Endophthalmitis can be subdivided into two forms: exogenous and endogenous.6,7 If the affecting organism breached the external ocular barrier as a result of surgery, trauma or an extension of corneal infection into the eye, it is classified as exogenous endophthalmitis.8 Endogenous endophthalmitis, on the other hand, is caused by a hematogenous (i.e., blood-borne) dissemination of the affecting pathogen.4,9 When the blood-ocular barrier becomes compromised, it creates circumstances that predispose the patient to endogenous infection; presentations can range from chorioretinitis alone to chorioretinitis with extension into the vitreous.4,10
Here, we review the case of a patient who presented with endogenous endophthalmitis secondary to Candida albicans––a fungal pathogen that’s frequently linked to a variety of oral and genital infections. Considering the potential severity of the presentation, we worked closely with two medical specialists to ensure the best possible visual prognosis.
History
A 74-year-old white female presented with a chief complaint of difficulty seeing with her left eye. She stated that her vision was blurry for six days, and believed that it was worsening. Her medical history was positive for hypertension, hyperlipidemia, vein stripping, hemorrhoidectomy, colon polyps, corticobasal degeneration, anxiety and osteoporosis. Ocular history included bilateral phacoemulsification cataract extraction––five years prior for the right eye and two years prior for the left eye.
The patient mentioned a recent, three-month hospital stay for an elective endoscopy, during which her esophagus inadvertently ruptured. She underwent surgery with thoracotomy, debridement of esophageal tissue, rupture repair and ligation of the thoracic duct. Treatment during the hospitalization period also included antibiotics for the esophageal rupture, urinary tract infections and an infected sacral pressure ulcer. Additionally, total parenteral nutrition was used for an extended duration to facilitate esophageal healing.
She also developed thrush while hospitalized. Subsequent blood cultures tested negative for yeast. One month after her release, the patient underwent a CT scan of the chest, which revealed pulmonary emboli. She had since been taking Coumadin (warfarin sodium, Bristol-Myers Squibb). The patient denied a history of smoking or recreational drug use; however, she noted occasional alcohol consumption.
Diagnostic Data
On examination, the patient’s best-corrected visual acuity measured 20/25 OD and 20/70 OS. (In an examination six months earlier, her left eye yielded a best-corrected visual acuity of 20/25.) The pupils were equally round and reactive to light and accommodation. Confrontation visual fields were full to finger counting. Her extraocular motilities were full in both eyes. Intraocular pressure measured 14mm Hg OU.
|
Predisposing Systemic Conditions and Risk Factors for Endogenous Endophthalmitis3-5,8,9,16,17
| |
|
• Diabetes mellitus
• Gastrointestinal disease • Infection • Cardiac diseases • Urological diseases • Hemodialysis • Malignancy • Immunosuppressive therapy/AIDS • Intravenous drug abuse • Chemotherapy • Steroid therapy • Antimicrobial drug therapy • Iatrogenic (i.e., dental surgery) • Septicemia • Liver abscess |
• Soft tissue abscess
• Pneumonia • Long-term parenteral feeds • Indwelling central venous catheter • Neutropenia • Recent surgery • Pregnancy • Massive trauma • Alcoholism • Hepatic insufficiency • Post-partum • Prematurity • Genitourinary manipulation • Endocarditis • Contaminated intravenous fluids |
An examination of the ocular adnexa was negative for suspicious pathology. Anterior segment examination revealed a clear cornea, quiet conjunctiva, clear aqueous humor and well-placed posterior chamber intraocular lens implants OU. A dilated fundus examination revealed healthy optic nerves, with a 0.3 x 0.3 cup-to-disc ratio in both eyes.
We determined that the right eye was negative for retinal pathology; however, the left eye exhibited grade 1+ vitreous cells and a hazy vitreous media that was concentrated anteriorly. These findings largely prohibited a clear view of the patient’s left macula. The media presented in a U-shaped pattern of yellow-white, connected spheres that closely resembled a string of pearls. The peripheral retinal exam was negative for pathology.
