Graft-vs.-host disease (GVHD)—an abnormal immune response to healthy host tissue following stem cell transplantation for the treatment of hematologic diseases—can be a complex condition for eye care practitioners to manage. It can lead to a host of ocular complications, the most significant of which is dry eye—seen in 40% to 76% of patients with GVHD.1 In particular, ocular GVHD can cause permanent severe aqueous-deficient dry eye (ADDE), as well as other dry eye etiologies, which can be visually debilitating. It is critical that practitioners correctly identify dry eye caused by GVHD and treat accordingly.
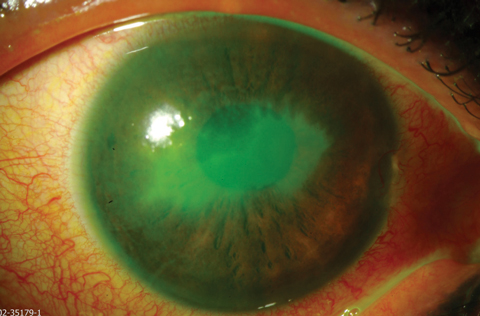 |
| Keratoconjunctivitis sicca is one manifestation that can help clinicians diagnose chronic GVHD. Photo: Christine W. Sindt, OD |
Disease Presentation
The onset of GVHD typically follows allogeneic hematopoietic stem cell transplantation (HCT), a therapy commonly used for patients suffering from a range of malignant and non-malignant hematologic diseases such as anemia, leukemia and thrombocytopenia.2
HCT most often follows the eradication of malignancies with chemotherapy, radiation or both, to replace stem cells lost from the blood and prevent patient mortality.3 GVHD occurs when the donor immune system attacks healthy recipient cells as foreign.2 Incidence of GVHD varies from 25% to 80% after allogeneic HCT, although most studies put it close to 50%.4
Human leukocyte antigen (HLA) disparity is by far the most powerful risk factor for the development of GVHD.5,6 Matching the donor HLA as close as possible to the recipient is critical before transplantation, as donor T-cells will view the host’s cells as foreign and attack if a large enough discrepancy is present.5 Other risk factors for GVHD include implantation of donor cells in a recipient of the opposite gender, advanced age of the recipient and significant damage during the conditioning process, a necessary step that serves to eradicate the host’s diseased cells and prevent rejection of the graft.7 However, it can also activate the host’s antigen presenting cells, in turn stimulating proliferation of donor T-cells.8
Forms of GVHD
GVHD can present as either acute or chronic. Most clinicians classify the disease based on the pathologic nature of the inflammation (Table 1). Acute GVHD, most commonly seen within the first 100 days after transplantation, resembles a toxic-like syndrome, which is typically seen in response to bacterial toxins. When acute GVHD affects the eyes, it usually responds well to steroids or other immunosuppressants.9,10
Chronic GVHD can have more dramatic ocular side effects and can cause significant long-term damage to the anterior segment of the eye. It is commonly seen more than 100 days after transplantation and has a relatively unknown pathophysiology. Successfully diagnosing chronic GVHD requires distinguishing from acute GVHD and other possible diagnoses, as well as identifying at least one distinctive manifestation of the disease, such as keratoconjunctivitis sicca, confirmed with diagnostic testing such as the Schirmer test.11,12 More recently, NaFl corneal staining is the preferred method for diagnosing keratoconjunctivitis.
Table 1. Acute vs. Chronic GVHD11,13-15 | ||
| Acute | Chronic | |
| Timing | Less than 100 days after transplant | More than 100 days after transplant |
| Pathophysiology | Direct donor T-cell response towards recipient cells | Loss of regulation of central tolerance causing indirect activation of the host’s T-cells against self |
| Tissues typically affected | Skin, bile duct and gastrointestinal system | Skin, lungs and mucous membranes |
Ocular Effects
GVHD can affect many ocular structures, including:
Lacrimal and meibomian glands. Chronic GVHD can affect several tissues of the eyes, the most common and most significant of which are the lacrimal glands. Research shows CD4+ and CD8+ T-cells cause cytotoxic effects on the periductal epithelial cells of the lacrimal gland.16,17 This damage causes permanent stenosis of the duct, resulting in moderate to severe ADDE similar to Sjögren’s syndrome (SS). One study found the tear turnover rate in GVHD patients is similar to that of patients with SS.18 The same study found the evaporation rate was highest in GVHD patients, and their lipid layer was the most unstable.18 Although the signs of ocular GVHD are similar to those with SS, ocular GVHD shows prominent fibrosis and an increase in activated T-cells and stromal fibroblasts in the glandular ducts, while SS shows the fibrosis and inflammation distributed through lesions in the acinar region of the lacrimal gland.16
Severe meibomian gland dysfunction (MGD) is also common in patients with GVHD. Interferometry confirms a decrease in the tear lipid layer in patients with ocular GVHD.19 However, research has yet to determine if the lipids are directly affected by the GVHD inflammation or due to the radiation during the conditioning process, which can destabilize the lipid layer.20,21 Regardless, it can cause further evaporation of the already-depleted aqueous of the tears.
