9th Annual Retina ReportFollow the links below to read other articles from annual update on retina: Busted Barriers: Triaging Retinal Hemorrhages The Larry Alexander Resident Case Report Contest: Cloudy with a Chance of Retinopathy |
Infectious retinitis, while rare, can have dire visual consequences. Many causes exist, including viral, bacterial, tick-borne, parasitic and fungal. Successful management of these patients requires prompt recognition to quickly initiate treatment and refer, though the outcome may still be unfavorable. This discussion of the various etiologies can help you hone your differentials and properly diagnose these patients in a timely manner.
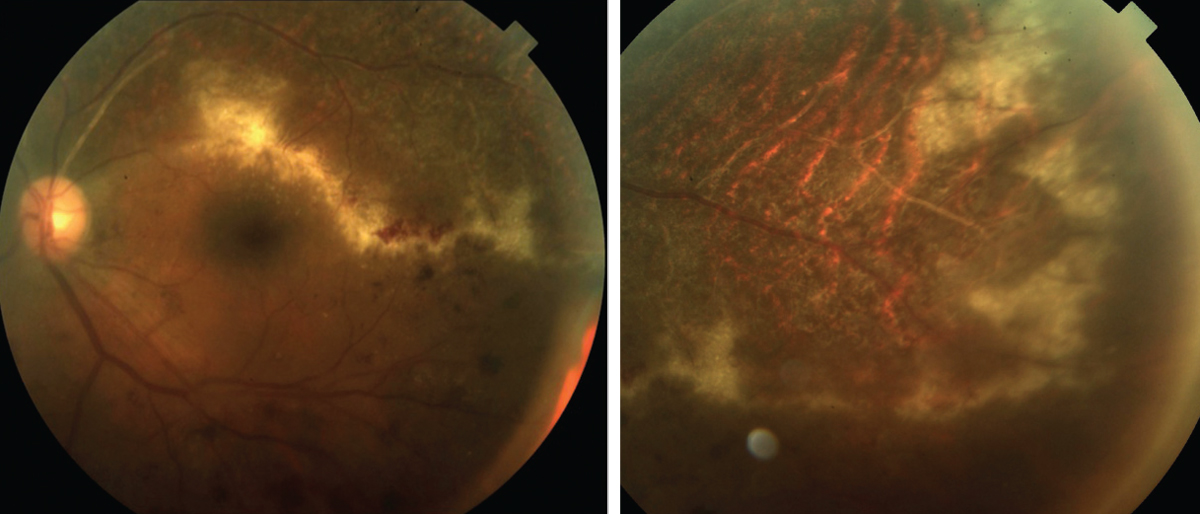 |
| Fig. 1. Along with the “ketchup and cottage cheese” appearance, CMV may advance like a “brush fire” or the tip of an ocean wave, leaving a necrotic retina. Click images to enlarge. |
Invasion of the Virus
Necrotizing herpetic retinitis describes a spectrum of infections caused by herpes viruses, mainly varicella zoster virus (VZV), herpes simplex virus (HSV) and cytomegalovirus (CMV).1,2 These conditions include acute retinal necrosis (ARN), progressive outer retinal necrosis (PORN) and CMV retinopathy.1,2 While separate conditions, they may represent a continuum whose clinical presentation depends on the patient’s immune status.1 These conditions are quickly progressive and can lead to devastating vision loss, even bilateral blindness.1,2,5
ARN. Patients with this condition may present with symptoms of blurry vision, floaters or ocular pain.4 It often presents with patches of retinal whitening and necrosis in the peripheral retina that extend posteriorly with time.5 Vitritis, anterior chamber cell, vasculitis and optic nerve involvement are common. Larger areas of necrotic retina increase the risk of retinal detachment (RD).5-7 Major causes of vision loss include RD, optic atrophy and retinal ischemia.6
ARN commonly presents in immunocompetent patients, but may also present in immunocompromised patients such as those with HIV/AIDS.8 Preceding signs of viral infection, such as cold sores, herpetic corneal disease, meningitis, encephalitis and disseminated shingles are common.6
Diagnosis is traditionally made based on clinical examination alone, often using the American Uveitis Society’s criteria: (1) One or more foci of discrete necrosis in the peripheral retina, (2) rapid progression without antiviral therapy, (3) arteritis and occlusive vasculitis, and (4) robust vitritis and anterior chamber reaction.9 Clinicians can also use polymerase chain reaction (PCR) to detect viral DNA and confirm infection—79% to 100% of ARN cases undergoing PCR testing revealed positive results for either VZV or HSV.8
PORN. This typically presents in immunocompromised patients, usually in those with HIV/AIDS whose CD4 count is less than 50cells/µl. Additional forms of immunosuppression, such as chemotherapeutic agents, are also risk factors.4 Patients with PORN often present with complaints of decreased peripheral vision or sudden vision loss. They may have floaters, but typically do not have pain.4