Due to the decreased visual acuity, unknown media in the vitreous and lack of macular visualization, we immediately referred the patient a retina specialist for consultation. In addition to these findings, the retinologist noted that the inflammatory aggregates were located in the posterior vitreous, and confirmed that the macula was attached. The patient was then scheduled for a diagnostic and therapeutic vitrectomy of her left eye.
|
Suggestive Causes of Endogenous Endophthalmitis18-21
|
|
|
• Transplacentally in newborns
• Fungal toenail infection • Oropharyngeal candidiasis secondary to inhaled steroid • Onychomycosis and vaginal candidiasis |
Diagnosis
A preliminary lab report showed “very light-growth yeast isolated.” However, the final report more specifically revealed “very light-growth Candidia albicans isolated.” We diagnosed the patient with fungal endogenous endophthalmitis.
A yeast susceptibility report was also performed. Then, we tested the following antifungals: anidulafungin, micafungin, caspofungin, 5-flucytosine, posaconazole, voriconazole, itraconazole, fluconazole and amphotericin B. Fortunately, C. albicans was shown to be susceptible to all aforementioned agents.
Treatment and Follow-up
Following a consult with an infectious disease specialist, we initially placed the patient on voriconazole. Ten days later, we switched her to 400mg fluconazole PO QD, after the final culture tested positive for C. albicans. The patient received antifungal therapy for a total of six weeks. Because fluconazole can increase the efficacy of anticoagulants, the patient’s international normalized ratio was monitored weekly and the Coumadin dosing was adjusted. A precautionary echocardiogram was ordered to assess the heart valves for possible endocarditis. This proved to be negative.
The patient was closely monitored and showed steady chorioretinitis resolution (figures 1-3). Five months after initial presentation, the patient’s chorioretinitis had resolved completely, and her visual acuity returned to 20/25 OS (figure 4).
Discussion
Endogenous endophthalmitis is a potentially blinding condition, and is usually associated with a poor visual prognosis.11,12 The condition accounts for just 2% to 15% of all cases of endophalmitis, and almost exclusively presents unilaterally.1-3 Postoperative endophthalmitis is responsible for more than 70% of cases, with an overall incidence of 0.81% in cataract procedures.13-15
| |
| |
| |
|
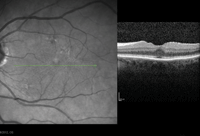
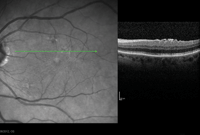
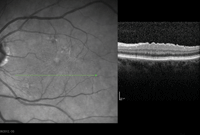
1-3. Optical coherence tomography (OCT) scan of our patient's left eye shows chorioretinitis of the fovea, superior macula and inferior macula three months after initial diagnosis.
|
A few underlying conditions or risk factors for endogenous endophthalmitis include diabetes mellitus, malignant tumor, gastrointestinal disease, immunosupression and intravenous drug use (see “Predisposing Systemic Conditions and Risk Factors for Endogenous Endophthalmitis” and “Suggestive Causes of Endogenous Endophthalmitis”).3,5,8,9
In patients with endogenous endophthalmitis, the choroid and retina appear highly vascular.10 Both components are separated from the intraocular components by the blood-ocular barrier, which is composed of the retinal pigment epithelium and Bruch’s membrane.10 Candidemia affects the choroid first, and there may be minimal or no initial vitritis.8 Infection by Candida species will manifest as choroditis or chorioretinitis, and usually is painless.8
Unless the infection site is near the macula, the patient is likely to be asymptomatic at this stage.8 Vitritus develops as inflammation escalates.8
This is the stage of progression that our patient reached when she noted a decrease in her visual acuity and decided it was time to undergo an examination. It is possible for extensive inflammation to involve the aqueous humor—if so, eye pain would likely be a presenting symptom.8
Although valuable information was gleaned from this case study, a number of unanswered questions remained. The etiology is unclear because the patient presented with multiple risk factors, such as parenteral feeds, antimicrobial drug therapy and recent surgery.
Also of note, the patient’s blood cultures were negative for yeast during her hospitalization.