Conjunctiva. Researchers estimate 12% of patients with acute GVHD and 11% of patients with chronic GVHD exhibit some form of conjunctival involvement.22 Inflammation can result in changes ranging from mild erythema to cicatrizing conjunctivitis similar to ocular cicatricial pemphigoid.22 Conjunctival inflammation tends to be more significant in acute GVHD and is more commonly hemorrhagic in presentation.23 Investigators have also linked GVHD patients to an increased incidence of conjunctival carcinoma.24
Cornea. Typically, this structure is not directly targeted in this disease process; however, significant secondary effects from lacrimal gland damage and altered tear makeup are often evident. This manifests as keratoconjunctivitis sicca, which, when severe, can lead to filamentary keratitis, corneal ulceration and corneal perforation.25,26 In rare cases, an immune-mediated limbitis can result in progressive neovascularization.27
Lenticular. Cataracts are a common ocular complication, with one study finding lens opacity in 39.4% of patients with GVHD.28 However, researchers note the lenticular changes were likely secondary to steroid use, radiation or both, as opposed to the disease itself.29
Posterior segment. When this is involved, which is rare, researchers speculate it may be secondary to the radiation or high-dose steroid treatments and not a direct complication of ocular GVHD.30
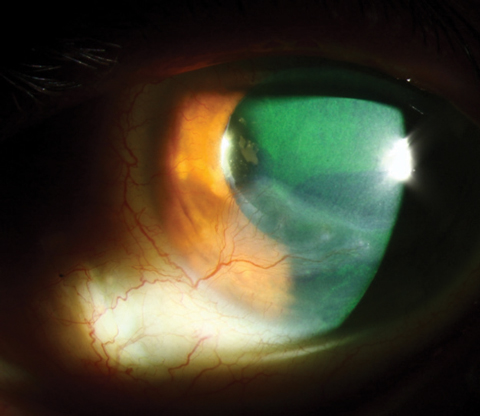 |
| Corneal neovascularization and scarring as a complication of GVHD. Photo: Christine W. Sindt, OD |
Dry Eye Diagnosis
In ocular GVHD, performing a diagnostic evaluation for dry eye is essential to determine the severity of the disease and monitor for improvement. Clinicians should use sodium fluorescein, rose bengal or lissamine green staining to determine both corneal and conjunctival involvement. Additional diagnostic tests include tear break-up time (TBUT), Schirmer scoring, meibomian gland evaluation, corneal sensitivity and tear osmolarity.31,32 A Schirmer score of less than 5mm at five minutes or new-onset dryness with a Schirmer score of 6mm to 10mm is sufficient for a diagnosis of chronic GVHD if distinctive manifestations are also seen in at least one other organ.11,31,32
In general, dry eye disease is characterized by hyperosmolarity of the tear film, which can lead to surface epithelium impairment by activation of proinflammatory cytokines released into tears.33 Research indicates a statistically significant correlation between tear hyperosmolarity and TBUT with osmolarity testing, but the same correlation is not seen with Schirmer testing or vital dye staining.33 Therefore, TBUT is an important measure of dry eye severity for patients with GVHD.