The condition presents with multiple regions of yellow-white retinal necrosis in either the posterior pole or peripheral retina that progress rapidly and coalesce. These regions initially affect the outer retina but eventually lead to full-thickness involvement. Hemorrhages may be present along with papillitis; however, a robust inflammatory response with dense vitritis and anterior chamber cell is not common. The necrotic retina becomes atrophic with a propensity to develop multiples holes, ultimately leading to RD in many patients.4
CMV. Present in its latent form in about 50% of the general population, this can lead to a severe necrotizing retinitis in patients who are immunocompromised.12,13 The condition usually presents in patients with CD4 counts of 50cells/L or less.13 CMV retinitis is the most common opportunistic ocular infection in patients with HIV/AIDS, although highly active antiretroviral therapy has significantly decreased the incidence of CMV retinitis in this population.13 However, more widespread use of systemic immunosuppressive agents for conditions such as autoimmune disease has also led to an increase in those without HIV/AIDS.12
Patients with CMV retinitis often present with blurred vision and floaters but may also be asymptomatic.14,15 The disease produces lesions of full-thickness retinal necrosis with a granular appearance (Figure 1). These lesions often occur along blood vessels and may be associated with significant retinal hemorrhaging, leading to descriptions such as “ketchup and cottage cheese.”14,15 Retinal vasculitis, optic nerve edema, vitritis and anterior uveitis may also be present. RD is common and is a leading cause of vision loss in patients with CMV retinitis.14
Diagnosis is chiefly based on clinical presentation.14,15 Most patients have a previous diagnosis of HIV/AIDS or history of immunosuppression. If not, testing is necessary. Serologic testing for CMV is available, but due to its high presence in the general population, it has little use. PCR testing may help to identify the virus.14,15
Using OCT, researchers describe CMV lesions as either full-thickness or cavernous necrosis.16 In full-thickness necrosis, there was thickening and hyperreflectivity of all retinal layers. The retinal pigment epithelium (RPE) was also thickened with a ruffling of its more anterior border, and the Bruch’s membrane was easily visible. In cavernous necrosis, hyperreflectivity in the inner retina with large empty spaces exists within the outer nuclear layer, leaving the RPE intact. OCT also revealed vitreous cell and thickening of the posterior hyaloid.16
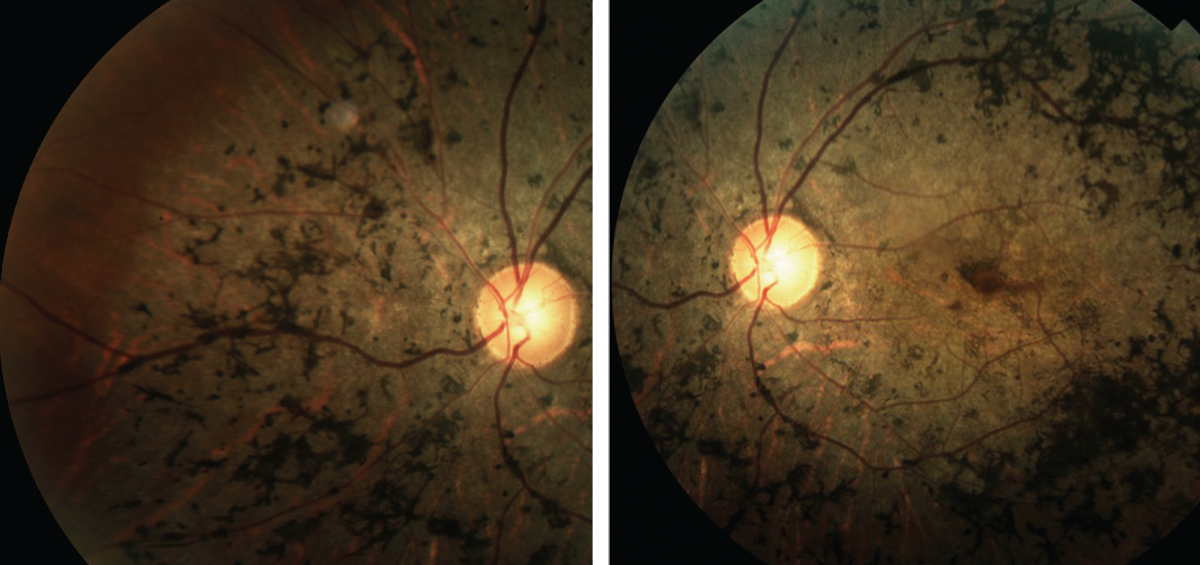 |
| Fig. 2. These images depict the classic presentation of syphilitic retinopathy. Click images to enlarge. |
Beware the Bugs
A bacterial infection can also cause retinal inflammation.
Syphilis. This is caused by Treponema pallidum.17 While ocular complications are uncommon, panuveitis is the most usual finding when they do occur.17,18 Others include interstitial keratitis, anterior and intermediate uveitis, scleritis, vasculitis, chorioretinitis, serous RDs, optic neuritis and optic nerve gumma, which are solitary inflammatory lesions (Figure 2).17-19