Several factors were crucial to the success of the care given to this patient, such as a prompt referral to the retina specialist and the decision to work closely in conjunction with an infectious disease specialist. Additionally, the laboratory cultures helped us select the most effective treatment for the offending pathogen. Multiple reports suggest that an early vitrectomy could reduce the infectious load and aid in the intraocular diffusion of systemic antifungals, as well as reduce the risk for secondary retinal detachment.22,23
There is no specific protocol to follow with such cases, likely due to the rarity of the condition. The current stage and severity of any endopthalmitis at the time of detection largly dictates the management course; however, most studies suggest early, aggressive treatment.24
Currently, one study and one case report indicate that an early vitrectomy can preserve vision––but most published cases of endogenous endophthalmitis result in a very poor visual prognosis, despite early vitrectomy and intravitreal injections.12,24,25
|
|
|
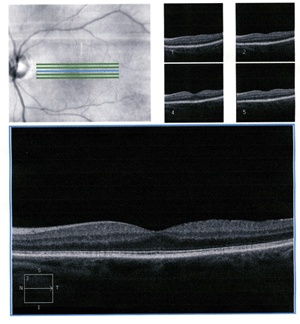
4. Five minutes after diagnosis, the OCT shows resolved chorioretinitis OS.
|
Literature specifically focused on endogenous endophthalmits secondary to C. albicans advises that patients with chorioretinitis alone, or chorioretinitis accompanied with mild vitritis, will respond to systemic antifungal therapy.5,8,10 However, for cases that exhibit moderate to severe vitritis, these reports recommend vitrectomy and intravitreal amphotercin or voriconazole in addition to systemic antifungal therapy.5,8,10 The intravitreal administration of medication was not performed in this case, as the vitreous sample had to be sent to the lab for culturing.
Candida albicans is the most common cause of endogenous endophthalmitis, which occurs most frequently in immunocompromised patients, and causes up to 92% of all candidiasis.24,26-28 When C. albicans is the isolated pathogen, a better visual outcome is expected—especially when compared to all other types of endogenous endophthalmitis.2,29,30 The fact that our patient’s immune system was intact may have contributed to her successful treatment.
Endophthalmitis is considered a true medical emergency. Various disease subtypes caused by multitude of affecting pathogens often yield a variable clinical presentation. So, it is important for clinicians to study the numerous ways that endophthalmitis can develop to ensure that prompt action is taken. In our patient’s case, she only exhibited ocular signs of endogenous endophthalmitis. But keep in mind that other patients also may present with a host of systemic symptoms secondary to the underlying infection.
Dr. Andrud is in private practice at Eye Microsurgery Ltd. in St. Louis.
1. Jackson TL, Eykyn SJ, Graham EM, et al. Endogenous bacterial endophthalmitis: a 17-year prospective series and review of 267 reported cases. Surv Ophthalmol. 2003;48(4):403-23.
2. Connell PP, O’Neill EC, Fabinyi D, et al. Endogenous endophthalmitis: 10-year experience at a tertiary referral centre. Eye (Lond). 2011;25(1):66-72.
3. Okada AA, Johnson RP, Liles WC, et al. Endogenous bacterial endophthalmitis: report of a ten-year retrospective study. Ophthalmol. 1994;101(5):832-8.
4. Zenith HY, Rose PS, Fiona OJ, et al. Review of clinical features, microbiological spectrum, and treatment outcomes of endogenous endophthalmitis over an 8-year period. J Ophthalmol. 2012(265078):1-5.
5. Flynn HW Jr. Endophthalmitis: diagnosis, treatment, prevention In: Spaeth GL (ed.) Ophthalmic surgery: principles and practice. Elseiver Science, Philadelphia; 2003:663-77.
6. Shrader SK. The clinical spectrum of endophthalmitis: incidence, predisposing factors, and features influencing outcome. J Infectious Disease. 1990;163(1):115-20.
7. Chee SP, Jap A. Endogenous endophthalmitis. Current Opinion in Ophthalmol. 2001;12(6):464-70.
8. Durand ML. Endophthalmitis. CMI 2013;19(3):227-34.
9. Hamada Y, Okuma R, Yusuke K, et al. Bibliographical investigation (domestic and overseas) on the treatment of endogenous Candida endophthalmitis over an 11-year period. Med Mycol J. 2013;54:53-67.
