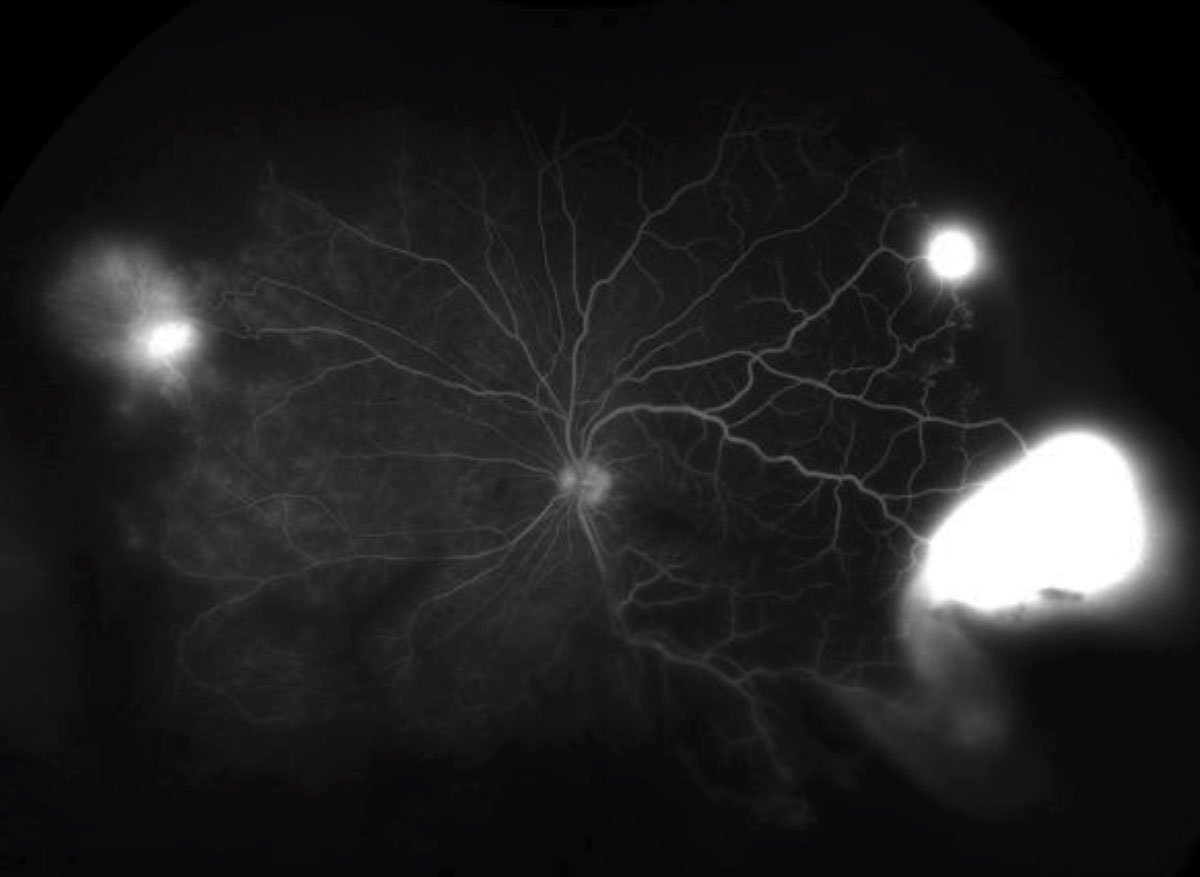 |
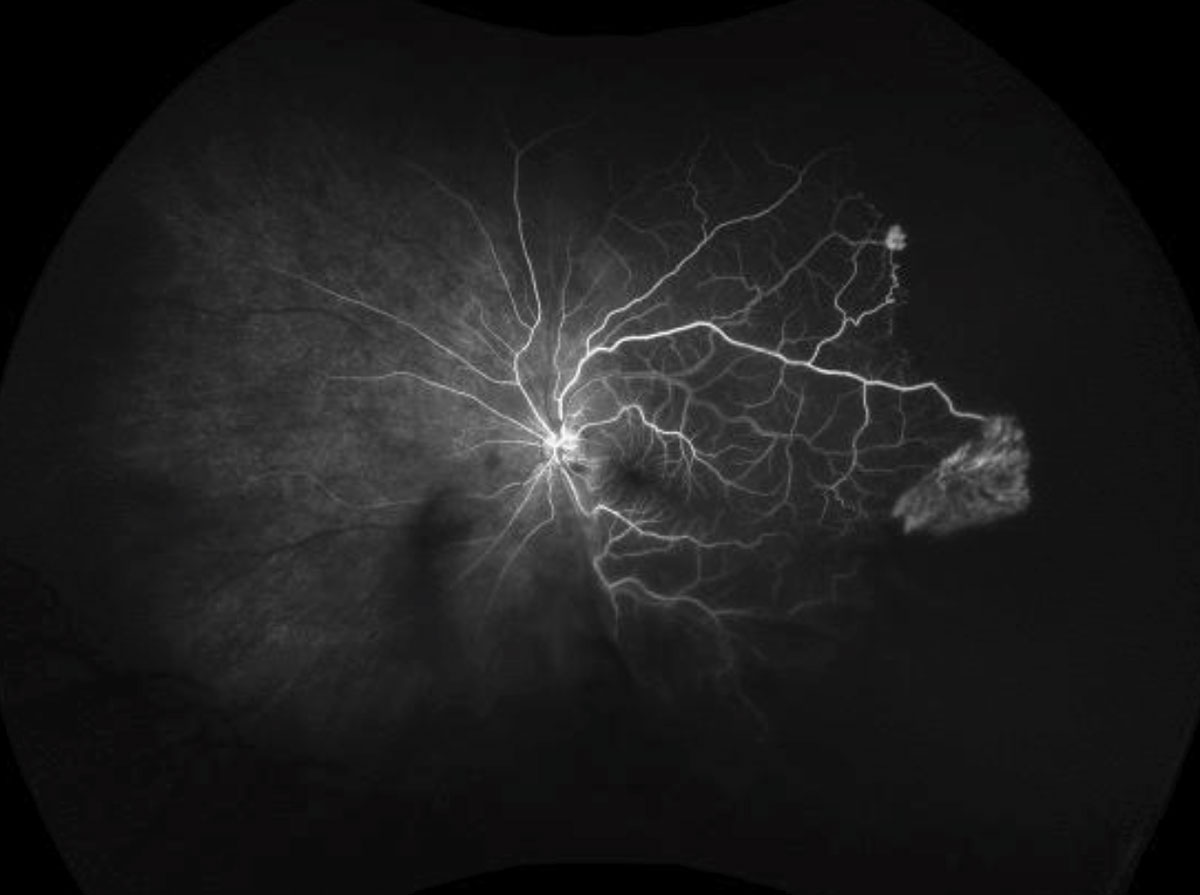
| Fig. 3. These ultra-widefield FA images depict the late phase in both eyes. Click images to enlarge. | |
After a discussion about the risks, benefits and alternatives, the patient was taken into surgery. Panretinal photocoagulation was applied anterior to the sea-fan lesions nasally and temporally in his right eye. The combined TRD/RRD in the left was repaired with the placement of a scleral buckle, pars plana vitrectomy, endoscopic photocoagulation and insertion of silicone oil. The patient had a good surgical result with reattachment of the retina and excellent regression of the sea-fan lesion in both eye. He continues to be followed closely.
Dr. Aboumourad practices at the Bascom Palmer Eye Institute in Miami. This case came courtesy of Jayanth Sridhar, MD, also of the Bascom Palmer Eye Institute.
| 1. Ryan SJ ed. Retina. 5th ed. Philadelphia: Elsevier Saunders; 2013. 2. Akhter M, Latting M, Scott Al. Management of proliferative sickle cell retinopathy. Eyenet magazine. www.aao.org/eyenet/article/proliferative-sickle-cell-retinopathy. October 2018. Accessed April 17, 2020. 3. Centers for Disease Control and Prevention. Data & statistics on sickle cell disease. www.cdc.gov/ncbddd/sicklecell/data.html. October 21, 2019. Accessed April 17, 2020. 4. Yanoff M, Duker JS, eds. Ophthalmology. 5th ed. Philadelphia: Elsevier; 2019. 5. Menaa F, Khan B, Uzair B, Menaa A. Sickle cell retinopathy: improving care with a multidisciplinary approach. J Multidiscip Healthc. 2017;10:335-46. 6. Gass J, Agarwal A. Gass’ Atlas of Macular Diseases, 5th ed. Philadelphia: Elsevier Saunders; 2012. 7. Platt O, Brambilla D, Rosse W, et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. N Engl J Med. 1994;330(23):1639-44. |
Retina Quiz Answers:
1) c; 2) d; 3) b; 4) d; 5) d.

