 |
The testing of the pupillary response to light is routinely performed by the eye care practitioner and imparts valuable information regarding the presence, laterality or magnitude of diseases that mainly affect the optic nerve, such as glaucoma.1 A relative afferent pupillary defect (APD), when present, is an important marker in the evaluation of unilateral or asymmetric function of the anterior visual pathway.1-4 It serves as an indication of reduced afferent input from the retina or optic nerve when compared with the fellow eye.3
Because it’s so commonplace, clinicians may tend to focus more on its diagnostic role than the physiology being tested. Understanding both, however, will provide a fuller picture of your patient’s pathology.
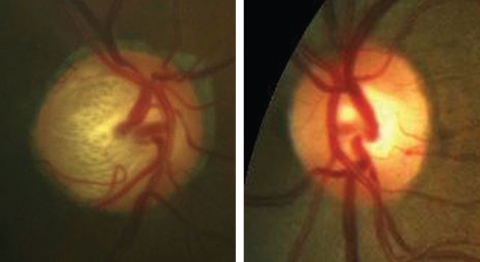 |
| Unilateral glaucoma, as seen in these fundus photos, more severe in the right eye than the left, could elicit an APD in the right eye. Click image to enlarge. |
The A&E Channel
The pupil response to light is best explained through the understanding of the light reflex pathway, which is comprised of two arms: the afferent and the efferent.
As the first arm, the afferent portion signifies the nerve impulse sent from stimulation of the retinal ganglion cells (RGCs) to the optic nerve by light, continuing down the optic tract, decussating at the chiasm and ultimately reaching the pretectal nucleus of the midbrain.
The efferent arm consists of the nerve impulse that exits the midbrain, traveling along the oculomotor nerve to the ciliary ganglion, resulting in constriction of both pupils in a healthy patient.5
APD and Glaucoma
Determining the presence or absence of an APD is of great clinical value, particularly in guiding the practitioner in the course of treatment for their glaucoma patients. Though glaucoma is typically a bilateral condition, it often initially presents unilaterally or bilaterally with variation in severity between the two eyes. It is well documented that patients with glaucoma exhibit lower amplitude, slower velocity and acceleration of pupil subjects when compared with healthy controls.6 Therefore, practitioners’ ability to determine the presence of an APD is a critical step in caring for patients suffering from glaucoma as well as those suffering from other types of optic neuropathies.
Table 1. Comparison of Pupil Testing Methods and Their Application | |||
| Pupil Test | Strengths | Weaknesses | Common Uses |
| Pupil Cycle Time | • Can be tested unilaterally | • Long test duration and lack of test standardization | • Not used |
| Infrared Video Pupillography | • Quantitative and objective method • Very sensitive | • Requires more high tech equipment • APD recorded in healthy subjects at times | • Clinical trials |
| Swinging Flashlight Test | • Easily performed chair side | • Subjective and requires technical skill for subtle APDs | • Routine eye exams |
Checking for an APD
Three major methods exist to determine the presence of an APD (Table 1). Although these tests are generally tried and true, one is best left alone with a preference for the first two.
Swinging flashlight test. Most commonly, the presence of an APD is evaluated in office using the swinging flashlight test, in which each pupil is illuminated and the velocity and amplitude of the pupillary response is compared.2 In a healthy patient, light stimulation into one pupil results in equal constriction of both. In a patient with an APD, the affected eye will have less pupillary constriction or will dilate in significant defects.6
The response can be quantified using a neutral density filter that is placed to dim the eye without the suspected APD. The filter is placed starting with 0.3 logs, and then gradually increasing the degree of light absorption. Once the APD is neutralized, the endpoint is reached as measured by the log unit necessary to achieve neutrality.5 Even in the case of a unilateral, fixed pupil, an APD is detectable through observation of the direct or consensual response of the reactive pupil.5 Animal studies show that a 0.6 log unit APD is observed in response to an approximate unilateral loss of 25% to 50% of RGCs.2,6
The test has its limitations, in that it requires a considerable amount of expertise by the examiner, especially in the detection of subtle defects.1 Due to its subjective nature, discrepancies between examiners are not uncommon, and small APDs are sometimes missed.7 Other difficulties in assessment of this test lie in patients with anisocoria, dark irises or very small pupils.2 Young patients may also exhibit hippus, which causes slight fluctuations in pupil size despite constant light stimulation.5
Infrared video pupillography. This technique, first introduced in 1958, illuminates the pupil by an infrared source and images it by a video camera, allowing observation of pupil characteristics in dark settings.6,7 By adjusting the stimulus between the two eyes until they reach the same amplitude, an APD can then be measured quantitatively by comparing the difference or latency between eyes.6 The precise calculation uses the ratio of the amplitude to the latency of the light reflex in each eye.7
The RAPDx (Konan Medical) device is used most often in clinical studies to objectively calculate an APD using log units.8 This high resolution device has demonstrated greater sensitivity in the detection of smaller APDs when compared with the swinging flashlight method.6 The machine’s duration, interval and intensity of the stimulus are fixed, maintaining a stable light reflex and eliminating examiner discrepancy.7