A few findings are considered distinctive, such as multiple, creamy-white preretinal precipitates in those with panuveitis.17,19 Involved retina tends to be mildly opacified compared with the dense retinal whitening seen in necrotizing herpetic retinitis.19 Necrotizing retinitis is a rare complication but can mimic ARN.19 An infrequent complication, acute syphilitic posterior placoid chorioretinitis, presents with deep oval plaques of retinal whitening.17,19 When imaged with OCT, these regions have outer retinal alterations including disruption of the ellipsoid zone, external limiting membrane and RPE as well as subretinal fluid.20 Congenital syphilis can lead to chorioretinitis with a salt and pepper appearance.
Serological testing is necessary for diagnosis and is divided into treponemal and non-treponemal tests. Treponemal tests include T. pallidum hemagglutination assay and fluorescent treponemal antiboday-absorption. These labs remain positive for life and cannot be used to determine disease activity and treatment efficacy, but they have a higher specificity than non-treponemal tests. The latter include venereal disease research laboratory test and rapid plasmin reagin test, both helpful for monitoring disease activity and treatment efficacy.17,19
Cat scratch disease (CSD). Caused by Bartonella henselea, this is the most common cause of neuroretinitis and is typically benign and self-limited.21,22 The bacteria is harbored in cats and kittens, and can be acquired in humans through a scratch or bite. However, the bacteria can also be transmitted by dogs, fleas and may even have unknown vectors of transmission.22
Systemic symptoms such as lymphadenopathy, fever and malaise typically occur prior to ocular findings, but can be coincident as well.21,22 Not all patients with ocular complications will report systemic symptoms.22
Posterior segment findings, either bilateral or unilateral, are variable.22 Many reports cite optic disc edema with macular star (neuroretinitis) as the most common finding, while others describe focal areas of either superficial or deep retinal whitening (Figure 3).22,23 Additional findings include optic nerve edema without macular star, vitritis, macular edema, serous detachments, hemorrhage, optic nerve granuloma and angiomatous lesions.21-25 Presence of macular star may initially be absent and form post-resolution of macular edema.22
Fluorescein angiography may reveal optic nerve leakage and hyperfluorescence of retinal lesions.24,25 Clinicians should demonstrate the extent of vascular nonperfusion in occlusive events to help determine visual prognosis and the likelihood of secondary complications such as neovascularization.24 Peripapillary telangiectasia and angiomatous lesions may be present.23,24,26 OCT can be a helpful tool to document and monitor macular edema, serous detachments and optic nerve edema.26
Ocular diagnosis is based on fundus findings, but readily available serological testing for Bartonella henselea should be obtained to help confirm the diagnosis.