10. Riddell J IV, Comer GM, Kauffman CA. Treatment of endogenous fungal endophthalmitis: focus on new antifungal agents. CID. 2011;52(5):648-53.
11. Kah TA. Disseminated fusariosis and endogenous fungal endophthalmitis in acute lymphoblastic leukemia following platelet transfusion possibly due to transfusion-related immunomodulation. BMC Ophthalmol. 2011;30(11):1-4.
12. Ishii K, Hiraoka T, Kaji Y, et al. Successful treatment of endogenous Klebsiella pneumonia endophthalmitis: a case report. Int Ophthalmol. 2011;31(1):29-31.
13. American Academy of Ophthalmology. Basic and clinical science course. Section 9. Intraocular inflammation and uveitis, 2001-2002. Presented at the Annual American Academy of Ophthalmology Meeting. San Francisco; 2001.
14. Doft B. Endophthalmitis management. Focal points: modules for clinicians. Presented at the Annual American Academy of Ophthalmology Meeting. San Francisco; 1997.
15. Aaberg TM Jr, Flynn HW Jr, Newton J. Nosocomal acute-onset postoperative endophthalmitis survey: a 10-year review of incidence and outcomes. Ophthmol. 1998;105:1004-10.
16. May DR, Peyman GA, Raichand M, et al. Metastatic Peptostreptococcus intermedius endophthalmitis after a dental procedure. Am J Ophthmol. 1978;85(5):662-5.
17. Gupta A, Gupta V, Dogra MR, et al. Fungal endophthalmitis after a single intravenous administration of presumably contaminated dextrose infusion fluid. Retina. 2000;20(3):262-8.
18. Basu S. Neonatal endogenous endophthalmitis: a report of six cases. Pediatrics. 2013;131(4):1292-97.
19. Chavan R. A case of Candida albicans endophthalmitis with no predisposing risk factors and a distant source of infection. Case Rep Ophthalmol. 2012;3(3):277-82.
20. Stanbury RM. Endogenous Candida endophthalmitis with no apparent predisposing factors. Eye. 1998;12:321-3.
21. Kostick DA, Foster RE, Lowder CY, et al. Endogenous endophthalmitis caused by Candida albicans in a healthy woman. Am J Ophthalmol. 1992;113:593-4.
22. O’Day DM. Intraocular penetration of systemically administered antifungal agents. Curr Eye Res. 1985;4:131-4.
23. Louie A, Liu W, Miller DA, et al. Efficacies of high-dose fluconazole plus amphotericin B and high-dose fluconazole plus 5-fluorocytosine versus amphotericin B, fluconazole, and 5-fluorocytosine monotherapies in treatment of experimental endocarditis, endophthalmitis, and pyelonephritis due to Candida albicans. Antimicrob Agents Chemother. 1999;43:2831-40.
24. Novosad BD, Callegan MC. Severe bacterial endophthalmitis: towards improving clinical outcomes. Expert Rev Ophthalmol. 2010;5(5):689-98.
25. Yoon YH, Lee SU, Sohn JH, et al. Result of early vitrectomy for endogenous Klebsiella pneumonia endophthalmitis. Retina. 2003;23(3):366-70.
26. Hamza HS. Fungal retinitis and endophthalmitis. Ophthalmology Clinics of North America 1999; 12:89-108.
27. Sallam A, Taylor SR, Khan A, et al. Factors determining visual outcome in endogenous Candida endophthalmitis. Retina. 2012;35:1129-34.
28. Lingappan A. Endogenous fungal endophthalmitis: causative organisms, management strategies, and visual acuity outcomes. Am J Ophthalmol. 2012;153:162-6.
29. Ness T, Pelz K, Hansen LL. Endogenous endophthalmitis: microorganisms, disposition and prognosis. Acta Ophthalmologica Scandinavica. 2007;85(8):852-6.
30. Zhang H, Liu Z. Endogenous endophthalmitis: a 10-year review of culture-positive cases in northern china. Ocular Immunol Inflam. 2010;18(2):133-8.