Because of the significance of APD testing and measurement in determining the diagnosis and progression of glaucoma, several studies investigated infrared video pupillography in its accuracy with glaucoma patients. One study reported the presence of an APD in 56% of glaucoma patients using pupillography, compared with only 29% detection using the swinging flashlight method.2,3
One disadvantage is that a small but detectable APD is sometimes observed in healthy subjects, limiting the specificity of this test modality.6
Besides the detection of an APD, pupillography may be used in other cases when pupil size is of diagnostic importance, such as differentiating physiological anisocoria from Horner’s syndrome or measuring pupil size for refractive surgery.9
Pupil cycle time (PCT). This technique, first presented in 1944, requires a much longer duration and observation of the patient through the slit lamp.6 Unlike the swinging flashlight test, this method does not require the fellow eye for comparison. Instead, a 0.5mm thick horizontal beam of light is oriented right at the inferior pupil margin, resulting in constriction. The beam remains in place until the constricted iris blocks the light and then re-dilates until the edge is placed in its initial position at the edge of the inferior pupil margin. The cycle continues and the oscillations are recorded to calculate the PCT using the average of five total measurements and multiplied by 30.6
No significant information is available regarding the exact correlation between prolonged PCT and optic nerve disease such as glaucoma. Additionally, the intensity of light stimulus has not been standardized, which renders this technique one with little clinical value.6
Clinical Significance
Aside from glaucoma, optic neuropathies such as ischemic optic neuropathy, optic neuritis, optic nerve compression from a mass lesion or traumatic optic neuropathies may also manifest with an APD in the affected eye.5
The correlation between an APD and macular disease is not yet well established. As 30% to 50% of all RGCs are located in the macula, it is likely to contribute to the pupillary light response.3 Recent emphasis has been placed on the detection of early glaucoma through analysis of macular integrity, but the degree to which the macula must be affected to elicit an APD is typically substantial.3 This is also true for macular diseases such as age-related macular degeneration.4
Anterior segment conditions, such as dense corneal opacities and amblyopia, do not affect the pupil response to light. Though it is rare for a cataract to cause an APD, one study reported that in severe, unilateral cases when vision is counting fingers or worse, an APD is often observed but, in the better-seeing eye, that later disappeared following extraction. The mechanism for this was undetermined, but the researchers concluded that in these particular cases, a defect in the visual pathway should only be suspected when the APD is noted in the eye with the dense cataract.10
Pupil testing is routinely performed on all patients, and can provide critical clinical insight, especially in cases of glaucoma, which is often difficult to diagnose in the early stages. Understanding the physiology governing our ability to evaluate pupil function and make sense of the data is imperative for not only narrowing down the disease differentials, but also in monitoring the course and progression of ocular disease.
Testing is a means to end. Prompt diagnosis allows us to heal our patients and stave off damage. Our abilities to diagnose, ultimately, are grounded in an understanding of the processes by which the various components that comprise the ocular system function and operate.
| 1. Kalaboukhova L, Fridhammar V, Lindblom B. Relative afferent pupillary defect in glaucoma: a pupillometric study. Acta Ophthalmologica Scandin. 2007;85:519-25. 2. Tatham AJ, Meira-Freitas D, Weinreb RN, et al. Estimation of retinal ganglion cell loss in glaucomatous eyes with a relative afferent pupillary defect. Invest Ophthalmol Vis Sci. 2014;55:513-22. 3. Gracitelli CP, Tatham AJ, Zangwill LM, et al. Asymmetric macular structural damage is associated with relative afferent pupillary defects in patients with glaucoma. Invest Ophthalmol Vis Sci. 2016;57:1738-46. 4. Takayama K, Ito Y, Kaneko H, et al. Cross-sectional pupillographic evaluation of relative afferent pupillary defect in age-related macular degeneration. Medicine. 2016;95(39):1-5. 5. Broadway DC. How to test for a relative afferent pupillary defect. Comm Eye Health J. 2016;29(96):1-2. 6. Chang DS, Xu L, Boland MV, Friedman DS. Accuracy of pupil assessment for the detection of glaucoma. Ophthalmology. 2013;120(11):1-15. 7. Takizawa G, Miki A, Maeda F, et al. Association between a relative afferent pupillary defect using pupillography and inner retinal atrophy in optic nerve disease. Clin Ophthalmol. 2015;9:1895-1903. 8. Tsukasa S, Ishikawa H, Asakawa K, et al. Evaluation of relative afferent pupillary defect using RAPDx device in patients with optic nerve disease. Neuro-Ophthalmology. 2016;40(3):120-4. 9. Wilhelm H, Wilhelm B. Clinical applications of pupillography. J Neuroophthalmol. 2003;23(1):42-9. 10. Lam BL, Thompson SH. A unilateral cataract produces a relative afferent pupillary defect in the contralateral eye. Ophthalmology. 1990;97(3):334-8. |