Tuberculosis. This is caused by Mycobacterium turberculosis and can have ocular involvement, even in isolation without pulmonary manifestations.32 All forms of uveitis are possible as well.32
The most common posterior finding is multiple choroidal tubercles, which look like deep, ill-defined, round, gray or yellowish lesions, possibly having overlying serous detachments.32,33 Larger choroidal granulomas may be present, often mimicking choroidal tumors.32,33 Choroidal involvement can also occur in the form of serpiginous-like choroiditis. This appears as multifocal plaque-like lesions in the peripapillary region that spread circumferentially. Additional findings can include retinal vasculitis (typically affecting veins more than arteries), neuroretinitis and endophthalmitis.32,33
Significant challenges exist in the diagnosis of ocular tuberculosis. Cultures and PCR testing have low yields, and biopsy is often impossible.34 Frequently, diagnosis relies on the clinical picture, tuberculin skin test, chest X-ray and, sometimes, response to treatment.34 The utility of interferon-γ release assays, such as the Qauntiferon-TB Gold (Qiagen) test, is still uncertain.35
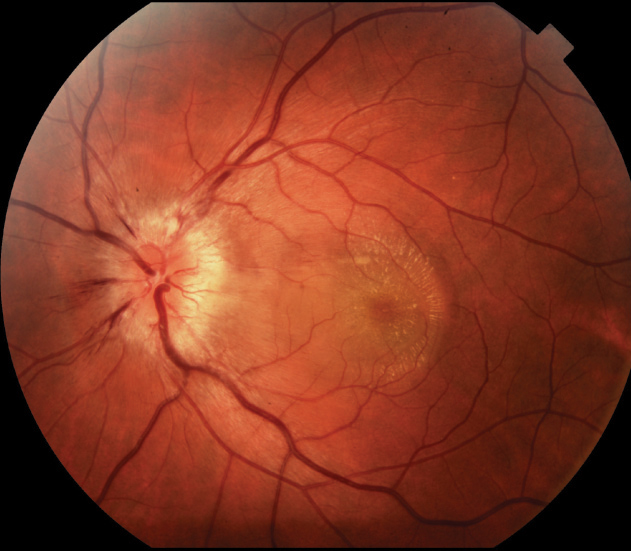 |
| Fig. 3. Neuroretinitis can present with a macular star formation, as seen here. |
Out of the Woods
Tick-borne diseases (TBDs) are often underdiagnosed, for several reasons. First, more than 75% of patients don’t remember having a tick bite.36 Second, signs such as arthralgia, skin involvement and neurological changes may be consistent with those of autoimmune diseases.37 Patients with TBDs may first present to their eye care provider due to ocular symptoms.
Lyme disease. The most common TBD in the United States, this disease had 27,000 confirmed cases in 2013.38 Borrelia burgdorferi spirochete is the causative agent in the United States, while B. garinii and B. afzelii prevail in Europe.40 Transmission occurs via the deer tick in the east and the Western black-legged tick in the west.41 Fortunately, the risk of contracting Borrelia from an infected tick is between 1% and 3%.39
In the early stage of Lyme disease, up to 80% of patients will have erythema migrans, or a bulls-eye rash roughly eight days post-infection.37,41 A flu-like illness and lymphadenopathy may accompany the rash, and ocular involvement, if any, is confined to follicular conjunctivitis and mild photophobia.40
Stage two (three to 10 weeks after inoculation) has the most ophthalmic changes. Ocular manifestations include optic neuritis, uveitis, choroiditis, venous occlusion, cotton wool spot and neurological pupil abnormalities.36,40 Keratitis is the most common finding in stage three disease, which occurs months to years later.
