 |
Laser procedures represent the next frontier in optometric scope expansion for many states. Knowing the risks, benefits and indications for these procedures is critically important for doctors who would perform them. Yet, understanding the periprocedural management of laser surgical patients is equally crucial for success in these endeavors.
This month, we review some of the more common laser procedures for the anterior segment, focusing on pre- and postoperative care to ensure the best possible outcomes.
Techniques
Currently, certified optometrists in Kentucky, Louisiana and Oklahoma can perform laser procedures, but only procedures that involve structures of the anterior segment. Three specific techniques account for the vast majority of laser procedures by ODs. They are:
• Laser capsulotomy for posterior capsular fibrosis, sometimes referred to as “after-cataract.”
• Laser peripheral iridotomy (LPI) for those at risk for or suffering from angle closure glaucoma.
• Laser trabeculoplasty using either the argon laser (ALT) or the Nd:YAG laser (selective laser trabeculoplasty or SLT).
Less frequently, lasers may be used to perform iridoplasty on narrow angles, or to remove lesions from the ocular adnexa. Techniques that are expressly forbidden in these states include retinal laser procedures (e.g., focal or panretinal laser photocoagulation), laser in situ keratomileusis (LASIK) and other refractive or cosmetic surgeries.
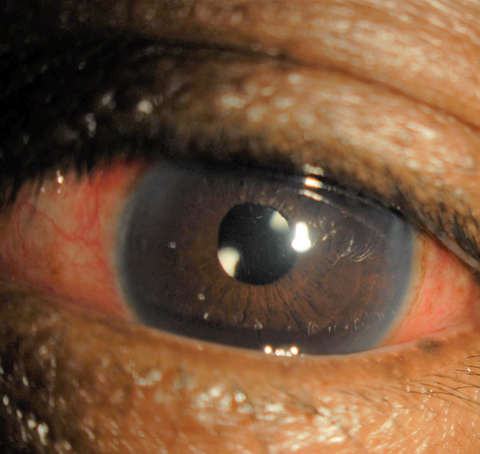 |
| This one-day postoperative cataract surgery patient displays redness indicating inflammation, similar to the inflammation laser procedure patients face. Click image to enlarge. |
YAG Capsulotomy
Opacification of the posterior capsule is a common complication of phacoemulsification surgery for cataracts, with estimates as high as 18% of cases.1 Researchers believe it occurs because of postoperative proliferation and migration of residual lens epithelial cells within the posterior capsular bag.2,3 Symptoms of posterior capsule opacification (PCO) may not become manifest for months or even years after the initial surgery, but can include such diverse complaints as blurred vision, decreased contrast sensitivity, increased glare and even monocular diplopia.1 The only remedy for this problem is laser capsulotomy.2 Essentially, this involves using the Nd:YAG laser to create an opening in the posterior capsule, eliminating obscuration of the visual axis.
Discharging laser energy into the eye can result in a host of undesirable sequelae. These commonly consist of transiently elevated intraocular pressure (IOP), postoperative inflammation (i.e., mild anterior uveitis) and an increase in floaters.1 Less frequent, but more serious, complications include retinal detachment, cystoid macular edema, pitting or displacement of the lens implant, iris hemorrhage and vitreous prolapse.1,4 To minimize these complications, the physician must make appropriate use of perioperative medications and perform careful postoperative assessment.
To guard against a pressure spike, it is customary to instill an IOP-lowering agent prophylactically several minutes before or immediately after performing laser capsulotomy.1,4 Historically, 1% apraclonidine was the drug of choice, but today 0.2% brimonidine is often used in a similar fashion.5 Studies show these agents can help minimize IOP spikes.5,6 The greatest incidence of IOP elevation occurs within three hours of the procedure and tends to return to normal within 24 hours.1 Those in whom higher amounts of laser energy are used during the procedure have a greater tendency toward more severe or prolonged IOP elevation, as do those with a prior diagnosis of glaucoma or ocular hypertension.7 For these individuals, recheck the IOP one to four hours after the procedure; if elevated, the continued use of a topical IOP-lowering agent during the postoperative period is recommended.4
Corticosteroid drops are also commonly given postoperatively after laser capsulotomy to curb associated inflammation. Prednisolone acetate 1% QID remains the standard for most physicians, although some practitioners prefer loteprednol 0.5% QID or even difluprednate 0.05% BID-TID. Follow-up is usually at one week following surgery, at which point IOP is measured and the anterior chamber assessed. If normal, drops can be discontinued at this point. Obviously, if pain, reduced vision, newly observed flashes or floaters occur at any point postoperatively, urge the patient to return immediately. Routine dilation following capsulotomy is generally not necessary unless symptoms dictate otherwise.
Peripheral Iridotomy
This procedure, which seeks to alleviate pupil-block, results in deepening of the anterior chamber angle and alleviating or preventing angle-closure glaucoma (ACG) by creating a pathway for aqueous egress from the posterior to the anterior chamber and into the trabecular meshwork (TM). This technique can be performed as a therapeutic measure for those in acute or chronic ACG, or as a prophylactic measure for those determined to be at increased risk (i.e., anatomically narrow angles).8 The Nd:YAG laser is typically the device of choice for this procedure, although in dark irides both argon and Nd:YAG lasers may be used sequentially.8
The risks associated with LPI are similar to those encountered with laser capsulotomy, including postprocedural IOP elevation and anterior uveitis. Less common sequelae include burns to the cornea or retina, or both, macular edema and retinal detachment.9 To prevent an associated IOP spike, one drop of apraclonidine or brimonidine is instilled about 20 minutes before the procedure.10 In addition to lowering IOP, pilocarpine is also used to induce miosis, stretching the iris and facilitating penetration of the tissue with less laser energy.10 IOP should be checked roughly 30 minutes to 60 minutes after the procedure. As with laser capsulotomy, the use of topical corticosteroids is helpful in minimizing post-procedural inflammation. The one-week follow up involves inspection of the iridotomy site to ensure patency, IOP measurement and assessment of intraocular inflammation. Physicians may also complete a one-month follow-up, which includes a dilated fundus examination. If IOP is elevated with dilation after LPI, the clinician should suspect alternative mechanisms of glaucoma (e.g., plateau iris or other non-pupil block conditions).