Numerous grading systems exist to assess the severity of ocular surface disease in these patients. The National Institutes of Health (NIH) research group developed one specifically for diagnosing and staging GVHD, which takes into consideration the severity of dry eye along with its effect on activities of daily living (Table 2).11 However, the NIH scoring system does not take into account the degree of corneal staining or other dry eye findings, instead focusing on the frequency of topical lubricant use. Because of this, some researchers and clinicians use the Dry Eye Workshop score, which is based on patient symptomatology, Schirmer score, TBUT, corneal and conjunctival involvement, as well as MGD.34
Table 2. NIH Ocular Scoring in Chronic GVHD11 | |
| Score | Definition |
| 0 | No dry eye symptoms. |
| 1 | Mild dry eye symptoms not affecting daily activities (requiring eye drops ≤3x/day) or asymptomatic signs of keratoconjunctivitis sicca. |
| 2 | Moderate dry eye symptoms partially affecting daily activities (requiring drops >3x/day or punctal plugs), without vision impairment. |
| 3 | Severe dry eye symptoms significantly affecting daily activities (special eyewear to relieve pain) or unable to work because of ocular symptoms or loss of vision caused by keratoconjunctivitis sicca. |
Ocular Treatment
Treating GVHD actually begins with prevention, which is accomplished through T-cell depletion. Prophylactic treatment is not without risks, which can include increased rates of graft failure, relapse of malignancy and infection.32 If preventative therapy fails, systemic immunosuppression is the mainstay for both acute and chronic GVHD. However, despite adequate systemic therapy, ocular complications may still arise. Ocular GVHD treatment focuses on increasing ocular surface moisture and decreasing ocular surface inflammation.26,31
Lubricants. Treatment typically begins conservatively with the use of preservative-free artificial tears and ointments, which are less likely to cause epithelial toxicity.35 Research also suggests hydroxypropyl cellulose ophthalmic inserts can improve dry eye symptoms and decrease the frequency of artificial tear use.26
Punctal Plugs. As lubricants alone are rarely adequate for the severe dryness seen in GVHD, punctal plugs may be a useful therapeutic option. Patients can show significant subjective improvement in symptoms and decrease in corneal staining with the use of punctal plugs over the course of twelve months. Also, there does not appear to be an increase in ocular inflammation, infection or other adverse events with the use of punctal plugs in patients with ocular GVHD.36
Topical corticosteroids. Local topical treatment with corticosteroids decreases the overall risk associated with prolonged and intense systemic immunosuppression and more effectively targets the affected area.26,37 In particular, ocular corticosteroid use promotes lymphocytic apoptosis and suppresses cell-mediated inflammation in the eye more directly than does systemic administration.37 Because long-term use of topical steroids carries the risk of steroid-induced glaucoma, cataracts, infectious keratitis and corneal thinning, many clinicians recommend short-term use combined with additional supportive therapy.26,31,37
Topical cyclosporine. This agent is another therapeutic option that shows great promise in the treatment of ocular GVHD. It is an immunosuppressive drug that inhibits T-cell proliferation on the ocular surface, increases goblet cell density in the conjunctiva, decreases epithelial cell apoptosis and interferes with the activity of proinflammatory cytokines in the conjunctiva.38,39 Several studies indicate statistically significant improvement in Schirmer basal secretion tests, TBUT, corneal fluorescein staining and patient symptoms after twice-daily cyclosporine 0.05% for at least three months.38,39 For refractory cases, compounding topical cyclosporine to 1% to 2% and increasing dosage to six to eight times per day may help promote healing during the early stages of ocular GVHD.40
Autologous serum. These eye drops contain epidermal growth factor, vitamin A, cytokines, nerve growth factors and fibronectin, all of which are essential for the proliferation, differentiation, maturation and integrity of the corneal and conjunctival epithelial surfaces.32,41-43 Studies show autologous serum is effective in treating severe dry eye, and one demonstrated 20% autologous serum dosed two to three drops ten times a day is effective in improving fluorescein and rose bengal staining, TBUT, corneal sensitivity and patient symptoms in ocular GVHD patients resistant to conventional artificial tear therapy.32,41-43