In patients suspected of exposure, an enzyme immunoassay should be ordered, and if positive, an immunoblot test. Because Lyme testing may be negative within the first few weeks of the disease, serological testing can be deferred if the clinical picture correlates with the disease and the patient portrays the bulls-eye rash.36
Rocky Mountain spotted fever (RMSF). This is a life-threatening disease caused by Rickettsia. The primary vector in most of the United States is the American dog tick. However, in the Rocky Mountain area and Canada, it is the Rocky Mountain wood tick. The brown dog tick, which mainly affects Mexico, has been implicated in eastern Arizona.42 The gram-negative bacteria targets the lining of small and medium-sized blood vessels, which leads to the triad of “Rickettsial vasculitis”—vascular inflammation, blood vessel damage and altered vascular permeability.37 Ocular findings include retinal hemorrhage (about 50% of RMSF have thrombocytopenia), retinal artery or vein occlusion, vascular sheathing, optic neuritis and neuroretinitis.36,40
Signs and symptoms include fever, nausea, headache, vomiting and possibly severe abdominal pain—especially in children—which may lead to an incorrect gastroenteritis diagnosis.43 The hallmark sign is the spotty rash, which starts around the wrist and ankles. Diagnosis relies more on clinical findings, as laboratory testing, such as a skin biopsy, is difficult to yield positive results.37 Also, given the severity of the disease, treatment should be initiated regardless of a negative test.
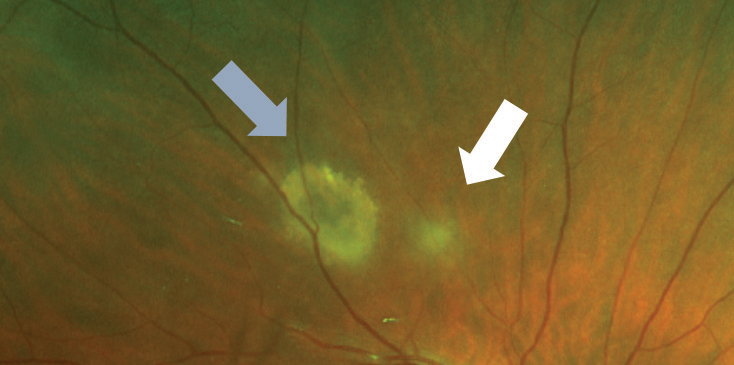 |
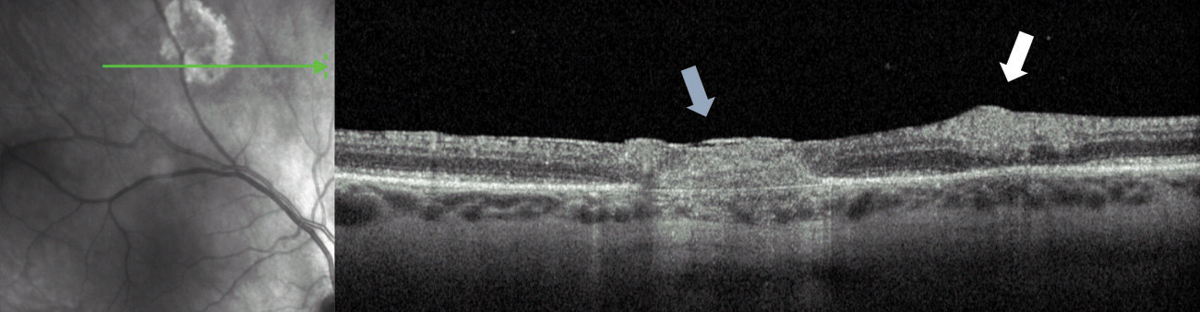
| Fig. 4. This resolving toxoplasmosis lesion (blue arrow) has a new region of active infection (white arrow). Note that the older lesions involve all retinal layers, whereas the early ones only affect the inner retina. |
 |
Unwilling Hosts
Other retinitis cases can be linked to any number of parasites:
Toxoplasmosis is the leading cause of posterior uveitis, and is due to Toxoplasma gondii—a ubiquitous human parasite.44 Infection occurs by ingestion of contaminated soil or tissue cysts in undercooked meat (pig and sheep).45,46 Ocular findings include retinochoroiditis, retinal vasculitis, branch retinal vein occlusion and anterior and intermediate uveitis.45 The complaint of “headlights in the fog” is the common presenting sign. A complication of infection is increased intraocular pressure, which may differentiate this process from other possible causes of retinochoroiditis.47