Dr. Sowka’s Personal Perspective |
| We detailed in an earlier column how we encountered a printed brochure extolling that SLT creates no side effect.1 However, risks are associated with any procedure and laser procedures are no exceptions. I am personally aware of a situation where a patient with ocular hypertension underwent a so-called benign SLT, only to develop an intractable IOP spike into the 40s necessitating bilateral trabeculectomy. I myself recently underwent one of the above-mentioned laser procedures without complications, until I awoke the next morning with hazy vision, steamy corneal edema, and an IOP spike into the 50s! Fortunately, the complication was short-lived and easily managed, but had I been a fragile glaucoma patient, the outcome could have been poor. 1. Sowka J, Kabat A. Irresponsible education. Rev. of Optom. 2016 April;153(4):88-9. |
Laser Trabeculoplasty
In this procedure, the laser is directed at the pigmented epithelium of the TM via a gonioscopy lens. Although the exact therapeutic mechanism remains incompletely understood, the successful result is diminished IOP by way of enhanced outflow through Schlemm’s canal.11 ALT acts through thermal alteration of the TM while SLT enhances biologic activity in this structure.11 Because SLT induces far less structural damage to the TM than ALT, SLT can potentially be repeated once if IOP control falters over time.12
As with LPI and YAG capsulotomy, the main complications of SLT include a postprocedural IOP spike and secondary inflammation.9 This paradoxical IOP spike seems to happen more commonly in heavily pigmented angles such as those encountered in exfoliative and pigmentary glaucoma.13 The use of apraclonidine or brimonidine before and immediately after SLT helps to blunt this effect.9,11
Postoperative corticosteroid use remains controversial, although a symptomatic anterior chamber reaction is not uncommon following SLT. While some continue to advocate for steroids, others suggest that their use may hinder the subsequent IOP-lowering effect.11 A randomized, controlled study evaluating the use of anti-inflammatory agents following SLT revealed little difference between groups using 1% prednisolone acetate, 0.5% ketorolac or artificial tears QID for five days postoperatively.14
An ancient Greek philosopher once proclaimed, “Change is the only constant in life.” Our profession has changed dramatically since its inception, and it will continue to evolve. As we prepare for surgical privileges in optometry, we must be certain that we first comprehend the impact of our actions and can manage the potential complications effectively.
| 1. Boucher K, Ellis B, Lighthizer N. Capping off cataract surgery. Rev of Optom. 2015 Jun;152(6):44-52. 2. Sharon Y, Livny E, Mimouni M, et al. Laser capsulotomy following cataract surgery: comparing time to capsulotomy with implantation of two broadly used intraocular lenses. Indian J Ophthalmol. 2017 Feb;65(2):144-7. 3. Wormstone IM, Eldred JA. Experimental models for posterior capsule opacification research. Exp Eye Res. 2016 Jan;142:2-12. 4. Steinert RF. Nd:YAG laser posterior capsulotomy. American Academy of Ophthalmology. www.aao.org/munnerlyn-laser-surgery-center/ndyag-laser-posterior-capsulotomy-3. November 4, 2013. Accessed May 20, 2017. 5. Singhal D, Desai R, Desai S, et al. Use of topical brimonidine to prevent intraocular pressure elevations following Nd: YAG-laser posterior capsulotomy. J Pharmacol Pharmacother. 2011 Apr-Jun; 2(2):104-6. 6. Kaur M, Singh A, Kailwoo S. Apraclonidine 1% to prevent post Nd:Yag laser capsulotomy rise of intraocular pressure. JARMS. 2012;4(3):237-42. 7. Bhargava R, Kumar P, Phogat H, Chaudhary KP. Neodymium-yttrium aluminium garnet laser capsulotomy energy levels for posterior capsule opacification. J Ophthalmic Vis Res. 2015 Jan-Mar;10(1):37-42. 8. de Silva DJ, Gazzard G, Foster P. Laser iridotomy in dark irides. Br J Ophthalmol. 2007 Feb;91(2):222-5. 9. Van Alstine A, Caruso JM. Managing the post-op glaucoma patient. Rev of Optom. 2016 Oct;153(10):58-65. 10. Shetler J, Miller J, Lighthizer N. Breaking down barriers: iridotomy in optometric practice. Rev of Optom. 2016 May;153(5):74-82. 11. Leahy KE, White AJ. Selective laser trabeculoplasty: current perspectives. Clin Ophthalmol. 2015 May 11;9:833-41. 12. Latina MA, Tumbocon JA. Selective laser trabeculoplasty: a new treatment option for open angle glaucoma. Curr Opin Ophthalmol. 2002 Apr;13(2):94-6. 13. Zhou Y, Aref A. A review of selective laser trabeculoplasty: recent findings and current perspectives. Ophthalmol Ther. 2017 Mar 3. doi: 10.1007/s40123-017-0082-x. [Epub ahead of print] 14. Jinapriya D, D’Souza M, Hollands H, et al. Anti-inflammatory therapy after selective laser trabeculoplasty: a randomized, double-masked, placebo-controlled clinical trial. Ophthalmology. 2014 Dec;121(12):2356-61. |