Tacrolimus. Another potential treatment option in the management of ocular surface inflammation from chronic GVHD is topical 0.03% tacrolimus ointment. This medication is FDA approved for the dermatological treatment of atopic eczema but has been used off-label to treat eczematous eyelid disease, atopic keratoconjunctivitis and other anterior segment inflammation.44 The mechanism of action is thought to be similar to that of cyclosporine, though the immunosuppressive potency of tacrolimus in vitro is 50 to 200 times greater than that of cyclosporine.31,45 Although beneficial effects of systemic tacrolimus in GVHD have been well documented, data regarding its use topically is limited.31,32,45
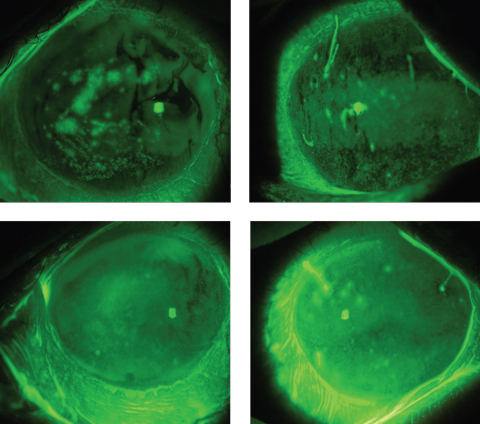 |
| These images depict a GVHD patient’s eyes before (top) and after scleral lens wear, showing an improved ocular surface, including a reduction in filaments, after just a few hours of wearing time. Click photos to enlarge. Photos: Alan Kwok, OD |
Contact Lens Wear
Several contact lens types are often used to treat ocular GVHD, including bandage silicone hydrogel lenses and gas permeable scleral lenses. Research suggests contact lenses reduce friction pain and control evaporation from the inflamed ocular surface.26 In one study, patients were fit in a monthly extended wear silicone hydrogel lens after failing to maintain adequate symptom control with topical lubricants and punctal plugs after three months.35,46 Investigators showed statistically significant improvement in patient symptoms in as little as two weeks with the use of these lenses, but little to no improvement in clinical signs of ocular surface disease and no significant changes to the tear film.35,46 Despite the absence of significant improvements in ocular surface disease, these lenses can still be considered in patients whose symptoms are not adequately controlled with conventional treatment. Patients fit in extended-wear contact lenses should be closely monitored due to the increased the risk of corneal edema, infiltrates, neovascularization and microbial keratitis.47
Scleral lenses may be the best option for treating ocular GVHD because the liquid reservoir beneath the lens allows for continuous hydration of the ocular surface, protecting against evaporation and aiding in resurfacing the damaged corneal epithelium, possibly by reducing the hyperosmolarity of the tear film.48,49 The liquid reservoir also helps to mask corneal irregularity secondary to dry eye and can significantly improve visual acuity in these patients. Patients with ocular GVHD successfully fit in scleral lenses often report substantial improvement in pain and photophobia associated with the disease.35,48
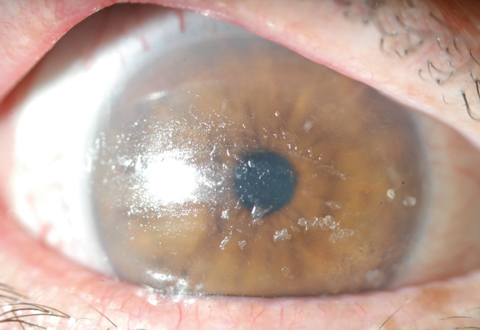 |
| Extreme poor wetting and lipid deposits in a GVHD patient. Photo: Karen G. Carrasquillo, OD,PhD |
Surgical Treatment
Patients with severe dry eye refractory to other treatment options can consider surgical management with tarsorrhaphy, amniotic membrane transplantation or punctal occlusion.26 Amniotic membranes can aid in epithelialization and prevention of fibrosis and inflammation.50,51 Research shows using sutureless amniotic membranes to treat moderate to severe dry eye significantly improves symptoms and ocular health for up to four months after treatment.51
Patients who do well with punctal plugs but experience recurrent extrusion may be good candidates for permanent occlusion with punctal cautery. In those with severe dry eye secondary to chronic GVHD, researchers found significant improvements in subjective symptom scores, Schirmer values, fluorescein and rose bengal scores and TBUT after thermal cauterization.52
Ocular GVHD is a complex and challenging condition to diagnose and manage. Because it can affect many ocular tissues, clinicians must properly identify all the ocular signs to determine the best treatment modality. Furthermore, early intervention is essential to reduce, or even prevent, the severe complications often associated with GVHD. A multidisciplinary approach is useful to determine when systemic, topical or other therapy options are best. These patients rarely return to a fully normal state due to lacrimal gland fibrosis and will need long-term treatment and follow up.
Dr. Spampinato is a staff optometrist at the Cincinnati VA Medical Center and an adjunct faculty member at The Ohio State University College of Optometry.
Dr. Hochwalt is a staff optometrist at the Cincinnati VA Medical Center and an adjunct faculty member at the Ohio State University College of Optometry and the Illinois College of Optometry.
1. Tabbara K, AlGhamdi A, Al-Mohareb F, et al. Ocular findings following allogenic hematopoietic stem cell transplantation (HSCT). Ophthalmology. 2009;116(9):1624-9. |