Acute infection leads to a superficial retinitis and vitritis. OCT imaging may show a thickened and hyperreflective inner retina, edema, subretinal fluid, vitreous cell and thickening of the posterior hyaloid. The condition can progress into deep layers, leading to chorioretinal scarring (Figure 4).
Diffuse unilateral subacute neuroretinitis (DUSN) results from nematode infestation, presumably in the subretinal space, and mostly afflicts healthy children and young adults.48 DUSN should always be suspected when a patient of this demographic presents with unilateral vitritis, retinal vasculitis, optic neuritis and outer retinal/inner choroid multifocal lesions—the early stage of the disease.49 Late-stage changes include atrophy of the retinal nerve fiber layer, inner and outer retina and optic nerve.48 Unfortunately, clinical visualization of the worm occurs in only 25% to 40% of patients.50 However, researchers have seen the worm’s location using OCT angiography.50
Toxocariasis, caused by ingesting Toxocara larvae from foods soiled with dog or cat feces, may lead to a unilateral ocular presentation of peripheral or posterior granuloma or panuveitis with no distinct nodular lesion.51 Children are especially at risk for exposure given their hygiene habits and the contamination rates (as high as 40%) of play areas.51 Only a few adult cases have been reported.52
 |
| Fig. 5. This patient presented with with the OHS triad: PPA, multiple punched out lesions and early development of CNV. Click image to enlarge. Image: Mohammad Rafieetary, OD |
Fear the Fungus
Given the choroid’s high blood flow, most fungal lesions begin as a choroiditis.54 Fortunately, research of adult and pediatric patients with fungemia shows the eye has less than 1% of fungal dissemination, possibly due to prompt initiation of antifungal treatment in clinically suspicious cases, even before culture results.55
Candida. This is the most common healthcare-associated bloodstream infection, with at least 50% of cases occurring in the intensive care setting.56 Community-acquired risk factors include an immunocompromised state, intravenous (IV) drug use, diabetes, catheter placement and broad-spectrum antibiotic use.56 Direct inoculation from ocular trauma or surgery can occur.57 Ocular manifestations include white fluffy chorioretinal lesions, focal vitritis and iridocyclitis.
Researchers described two patterns of OCT findings in Candida endophthalmitis—chorioretinal and intraretinal—which they believe represent the site of ocular invasion. The chorioretinal pattern includes the rain-cloud sign (preretinal hyperreflective dense masses with a strong shadowing effect) and the sawtooth-shaped sign (vertically oriented hyperreflectivity bands adjacent to the lesion). The less indolent intraretinal pattern (hyper-reflective sphere) spared structures beyond the inner nuclear layer.58
Aspergillus. The vast majority of exogenous cases are molds (87%), and the most common agent is Aspergillus.59 It grows in soil and decaying vegetation with air-borne spores, which place the immunocompromised, those with pulmonary disease and IV drug use at risk. Macular infiltration is common, and patients often present with a painful decrease in visual acuity. Other signs include vascular occlusion, retinal necrosis and retinal hemorrhages.57
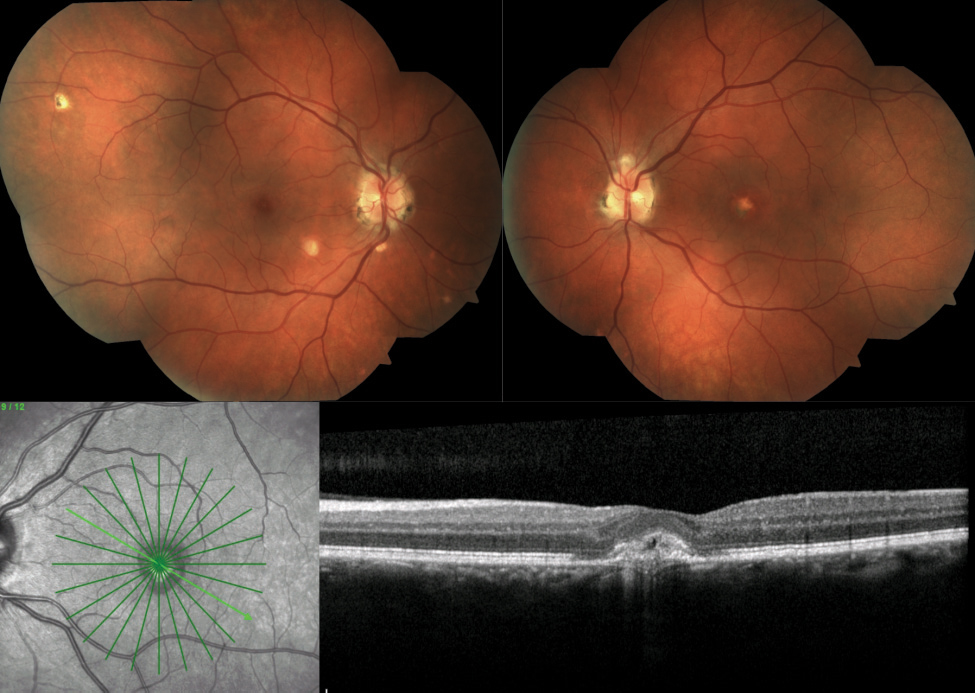
Ocular histoplasmosis syndrome (OHS). A mild and transient pulmonary infection in otherwise healthy people, this is caused by Histoplasma capsulatum or H. duboisii.60 The Mississippi and Ohio river valleys form the “histo belt.” Clinically, OHS appears as peripapillary atrophy (PPA), “histo spots” or “punched out” chorioretinal scars in the macula and midperiphery and choroidal neovascularization (CNV) or its disciform scar sequelae (Figure 5).61
Diagnosis of OHS is based on observation, as skin testing may induce flare-ups. Two of the three ocular findings listed above should be met.61 Smoking is a significant risk factor for CNV development.62 In addition, one study found that the presence of PPA and macular histoplasmosis spots invoked a respective 4% and 25% risk of vision loss to the fellow eye.62
Retinitis, although a rare condition, highlights the importance of a thorough funduscopic examination, even in asymptomatic patients. With myriad etiologies on the differential, knowing the classic presentation of each is crucial to narrowing down the diagnosis and initiating treatment as early as possible. Management depends on the entity and can range from in-office monitoring to an infectious disease or retina specialist referral. Despite prompt diagnosis, positive outcomes are not always possible. For those cases, ODs should employ low vision tactics to enhance patients’ remaining vision and help them maintain functional independence.
Dr. Williamson is the residency supervisor at the Memphis VA Medical Center and is adjunct faculty at multiple optometry schools.
Dr. Haynes is a clinical optometric fellow at Charles Retina Institute in Germantown, Tenn.
1. Guex-Crosier Y, Rochat C, Herbort CP. Necrotizing herpetic retinopathies. A spectrum of herpes virus-induced diseases determined by the immune state of the host. Ocul Immunol Inflamm. 1997;5(4):259-65. |

